PATIENTS
Retrospective study along 6 years including 61 patients aged 1 to 62 years (mean age:21 years) diagnosed with AL (ALL-65% of cases and AML-35% of cases) and acute or progressive neurological signs were evaluated in our imaging department by CT and/or by MRI.
The pediatric group was affected mostly by ALL in 82% of cases and the adult group preponderently by AML in 95 % of cases.
TECHNIQUES
Computed tomography (CT). Nonenhanced and/or enhanced CT head (1ml/kg of non ionic iodinated contrast material) protocol were done according to the age of the patient and regarding the unenhanced CT findings.
Magnetic resonance imaging (MRI).
Brain MRI protocols consisted of nonenhanced T1,
T2,
Flair,
T2 EG and diffusion weighted sequences and T1 after Gd-DTPA injection (0,1 ml/kg).
IMAGING FINDINGS
9% of AL patients were within normal limits.
91% of AL patients presented differentes types of cranio-cerebral lesions.
1.
DIRECT CRANIO-CEREBRAL COMPLICATIONS of ACUTE LEUKEMIA
TUMORAL INVOLVEMENT. In AL,
leukemic cells can involve the calvarial bone marrow, dura,
leptomeninges, and more rare the brain parenchyma.
The precise way by which leukemic cells enter the CNS is unknown but may involve hematogenous mechanisms as well as direct spread from adjacent involved bone marrow [4-6,
9-13].
12% of our patients had different types of cranio-cerebral tumoral involvements.
- Meningeal Disease. Infiltration of the meninges by leukemic cells may affect the dura,
the leptomeninges,
or both,
and may be diffuse or focal [13].
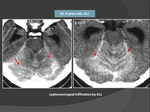
In our study we have found in 5 cases a leptomenigeal tumoral infiltration imaging aspects being correlated with the clinical signs and cytology of the CSF.
Signs and symptoms of leptomeningeal disease,
known as leukemic meningitis were represented by increased intracranial pressure,
including headache,
nausea and vomiting,
irritability,
lethargy,
and papilledema [4,5].
CT and MRI.
Involvement of the meninges may be diffuse or focal with abnormal meningeal enhancement at contrast material–enhanced CT (Fig. 2) and particularly at MRI (Fig. 3).
Cytologic confirmation is necessary for diagnosis,
but repeated CSF analyses may be necessary because of the high frequency of false-negative cytologic findings [4,5].
Imaging is complementary to CSF cytologic analysis.
As in leptomeningeal cancinomatosis,
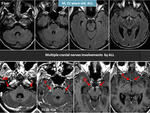
leukemic involvement of the subarachnoid space can be identified on MRI images as either an abnormal appearance of the CSF or diffuse enlargement of the cranial nerves (2 cases in our study) in association with an abnormal enhancement of the meninges.
In our study,
like in all imaging publications focused on this topic,
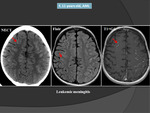
contrast-enhanced MRI was more sensitive than CT in the detection of tumor involvement of the subarachnoid space (see Fig.3).
The best sequence used to evaluate the abnormalities of the subarachnoid spaces is FLAIR (fluid attenuation inversion recovery),
in which,
on precontrast brain MRI,
we can visualize the hyperintensity of the CSF (see Fig.3).
Imaging findings after administration of Gd-DTPA are represented by: abnormal linear or nodular enhancement of the subarachnoid space,
cisternal and pial enhancement,
enhancement and thickening of nerve roots (Fig. 4) suggestive for leukemic meningitis. In rare cases,
a unique meningeal leukemic mass may be imposibile to be differentiate from other tumoral lesions [7,8].
Diffuse dural infiltration by AL is less common than leptomeningeal involvement,
best detected by contrast MRI as abnormal diffuse dural thickening and as abnormal enhancement of the dura [5].
Differential diagnosis must be done with infectious or inflammatory meningitis, as well as irritation from intrathecal chemotherapy or hemorrhage that may be associated with abnormal leptomeningeal enhancement in the absence of leukemic involvement.
Abnormal thickening and enhancement of the dura may also be seen in meningeal fibrosis,
resulting from subdural hematomas related to shunting,
or prior trauma [5,7,8].
MRI alone may be unable to distinguish between benign and malignant causes of an abnormal appearance of the meninges (Fig. 5).
- Intracranial masses. Intracerebral masses may occur in leukemia,
although they are rare.
