We describe and illustrate the role of imaging techniques (computed tomography -CT-,
whole body positron emission tomography -PET-,
integrated PET-CT and magnetic resonance imaging -MRI-) in the diagnosis,
staging and follow-up of NSCLC,
highlighting their indications,
limitations,
and complementary function in oncologic imaging.
IMAGING TECHNIQUES
All patients with suspected NSCLC should have a history and physical examination performed,
as well as laboratory testing and imaging techniques.
Imaging aims to accomplish three goals: a) determine the disease stage; b) identify lesions that warrant tissue sampling; and c) assist in planning surgical therapy,
radiation therapy (RT) or chemotherapy (ChT).
Abnormal findings on imaging tests should be confirmed by tissue sampling,
as noninvasive techniques,
such as imaging alone,
are insufficient.
It should be emphasized that sampling the primary tumor is diagnostic only.
In contrast,
sampling abnormalities that may be related to metastatic sites can be diagnostic and provide additional staging information.
In general,
the sampling target should be the lesion that will establish the highest disease stage assuming that the lesion is not inaccessible or particularly high risk.
Sampling both the primary tumor and the potentially metastatic lesion is occasionally required.
Selection of a sampling technique should take tumor location,
safety,
availability,
ease,
local expertise,
diagnostic accuracy and patient preference into consideration (imaging-guided percutaneous needle aspiration or biopsy,
endobronchial ultrasound-guided forceps biopsy ,
conventional flexible bronchoscopy with forceps biopsy,
blind transbronchial fine needle aspiration,
or both,
electromagnetic navigational bronchoscopy-guided forceps biopsy …).
Following successful tissue sampling,
there is usually enough information to confirm NSCLC and determine the disease stage according to the TNM staging system for NSCLC.
An exception to this approach applies to patients who are good surgical candidates,
have a well defined primary lesion,
and whose noninvasive evaluation found no evidence that the disease has spread beyond the primary lesion.
Such patients may undergo surgical resection of the lesion without prior tissue sampling.
In this situation,
NSCLC is confirmed and staged after the histopathology from the surgical resection is known.
The rationale for this approach is that,
in such patients,
the benefit of early resection (ie,
cure) outweighs both the surgical risks and the possibility of false-negative or false-positive clinical-diagnostic staging.
Contrast-enhanced CT that extends through the lungs,
liver,
and adrenal glands is ideal for TNM staging,
as will be discused below.
Based upon this initial evaluation,
most patients require additional imaging.
This may include whole body PET,
integrated CT/PET,
radionuclide bone scintigraphy,
MRI of the chest wall or brain,
and/or CT of the brain.
Each of these modalities and a recommended approach to imaging patients with suspected NSCLC are reviewed separately.
CHEST RADIOGRAPHY
Despite their limited utility in staging,
chest radiographs have an important role in the assessment of a lung nodule or mass.
Comparing a current chest radiograph to prior radiographs is often the only way to determine whether a lung lesion is new,
enlarging,
or stable,
since patients are more likely to have had a prior chest radiograph than any other type of chest imaging.
A new or enlarging lesion is more likely to be lung cancer than a lesion whose size and appearance have been stable for years.
Lesions that are malignant tend to have a doubling time between approximately 20 and 400 days.
Benign lesions generally have a doubling time that is either less than 20 days (eg,
with some infectious causes) or greater than 450 days However,
using the growth rate to distinguish benign from malignant lesions has pitfalls:
- Difficulty with accurate measurements of small lesions.
- Doubling time is the time it takes for a nodule to increase two-fold in volume (NOT diameter).
Since the formula for the volume of a sphere is [4π]r3 ÷ 3 (where r is the radius),
the doubling of volume corresponds to an approximately 26 percent increase in diameter.
Thus,
doubling the volume of a nodule that has a diameter of 1,
2 or 3 cm,
means the diameter will increase by 3,
5 and 8 mm,
respectively.
Volumetric three-dimensional rendering with modern multislice CT scanners and advanced post-processing software can detect early changes in configuration and subtle growth that would elude conventional two dimensional measurements.
CT
All patients with suspected NSCLC should undergo contrast-enhanced CT that extends through the lungs,
liver,
and adrenal glands.
CT is ideal for TNM staging,
as it can characterize the primary tumor and define its relationship to the chest wall and mediastinal structures,
it can identify mediastinal lymph nodes that are enlarged and suspicious for malignant involvement and it can detect contralateral lung,
chest wall,
or upper abdominal lesions that are suspicious for metastasis.
