Keywords:
Interventional non-vascular, Oncology, Abdomen, Catheter arteriography, CT, Ultrasound, Ablation procedures, Chemoembolisation, Neoplasia, Cirrhosis
Authors:
F. Turini, I. Bargellini, A. Cicorelli, S. Mazzeo, E. Bozzi, S. Vannucci, R. Lencioni, R. Cioni, C. Bartolozzi; Pisa/IT
DOI:
10.1594/ecr2011/C-0686
Results
SAFETY
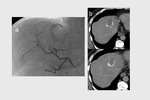
The procedure was technically successful in all cases.
No major complications were observed.
Mild post-embolization syndrome was observed in 16 patients.
No relevant deterioration of liver function was recorded.
The mean hospital stay was 2.1 days ± 0.8 (2-4 days).
TUMOR RESPONSE
At 1-month imaging follow-up,
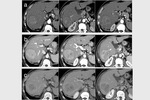
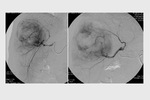
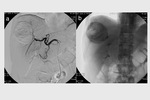
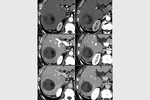
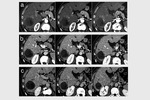
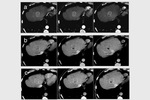
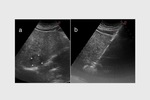
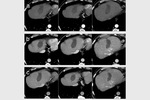
complete tumor response (CR) was obtained in 26 target lesions (76.5%) (Figure 1-9) and partial response (PR) in 8 lesions (23.5%).
CR was significantly (P=.004) higher in smaller lesions (Figure 10).
At 6-months,
CR rate was 66.7% (22/33 target lesions) and PR rate was 30.3% (10/33 lesions) (Figure 5); one case (3%) of tumor progression was observed.
SECONDARY ENDPOINTS
Mean follow-up was 25.4 ± 16.9 months (range 1-63 months).
The median expected time to local tumor recurrence was 14 months (Figure 11),
whereas the median expected time to tumor recurrence in non-target lesions was 18 months (Figure 12).
Radiological disease progression was observed in 8 (23.5%) patients with a mean expected time of 30.7 months (Figure 13).
Finally,
6 patients (17.6%) died on follow-up.
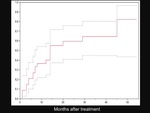
The overall expected 1-,
2- and 3-years survival rates were 89.1%,
84.4% and 77.9%,
respectively (Figure 14).