Construction of the phantom
The phantom uses scans of an epoxy-filled human skull to provide a homogeneous background with realistic noise patterns derived from a 320-row CT scanner.
The epoxy-filled human skull was scanned multiple times at various exposure settings ( Fig. 1 a).
A volume of interest (VOI) of 64 x 64 x 30 voxels is selected from the epoxy-filled human skull ( Fig. 1 b) in which ring and cupping artefacts are least present.
This VOI contains a realistic noise pattern that simulates the situation in patients.
The values were corrected for the offset due to the CT values of plexiglas and inhomogeneities of the acquisition.
The resulting pure noise (average HU after offset correction = 0 HU) is given in Fig. 1 c.
A cubic-VOI of 64 x 64 x 40 was created and filled with a geometrical shape,
for example a cylindrical object ( Fig. 2 a).
The extracted VOI demonstrating the noise pattern at the chosen exposure setting ( Fig. 1 c and Fig. 2 b) is superimposed on the cubic-VOI with cylindrical object.
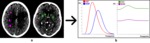
Multiple Regions of Interests (ROIs) were annotated in white and gray matter (WM,
GM) in the patient data ( Fig. 3 a).
To obtain smooth curves mean Time Attenuation Curves (TAC) for WM and GM were calculated by averaging all the ROIs of a tissue ( Fig. 3 b).
The TAC of GM is digitally added to the cylindrical object and the TAC of WM is added to the background ( Fig. 4 b).
The resulting VOI therefore consists of the realistic noise chosen at an exposure setting superimposed on a cylindrical object simulating GM and the background simulating WM which have been extracted from patient data ( Fig. 4 b).
Different CTP acquisition strategies can be simulated by manipulating the time tags of the image data and by using noise patterns derived with various exposure settings.
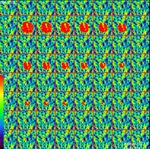
The resulting perfusion maps allow for studying contrast-to-noise ratios (CNR) of the geometrical shapes ( Fig. 5).
Clinical Testing
The hybrid digital phantom was used to simulate a clinical CTP acquisition.
We randomly selected 9 patients who participated in the Dutch Acute Stroke Trial (DUST).
These patients were scanned using a 320-row CT scanner for the indication of stroke at the Leiden University Medical Center (LUMC) between December 2009 and September 2011.
Fifty milliliters of non-ionic contrast agent was injected into the cubital vein at a rate of 5mL/s,
followed by a 40 mL saline flush with a 5 mL/s injection rate.
We tested the phantom by simulating the clinical CTP protocol used for this acquistion.
A total of 24 aquisitions covering 16 cm were obtained with the following scan parameters: 80 kV,
rotation time of 0.75 s and an axial slice thickness of 0.5 mm.
A variable tube current and temporal spacing of scans was used for the different phases (1 x 310 mA (mask),
4 x 160 mA every 2 s (start of arterial inflow),
6 x 300 mA every 2 s (arterial phase),
3 x 160 mA every 2 seconds (venous phase),
5 x 130 mA every 5 seconds (permeability phase),
5 x 130 mA every 30 s (permeability phase)).
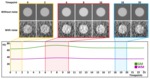
An example is given of this clinical CTP protocol in Fig. 6.
Calculation of perfusion maps
One observer annotated gray and white matter (GM,
WM) in 9 patients (see figure 3) in 5 mm thick reconstructed sections.
The Perfusion Mismatch Analyzer (PMA) [1] software is used to automatically select the AIF and VOF in the patient and to calculate the perfusion maps.
The curve of the AIF and VOF was fitted in PMA using a gamma variant fit.
We used the fitted curve as AIF to calculate the CBF perfusion maps for both the patient and the phantom in PMA.
There was no noise superimposed on the AIF and VOF.
Standard smoothing (denoising) in the PMA software was turned on for both phantom and patient perfusion maps.
White matter TACs derived from the patient were superimposed on the background and grey matter TACs were superimposed on a cylindrical object with a diameter of 20 mm.
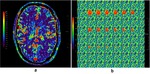
CBF perfusion maps were calculated for both patient data and hybrid digital phantom data ( Fig. 7 ).
Analysis
In order to test whether the digital phantom realistically reflects the noise properties of patient and can predict how these noise propagates to patient measurements,
we compared the contrast to noise ratio (CNR) and noise in the perfusion maps in patients to that in the phantom.
The CNR in phantom was compared to CNR in patients for noise measured in WM.
Spearman’s rank correlation coefficient was used to assess the relationship of patient CNR and phantom CNR.
To avoid measuring the effects of partial volume and smoothing,
CNR measurements were carried out in cylindrical ROIs with a diameter of half the object diameter.
CNR was calculated by the difference between the mean CBF in GM and WM divided by the standard deviation of CBF in WM: (mean(CBFGM) - mean(CBFWM)/SD(CBFWM)).
Noise in CBF maps was measured in the WM simulated background.
The difference in noise in the CBF perfusion maps between the patient and phantom was presented as a percentage and was calculated by the difference of the standard deviation of the CBF of WM in the patient and the standard deviation of the CBF of WM in the phantom divided by the standard deviation of the CBF of WM in the patient multiplied by 100 ((|patient SD CBFWM - phantom SD CBFWM|/ patient SD CBFWM) x 100%).