Helical (spiral) CT performed with a sustained breath hold also eliminates the inconsistency associated with respiratory motion and assures contiguity from section to section.
As a result,
helical CT detects up to 40 percent more nodules than conventional CT,
as the accuracy of CT attenuation is enhanced if the collimation is less than one half the diameter of the nodule,
thereby eliminating partial volume averaging.
Besides,
densitometric and volumetric analysis of nodules and reformatted images with isotropic resolution in sagittal and coronal planes,
free of respiratory motion and steplike artifacts are easily obtained in the work station.
T descriptor: CT assessment of the nodule or mass is generally enough to designate a tumor as T1a,
T1b,
T2a,
T2b,
T3,
or T4.
The presence of atelectasis or obstructive pneumonitis may obscure the true extension of the tumor,
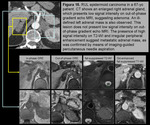
although the use of PET-CT has overcome this limitation (Figure 7)
Most patients who have metastatic disease will have multiple nodules on CT (ie,
it is sensitive); however,
many patients without metastatic disease will also have multiple pulmonary lesions (ie,
it is not specific).
Nonmalignant causes of multiple pulmonary nodules include granulomatous infections,
intrapulmonary lymph nodes,
septic emboli,
sarcoidosis,
Wegener's granulomatosis,
amyloidosis,
and rheumatoid arthritis-associated pulmonary disease.
Clinicians should be aware that the proximal extent of the tumor can only be estimated by CT.
Bronchoscopy is required for confirmation.
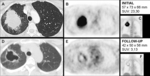
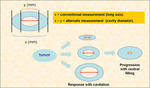
At follow-up studies of response assessment,
not only changes in tumoral volume or size should be taken into account,
but also the presence or absence of cavitation,
as it may indirectly reflect changes in tumor volume and may also be associated with response to treatment.
Cavitation of lesions is more common in NSCLC patients treated with vascular endothelial growth factor receptor inhibitor and platinum-based ChT than in patients treated with conventional ChT.
A lesion may undergo partial response if cavitation is incorporated into assessment,
regardless of whether the overall diameter decreases,
remains stable,
or increases.
Central filling in of a cavity also allows for a declaration of disease progression if cavitation is included in the assessment of the lesion without an increase in overall diameter (Figures 8 and 9).
The most appropriate method to measure volume of tumor tissue remains to be defined.
Advances in the technical aspects of tumor volume and prospective evaluation of such approaches will be required.
However,
pathologic data to support this hypothesis in humans are lacking,
and functional imaging (such as PET) would be one method to assess this in future studies.
N descriptor: Lymph node enlargement on CT (measured in the short axis) presumes lymph node metastasis in the context of a newly diagnosed or suspected NSCLC.Most normal mediastinal lymph nodes are less than 10 mm,
although normal subcarinal lymph nodes can reach a diameter of 13 to 15 mm.
Nodes exceeding these sizes are significantly more likely to contain malignant disease in patients with lung cancer.
Normal lymph nodes are rarely seen in the retrocrural region,
para-aortic region,
or pericardial fat.
Lymph nodes exceeding 8 mm in these regions should be considered suspicious (Figure 10).
However,
the assignment should be considered tentative,
since the use of lymph node enlargement as a surrogate for malignant disease is imperfect.
Metastatic disease can exist in normal-sized lymph nodes (up to 21 %),
while hyperplastic,
benign lymph nodes can exceed 10 mm (up to 40%).
A metaanalysis found that CT for mediastinal staging had a sensitivity (S) of 57%,
a specificity (Sp) of 82%,
and positive and negative predictive values (PPV and NPV) of only 56 and 83%,
respectively.
Thus,
tissue sampling is required to confirm the presence or absence of regional lymph node involvement.
N descriptor: CT of the chest may identify pleural nodules,
a pleural or pericardial effusion,
or separate tumor nodule(s) in a contralateral lobe.
These abnormalities define M1a disease.
Contrast-enhanced CT of the brain detects brain metastases with a S and Sp of 76 and 82 percent,
respectively,
in a population with a prevalence of 14 percent.
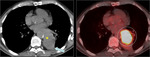
CT of the abdomen may detect focal lesions in the liver or adrenal glands (Figure 11).
These abnormalities define M1b disease.
CT alone is insufficient to exclude bone metastases,
although it may detect lytic or sclerotic bone metastases.
Assignment of M1a or M1b disease on the basis of abnormalities detected by CT should be considered tentative and tissue sampling is required to confirm that lesions are due to metastatic disease.
PET
PET is unique in that it provides metabolic rather than anatomic information.