Chloromais (granulocytic sarcoma or myeloblastoma) is a rare solid tumor composed of primitive precursors of the granulocytic series that include myeloblasts,
promyelocytes,
and myelocytes [10].
This lesion was first described by Burns in 1811.
Granulocytic sarcoma (GS) occur in 2.5–9.1% of patients with acute myelogenous leukemia, having approximately the same rate of occurrence in both sexes.
Children are more often affected than adults: 60% of patients are younger than 15 years old.
Intracranially GS or other leukemic masses may involve the brain parenchyma or be dural-based [14-18].
More frequently GS are contiguous to meninges or ependyma.
When dural-based,
GS may be indistinguishable from meningioma [5].
Intracranial parenchymal masses are rare and may be unique or multiple [4].
In our study we found 4 cases with granulocitic sarcoma, 3 cases with intraparenchymal nodular lesions and 1 case with a mass involving the cavernous sinus and the right trigeminal nerve (Fig. 6) .
CT. At non enhanced CT (NECT),
these lesions are typically isodense or hyperdense and on contrast-enhanced CT (CECT) are brightly [14-18] (Fig. 7).
MRI. At MR imaging,
intracranial leukemic masses are isointense or hyperintense relative to the brain on T1 -weighted images,
isointense or slightly hyperintense on T2- weighted images and presents homogeneous enhancement after Gd-DTPA iv.injection (Fig. 8).
One of our cases with GS presented also bilateral cranial nerves involvements and linear enhancement of the ventricles ependyma (Fig. 9 ).
Cysts or necrosis are not common observed.
They may be associated with small/ moderate edema ( Fig. 10 ) and mass effect.
- Orbital involvement may occur in various forms,
especially in children.
Orbital involvement is common in granulocytic sarcoma [4-6].
The lesions may arise primarily within the orbit,
or they may extend into the orbit from adjacent anatomical structures.
Focal masses may be bilateral intraconally or extraconally (Fig. 11).
Other ophthalmic locations include extraocular muscle infiltration,
optic nerve infiltration,
and intraocular involvement of the anterior chamber,
ciliary body,
choroid,
retina or ocular adnexal involvement [4-6,
19].
2 patients from our study presented bilateral orbital involvments by leukemic cells.
CT. On NECT,
generally, orbital GS are homogeneously isoattenuating to slightly hyperattenuating relative to muscle and less attenuating than the sclera.
Invasion of the orbital fat and extension to the eyelid are commonly observed.
No calcification is detected.
After after intravenous administration of iodinated contrast media,
there is a uniform enhancement of the mass (Fig. 12).
MRI. On T1-weighted MR images,
orbital GS are iso- to hypointense relative to muscle and they replace the high; they are heterogeneously iso- to slightly hyperintense on T2-weighted images. Homogeneous enhancement is seen after intravenous administration of Gd-DTPA [4-8] .
Differential diagnosis. The differential diagnoses of a retrobulbar orbital mass in a child include rhabdomyosarcoma, metastatic,
lymphoma,
neuroblastoma,
African Burkitt,
and idiopathic inflammatory pseudotumor [19].
- Paranasal sinuses and nasopharinx.
GS has also been reported to affect the paranasal sinuses and the nasopharynx [4-6].
- Skull base. Rarely,
the skull base is a primary site for a leukemic mass [4-6].
2.
INDIRECT CRANIO-CEREBRAL COMPLICATIONS of ACUTE LEUKEMIA
CEREBRO-VASCULAR COMLICATIONS. Hematologic cerebrovascular events are among the most severe of CNS complications in patients with AL and contribute to the morbidity and mortality of the disease [4-6].
Every coagulation factor has,
at one time,
been reported to be abnormally elevated on decreased in patients with AL,
and multiple coagulopathies in the same patient are common [4-8,
20,21].
The mechanisms involved in the alteration of coagulation factors include interference with production,
accelerated break down,
and the effect of chemotherapeutic agents,
that can result in a hypercoagulable state or,
alternatively,
in a bleeding diathesis.
AL patients also may develop disseminated intravascular coagulation (CID,) manifested by hypofibrinogenemia,
thrombocytopenia,
and bleeding from multiple sites,
including,
sometimes,
the CNS [5].
Cerebrovascular complications in AL patients include:
- hemorrhage (intraaxial or extraaxial),
- sinovenous thrombosis,
and
- cerebral infarctions.
