We describe transplantation surgical technique and analyse gray scale, colour and spectral Doppler findings in cases with normal post transplantation renal function: examination includes transplant length, cortical thickness, parenchymal morphology and possible fluid collections. Colour Doppler assessment of arterial and venous anastomoses and spectral analysis of main and intrarenal arteries are also performed. Resistive Index and Peak Systolic Velocity values are obtained. We present the commonest complications (acute tubular necrosis, acute and chronic rejection, infection, renal artery or vein stenosis and thrombosis, infarction, collecting system obstruction, arteriovenous malformations and pseudoaneurysms, fluid collections). A step by step protocol assures inclusion of all parameters that should be assessed in the examination.
Discussion
Renal transplantation has been the treatment of choice for end stage renal failure since the 1960s. It is achievable with cadaveric or living related donor transplants. The latter results in a 90% 2 year graft survival, as well as low recipient morbidity. It is therefore the preferred option [1]. On the other hand, cadaveric transplantation has a 5 year survival of 65%.
Imaging, especially ultrasound, has a key role in the selection of living donors. They are examined preoperatively in order to ensure the transplant kidneys satisfying condition and to exclude any major patient pathology present. Recipients are similarly examined with ultrasound to assess the iliac vessels patency.
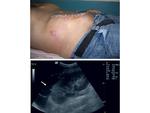
The transplant kidney is placed extraperitoneally in the right iliac fossa. The renal artery is anastomosed end-to-end to the internal iliac artery or via an end-to-side anastomosis of an aortic patch on to the externac iliac artery (cadaveric transplants). The renal vein is anastomosed end-to-side with the iliac vein. The urinary tract reconstruction is performed by antireflux ureteroneocystostomy. Ureter length is kept short in order to avoid ischaemia. Anatomic variations present additional problems.
Immediate post surgery graft dysfunction causes are non mechanical (acute tubular necrosis, dead donor kidney, acute rejection, cyclosporine nephrotoxicity) and mechanical (occlusion or stenosis of the renal artery, pseudoaneurysms, arteriovenous fistulas, renal vein thrombosis, urinary obstruction, fluid collections) [1].
According to a different classification, complications can be categorised into vascular complications, complications related to the collecting systems (urological complications), perinephric fluid collections and parenchymal complications. Additionally, there are some complications that may occur secondary to a transplant renal biopsy [2].
Ultrasound is very useful for detecting vascular complications (early occlusions, arterial stenosis, perinephric complications, hydronephrosis etc). All these are clinically significant complications that affect management. Ultrasonography can also detect many of the late acquired diseases, especially intercurrent tumours that require surgery. It is also the best method to guide interventions (collection aspiration, nephhrostomy drain insertion). However, it fails to discriminate between important causes of early graft dysfunction (acute tubular necrosis, rejection, drug toxicity): these distinctions still rely on biopsy [3].
An examination protocol includes:
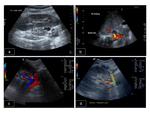
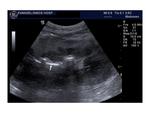
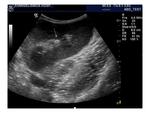
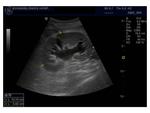
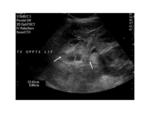
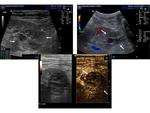
B mode examination of the transplant kidney: Renal size is measured. Normal echogenicity with corticomedullary differentiation is suggestive of normal function. Increased echogenicity throughout the kidney may be a sign of rejection. Perinephric collections are also located in order to assess their size, possible increase or decrease in fluid quantity and ensure correct drainage catheter placement. Besides the immediate perinephric area, all parts of the abdomen should be examined for collections. The kidney is imaged for pelvicalyceal dilatation, stones or masses present.
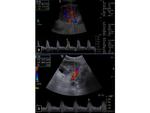
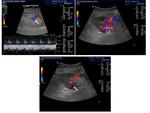
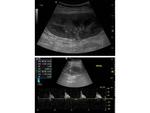
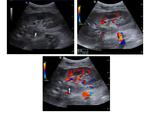
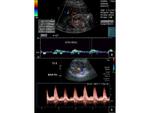
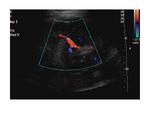
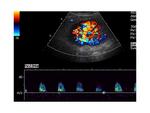
Colour and Spectral Doppler is used to evaluate perfusion. Iliac and renal vessels are checked for patency and normal waveforms present. Renal artery stenosis criteria used for native kidney evaluation also apply in transplants: The main renal artery is imaged in a transverse or oblique level, ideally at its origin from the external iliac artery and its course towards the renal hilum. The waveform is normally of a splanchnic, low resistance type. The peak systolic velocity is calculated. If higher than 180-200cm/sec, it is suggestive of stenosis [4-8]. Intrarenal arterial branches are also evaluated, with special attention to the systolic acceleration time and the Resistive Index (RI). Findings suggestive of significant renal artery stenosis is a slowly increasing velocity with a low peak value (tardus parvus waveform). Elimination of the early systolic peak, as well as an RI less than 0.5 are also consistent with the same entity. In particular, a stenosis higher than 70% [8] is connected to a slow increase to the peak systolic velocity (tardus), the acceleration time being longer than 0.07 sec, while the peak systolic velocity is low (parvus) [4, 6, 8].
Finally, the use of echo enhancers has potential to diagnose acute kidney graft rejection in its early stages [9], image mass-like lesions, as well as elucidate cases with suspected ischaemia.