FDG-PET has become an important clinical tool for the identification of malignant thoracic tumors,
unsuspected metastases in the mediastinum and extrathoracic sites as the technology has become more accessible.
The most commonly used radionuclide in thoracic PET is fluorine-18.
Fluorine-18 is bound to a D-glucose analog,
which yields 2-(fluorine-18) fluoro-2-deoxy-D-glucose (FDG).
A tumor's metabolic activity can be measured using the standardized uptake value (SUV,
formerly called standard uptake ratio).
A high SUV indicates robust FDG uptake due to high metabolic activity,
which suggests malignancy (or active inflammation).
The SUV can be evaluated quantitatively (a numerical value is determined from the PET scan.
An SUV exceeding 2.5 is generally considered highly suggestive of malignancy or active inflammation) or qualitatively (FDG uptake by the tumor is compared to background activity by visual inspection),
with comparable accuracy.
The SUV depends on multiple parameters - the activity of the FDG,
the timing between injection and scanning,
and the size and location of the lesion.
As a result,
each institution must establish its own cutoff value.
Whole body PET should be performed next in most patients with suspected NSCLC.
Exceptions are patients whose CT of the chest revealed possible M1a or M1b disease.
Such patients may proceed directly to tissue sampling to confirm the diagnosis of stage IV NSCLC,
since new findings on PET will not change the disease stage.
T descriptor: PET does not provide sufficient detail to help determine whether a tumor is T1a,
T1b,
T2a,
T2b,
T3,
or T4.
Nevertheless,
PET is more accurate than CT in differentiating malignant lesions (increased uptake) from benign lesions (normal or decreased uptake) as small as 1 cm (Figure 12).
It is estimated that 96 percent of patients with lung cancer will have an abnormal PET (ie,
S),
79 percent of patients without lung cancer will have a normal PET (ie,
Sp),
with diagnostic accuracy of 91 percent.
PET may also help distinguish tumor from peritumoral atelectasis,
thus preventing overestimation of the tumor's size (Figure 7).
N descriptor: PET can detect malignant disease in lymph nodes of normal size,
thus overcoming one of the major limitations of CT (Figure 10).
PET is more sensitive than CT for detecting lymph node metastases,
and also more specific.
This was illustrated by a study that found that PET distinguished N0 or N1 disease from N2 or N3 disease with a S and Sp of 89 and 92,
respectively.
In a meta-analysis comparing PET and CT in nodal staging the S,
Sp,
PPV,
NPV and accuracy of PET were 79,
91,
90,
93,
and 92% compared with 60,
77,
50,
85,
and 75% for CT,
respectively.
PET has a good NPV,
but poor PPV.
This means that false positive (FP) results are relatively common with PET,
which could lead to the assignment of an incorrectly high stage of disease and a missed opportunity for surgical cure.
Thus,
a positive PET should not be considered proof of lymph node metastasis.
Tissue sampling is required to confirm the presence or absence of regional lymph node involvement.
M descriptor: Whole body PET detects metastatic NSCLC nearly anywhere in the body with greater S than CT,
and is particularly usefull in detecting adrenal and bone metastases.
Whole body PET may lead to a change in the M designation in 14-50% of the patients.
There are two exceptions: a) PET is not suitable for the detection of brain metastases because of the high glucose uptake of normal surrounding brain tissue.
Patients who undergo whole body PET require dedicated brain imaging,
generally with contrast-enhanced CT or MRI; b) It is uncertain whether PET is superior to CT for detection of liver metastases.
Some series suggest that PET may be superior to CT in patients whose primary tumor is NSCLC,
but not in patients with other types of malignancy.
Identification of presumed metastatic disease on the basis of PET should be considered tentative.
Tissue sampling is required to confirm that such lesions are due to metastatic disease.
Although bone scan and PET present similar S values in the detection of bone metastases,
the former has been largely displaced by PET for two major reasons.
First,
PET detects bone metastases with similar S and better Sp than bone scans.
Second,
PET has the added advantage of being able to identify metastases in the visceral organs.
Advantages of bone scans include that they are less time-consuming and less likely to have false negative (FN) results from osteoblastic lesions,
as compared to PET.
According to a meta-analysis of eight studies (723 patients),
bone scans identified bone metastases with a S,
Sp,
PPV and NPV of 82,
62,
32,
and 90 percent,
respectively,
in a population with a prevalence of 20 percent.
PET correctly excludes cancer in most cases (high NPV).
However,
it presents poor PPV,
which means that FP results are more common than FN results.
They are generally due to metabolically active infectious or inflammatory lesions and granulomatous diseases such as rheumatoid nodules,
tuberculous granulomas,
and fungal granulomas.