In our study vascular abnormalities were represented by intracranial haemorrhage (30% of cases), ischemic lesions (7% of cases) and venous thrombosis (4% of cases).
- Intracerebral hemorrhage. Patients with the acute promyelocytic leukemia are at risk for massive intracerebral hemorrhage (ICH),
which represents the cause of death in more than 60% of patients [5].
In AL patients who have disseminated intravascular coagulation (Fig. 13),
a pattern of multiple small hemorrhages with small collars of edema may be seen in the subcortical white matter,
difficult to differentiated only on NECT images from leukemic nodular lesions.
Patients with ICH may present with a sudden onset of headache or neurologic deterioration and seizures (Fig. 14 and Fig. 15) .
- Extraaxial hemorrhage.
Extraaxial hemorrhage is far less common,
may be subdural (Fig. 16 and Fig. 17) or subarachnoid ( Fig. 18 ).
- Dural sinus thrombosis. Patients with AL are at increased risk for dural sinus thrombosis (DST) and cortical venous thrombosis.
Occlusion of DST in AL patients include CNS infiltration,
leukostasis,
and chemotherapeutic agents,
particularly L-aspanaginase.
L-asparaginase is known to produce numerous hemostatic defects including hypofibninogenemia,
prolonged clotting time,
and multiple clotting factor deficiencies.
The reason for thromboses in patients receiving L-asparaginase is poorly understood [21].
Coagulopathies have also been reported with vincnistine and prednisone therapy in leukemic patients.
Signs and symptoms of DST (headache,
papilledema,
lethargy) can mimic CNS leukemic infiltration,
and imaging may be required for differentiation [5].
CT. At CT,
signs of DST are represented by abnormal hyperdensity of a sinus on precontrast scans,
abnormal central hypodensity of a sinus on postcontrast scans (empty delta sign),
or excessive meningeal collaterals.
MRI. MRI findings of the dural sinus thrombosis are: abnormal signal intensity or lack of flow void on precontrast images and empty delta sign (enhancing dura surrounds non enhancing thrombus) on post contrast MR images.
Hemorrhage or venous may complicate sinus occlusion (Fig. 19).
- Cerebral infarction.
Patients with AL,
are at increased risk for cerebral infarction,
although cerebral infarction is far less common than hemorrhagic events [5-9].
Causes are represented by: nonbacterial thrombotic endocarditis,
intravascular coagulation,
SDT,
tumor emboli,
septic emboli,
miscellaneous (vasculitis,
artery compression by a tumor) L-asparaginase therapy [21,22] (Fig. 20).
INFECTIONS. In or study,
head infections weere found in 8% of cases.
Infections are amongst the most common and serious complicationsin patients with AL.
Patients with AL are susceptible to infection for a number of reasons,
including abnormal or decreased granulocytes,
mucosal damage(mucositis related to chemotherapy,
rendering mucosal surfaces vulnerable to microbial invasion),
steroid therapy,
and diminished mucociliary clearance [23-26].
- Sinus infection. Sinusitis are common in AL patients [25,26].
Typical organisms affecting the paranasal sinuses and other mucosal regions (Fig. 21) are the gram-negative bacilli such as Escherichia coli,
Pseudomonas aeruginosa, and Klebsiella pneumoniae and the gram-positive Staphylococcus aureus [23].
Aspergilus sinusitis can produce variables ymptoms and may be accompanied by a foul-smelling discharge [25].
In AL or immunocompromised patients,
this infection may be aggressive,
resulting in bone destruction and extension into adjacent spaces (Fig. 22) or intracranial extension [26].
- Intracranial infection can occur by a number of routes in AL patients.
The most importants routes are represented by direct intracranially spread in aggressive sino-nasal infections and hematogenous intracranial spread [4-6].
Infections caused by Aspergillus or the Zygomycetes fungal groups such as Mucor, Candida and Aspergillus organisms are associated with bone destruction,
vascular invasion,
and rapid intracranial spread,
causing meningitis,
brain abscess,
ischemic or hemorrhagic infarction.
The progression from sinusitis to intracranial spread and death can occur in just a few days in AL patients [4-6,
23].
Hematogenous intracranial spread can occur with various organisms including fungal species such as Aspergillus,
Cryptococcus, and Candida; bacteria such as Listeria monocytogenes;and viral species such as varicella zoster [23].
