Gestational trophoblastic disease (GTD) represents a spectrum of conditions that,
although usually benign and easily treatable,
occasionally progress to an aggressive,
potentially fatal process [1].
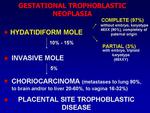
GTD includes pregnancy-associated trophoblastic abnormalities,
which are divided in (fig.
1):
- hydatidiform moles (CHM)
- partial hydatidiform moles (PHM)
- invasive hydatidiform moles(IHM)
- choriocarcinoma
- placental site trophoblastic tumors (PSTT)
Epidemiology
GTD usually occurs in a pregnant woman of any age,
more commonly in teenagers and in women of advanced maternal age (40–50 years).
It affects approximately 1 in 2500 pregnancies in Europe,
1 in 1000 in United States and 1 in 250 in Asia (fig.
2) [2].
Mainly two risk factors increase the likelihood for the development of GTD: woman age < 20 y.o,
or > 35 y.o and a previous GTD (fig.
2) [3,
4,
5].
Pathogenesis
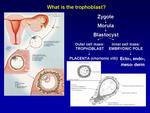
GTD is the result of the abnormal growth of the zygote under paternal nucleus (spermatozoon),
while maternal nucleus (oocyte) is absent or inactivated.
This leads to an anomalous growth of trophoblast,
the functional unit of the placenta,
that covers the early blastocyst and provides the nourishment between the maternal endometrium and the developing embryo [1].
In GTD,
the anomalous trophoblast maintains the ability to infiltrate the deciduas and its endocrine properties (β-hCG production) (fig.
2,
3).
Clinical features
Vaginal bleeding during pregnancy,
sometimes accompained by vesicles expulsion,
is the chief presenting complaint of GTD in early pregnancy.
At physical examination uterus is large for gestational age,
and foetal heart tones are usually absent.
Abdominal pain,
cramps of the lower abdomen can be present in association with elevated serum β subunit of hCG (β-hCG) at laboratory tests (fig.
4) [6]. Because of the excessive production of β-hCG,
ovaries and thyroid hyperstimulation symptoms can be present.
Pathology
The early clinical diagnosis of GTD should be confirmed histologically,
after the evacuation of pregnancy [7].
The histologic appearance of the villous trophoblast changes during different periods of the pregnancy.
Normally,
villous trophoblasts have a polar distribution,
with cytotrophoblastic cells at the center of the group and syncytiotrophoblastic cells at the periphery.
The presence of highly atypical villous trophoblasts with or without hydropic villi is diagnostic of trophoblastic disease (fig.
5,
6) [8].
Diagnostic imaging
After clinical and histological evaluation,
Ultrasound (US) is the examination of choice in GTD.
Computed Tomography (CT) and Magnetic Resonance (MR) imaging play a key-role in determining the extent,
the complications of GNT and during follow-up [9].
Treatment
Because of the invasive nature of trophoblast and the malignant potential of GTD,
surgical evacuation (suction curettage is preferred than hysterectomy) followed by weekly gonadotropin follow-up until β-hCG levels became undetectable,
is preferred above medical methods [10,
11].
Patients are then followed for 6 months: an increase or a plateau in the β-hCG level during the follow-up period defines persistent gestational trophoblastic neoplasia (GTN),
that is an indication for
chemotherapeutic treatment [12].
The choice of a chemotherapeutic regimen is based upon the characteristics of the presenting GTN.
Patients are stratified into low and high risk GTN by combining the International Federation of Obstetrics and Gynecology (FIGO) stage with the World Health
Organization (WHO) prognostic scoring system.
Single-agent therapy is indicated for low risk GTN,
while multi-agentchemotherapies are utilized for high risk GTN (fig.7,
8).