Other sources of FP results are brown adipose tissue in adult humans at the base of the neck,
supraclavicular region,
and superior mediastinum,
and normal or hyperplastic thymic tissue,
both of which can be glucose-avid and lead to high uptake of FDG.
This is why suspicious lesions should be sampled to avoid incorrect upstaging and a missed opportunity for surgical cure.
FN results are less frequent,
and generally occur in one of three settings:
- Tumors with relatively low metabolic activities such as bronchioloalveolar carcinomas well differentiated adenocarcinomas,
and carcinoid tumors.
Rare metastatic lesions from renal cell,
prostatic,
or testicular carcinomas that do not concentrate detectable amounts of FDG have also been described.
- Small lesions (a critical mass of metabolically active malignant cells is required for detection by PET).
- Uncontrolled hyperglycemia: An elevated serum glucose level results in decreased intracellular FDG uptake because FDG and glucose compete for the same cell surface receptor.
To sum up,
the indications of PET in the evaluation of NSCLC are enlisted as follows:
- Solitary pulmonary nodules.
FDG-PET may be useful in distinguishing benign versus malignant pulmonary nodules
- Carcinoma staging,
as discussed previously (particularly in patients with M0 disease and no evidence of lymph node involvement on CT,
in order to plan tissue sampling if lymphadenopathies are detected).
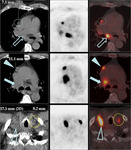
- Follow-up: FDG-PET can be helpful the assessment of recurrent or persistent malignant and differentiate them from changes caused by RT or surgery.
PET has a greater S and Sp for detecting recurrence in a previously irradiated part of the body than CT and MRI.
Increased uptake of FDG beyond eight weeks after the completion of RT represents probable recurrent or persistent tumor,
rather than an effect of radiation (Figures 12,
13,
14).
INTEGRATED PET/CT
Integrated PET/CT scanners fuse images obtained in tandem from PET and CT,
thereby combining the detailed anatomical information generated by CT scans with the metabolic localization of PET scans.
It is an acceptable alternative to performing an initial CT of the chest followed by whole body PET.
T descriptor: PET/CT has the anatomic detail that is necessary to designate the primary tumor as T1a,
T1b,
T2a,
T2b,
T3,
or T4 (Figure 14).
It also appears to more accurately characterize solitary pulmonary nodules in patients who have a history of cancer (eg,
colorectal,
breast,
NSCLC) as benign or malignant than either modality alone,
with S,
Sp,
PPV,
NPV and accuracy ranging between 89-96%,
83-93%,
84-92%,
90-98% and 89-91%,
respectively.
N descriptor: PET/CT has a good NPV and a poor PPV.
This suggests that FP results are common with integrated PET/CT,
which could lead to the assignment of an incorrectly high stage of disease and a missed opportunity for surgical cure.
S,
Sp,
PPV,
NPV and accuracy of regional lymph node staging,
respectively,
were 89,
94,
89,
94,
and 93%,
with CT-PET; 89,
89,
80,
94,
and 89% with PET; and 70,
59,
50,
77,
and 63% with CT.
In view of these findings the 2003 ASCO recommendations are that FDG-PET imaging should be performed in patients with no CT findings of nodal metastatic disease to corroborate the CT findings when there are no distant metastasis (M0) or to redirect nodal sampling by identifying an otherwise undetected site of metastasis (Figure 10).
M descriptor: Integrated PET/CT is clearly superior to CT or PET alone at identifying distant (extrathoracic) metastases.
The reported S,
Sp,
PPV,
NPV of integrated PET/CT for detecting malignant extrathoracic lesions was 98,
92,
89,
and 98 percent,
respectively.
These values were better than those obtained by PET alone (98,
22,
75,
and 89,
respectively) or CT alone (98,
18,
71,
and 89,
respectively) (Figure 11).
Given the frequency of FP results,
integrated PET/CT that is positive for a possible metastatic lesion should not be considered proof of metastasis.
Tissue sampling is required to confirm the presence or absence of metastatic disease.
The impact of integrated PET/CT on staging and therapy sometimes implies that patients undergoing conventional staging plus integrated PET/CT are less likely to be offered surgical therapy.
While it is desirable to correctly increase a patient's clinical-diagnostic stage and potentially spare the patient from unnecessary surgery,
there is evidence from a randomized trial that integrated PET/CT can incorrectly increase a patient's clinical-diagnostic stage.
This could lead to surgical therapy being inappropriately withheld from some patients.
The clinical-diagnostic stage is correctly upstaged approximately three times more frequently than it is incorrectly upstaged.
Patients whose CT of the chest (including the liver and adrenal glands) and either whole body PET or integrated PET/CT do not indicate potential lymph node metastases,
intrathoracic metastases,
or distant metastases do not require additional imaging.
This reflects the low rate of FN results among both whole body PET and integrated PET/CT.
Such patients can be tentatively staged as having N0M0 disease.
Some patients will still undergo tissue sampling to confirm the diagnosis of NSCLC,
while others may be considered for surgical therapy without further imaging or tissue sampling,
as previously mentioned.
MRI
The role of MRI in the diagnosis and staging of NSCLC is more limited than the role of CT.
This is the result of many factors,
including poorer spatial resolution than CT,
low proton density,
long imaging times causing physiologic motion artifact,
and magnetic susceptibility-induced signal loss induced by the air/tissue interfaces in lung.
The use of MRI is reserved for those cases in which the tumor is suspected to be unresectable,
but CT findings are not definitive (as in Pancoast [superior sulcus] tumors) or cases in which the patient has a history of reaction to iodinated contrast agents,as paramagnetic contrast agent used in MRI tends to be better tolerated.
However,
it should be avoided in patients with renal failure due to the risk of inducing nephrogenic systemic fibrosis.
MRI is comparable to CT in evaluating mediastinal and hilar disease.
Due to its multiplanar capability,
the assessment of subcarinal and aorticopulmonary lymph node masses can be easier.
Nevertheless,
it should be always considered as a complementary tool to CT.
However,
MRI may be helpful when brain or adrenal metastasis,
mediastinal invasion,
chest wall invasion,
pleural invasion,
brachial plexus invasion or vascular encasement are suspected,
as well as in the evaluation of transdiaphragmatic growth of tumor or spinal involvement (Figures 12,
13, 14).
MRI is well suited for evaluation of paraspinal masses because of its multiplanar imaging capability.
It can clearly demonstrate the craniocaudal extent of disease,
involvement of the vertebral column,
and/or extension into the spinal canal.
CT evaluation of intraspinal extent of paraspinal masses can suffer from beam-hardening artifact produced by the high attenuation vertebral column: MRI has no similar limitation.
Concerning brain metastases,
imaging the brain with contrast-enhanced MRI is preferable,
although contrast-enhanced CT is a reasonable alternative if MRI is not available.
MRI detects brain lesions and differentiates metastases from other central nervous system lesions with greater S than nonenhanced MRI,
contrast-enhanced CT,
or nonenhanced CT.
Imaging the brain is indicated in all patients with neurological symptoms or signs.
Patients without neurological abnormalities do not require dedicated imaging of the brain unless there are other reasons to suspect brain metastases,
such as extensive local disease,
regional lymph node involvement,
or other distant metastases.
Patients who undergo an initial contrast-enhanced CT should proceed to contrast-enhanced MRI if the contrast-enhanced CT is negative and there remains a high clinical suspicion for brain metastases.
MRI may also be helpful in distinguishing benign,
fat-containing adrenal adenomas from adrenal metastases.
Adrenal adenomas have low signal intensity on out-of-phase gradient echo MRI due to their intracellular fat content.
CT and MR features suggestive of metastases include size greater than 3 cm,
poorly defined margins,
irregular enhancing rim,
and high signal intensity on T2-weighted images (Figure 15).
An adrenal mass in patients with newly diagnosed NSCLC should be evaluated by CT,
followed by MR imaging if needed,
and FDG-PET (two series have shown a S of 100% and Sp of 80 to 90% of PET in identifying adrenal metastases).
Concerning bone metastases,
although cortical destruction is better demonstrated by CT,
bone marrow involvement by tumor is better visualized on MRI.
The ability of MRI to image in arbitrary planes of section is an advantage over CT in assessment of chest wall or local mediastinal invasion.
The newer generation of multislice CT scanners,
which have enabled clinically practical imaging of the entire thorax at submillimeter resolution,
has considerably narrowed this advantage.Both CT and MRI are accurate in confirming gross invasion of mediastinum structures and chest but unreliable in differentiating subtle invasion from anatomic contiguity.
To sum up: Although CT plays a primary role in noncardiac chest imaging,
the multiplanar capabilities and excellent tissue contrast of MRI make it equal or superior to CT in several areas including: Assessment of the apices,
diaphragm,
and spinal column; evaluation of pleural disease and paraspinal masses,
assessment of chest wall invasion,
delineation of blood vessel invasion and metastatic invasion of the bone marrow.