Endocarditis from systemic fungemia can result in septic emboli and subsequent brain abscess [23,
24].
CT. Abscesses typically appear on CT scans,
as areas of low density,
single or multiple,
with peripheral ring like enhancement.
MRI. At MR imaging,
abscesses are typically hypointense on T1 -weighted images and hyperintense on T2-weighted images,
sometimes with a ring of signal hypointensity on the T2-weighted images that enhance after Gd-DTPA injection associated with edema (Fig. 23). Three imaging patterns have been described in neutropenic patients with cerebral invasive aspergillosis [26]: (a) cortical-subcortical hypoattenuating areas on CT scans or hyperintense areas on T2-weighted images,
(b) multiple ring-enhancing lesions ( Fig. 24),
and (c) dural enhancement adjacent to sinonasal disease.
Candidiasis can also be seen in immunocompromised patients [23,
26].
It appears as numerous ring-enhancing microabscesses less than 3 mm in diameter at the gray-white matter junction,
basal nuclei,
and cerebellum.
Candida affect more frequent vascular structures causing vasculitis,
intraparenchymal hemorrhage,
mycotic aneurysms,
and thrombosis of small vessels with secondary infarction [26].
3.
TREATMENT-RELATED CRANIO-CEREBRAL COMPLICATIONS in ACUTE LEUKEMIA
Side effects of therapeutic procedures (chemotherapy,
radiotherapy and bone marrow transplantation) were observed in 32 % of our AL cases.
Aggressive treatment of acute AL,
may directly result in neurotoxicity.
Many chemotherapeutic agents are known to be associated with neurologic syndromes such as:
- cerebellar degeneration (Cytarabine);
- disseminated necrotizing leucoencephalopaty (Methotrexate); radiation therapy;
- leukoencephalopathy (Cytarabine);
- meningitic syndrome (Methotrexate);
- neuroendocrine dysfunction (radiotherapy);
- vascular occlusion (radiotherapy);
- blindness,
seizures (Cyclosporin A);
- decreasing mental status; developmental delay,
neuropsychiatric decline (many agents) [5].
Clinical features include meningeal irritation,
headache,
nausea and vomiting,
encephalopathy consisting of confusion and disorientation,
and signs of neurologic dysfunction such as slurred speech,
ataxia,
incontinence,
and seizures [4-5].
ANTILEUKEMIC CHEMOTERAPHY
White matter lesions and hemosiderin,
are two of the most common abnormalities on MRI that may be related to cranial irradiation and intrathecal Methotrexate (MTX) [37-39].
MTX is a cell cycle-specific agent that inhibits the enzyme dihydrofolate reductase,
preventing the conversion of folic acid to tetrahydrofolic acid and inhibiting cell replication.
Both high-dose intravenous MTX and intrathecal MTX are associated with demyelination,
white matter necrosis,
loss of oligodendroglia,
axonal swelling,
microcystic encephalomalacia,
and atrophy relatively selective for the deep cerebral white matter [27-29,
37-39] .
The incidence of acute MTX neurotoxicity ranges from 3–10% and varies with the dose, the route of administration of MTX,
the frequency with which MTX is given,
and the use of leucovorin [39].
Transient T2 prolongation in the infra-/ and supratentorial white matter and cerebral cortex has been described [37].
DW imaging showed restricted diffusion limited to the white matter or involving the cortex.
White matter lesions observed on conventional MR imaging are known to be temporary and reversible in some patients,
as in our cases ( Fig. 25 ).
Leucoencephalopaty. MRI spectrum of leukoencephalopathy (LP) has been reported in about 40% of patients during therapy [27-29] and 0%–31% of patients in long-term remission.
LP may be either persistent or transient [44-46].
MRI. White matter (WM) abnormalities seen on the T2-weighted or FLAIR sequences were defined [27] as patchy mildly increased signal intensity in the periventricular WM,
as moderate changes that extend almost to the gray-white junction,
sparing the subcortical U-fibers,
as severe changes,
confluent from the level of the frontal horns to that of the trigones,
with or without involvement of the U-fibers (Fig. 26).
One patient with ALL trated by systemic chemotherapy (Metothrexate) developed left optic neuritis,
that was reversibile in time ( Fig. 27).
Disseminated necrotizing leukoencephalopathy.
The term disseminated necrotizing leukoencephalopathy was introduced by Rubenstein in 1975 to describe the pathologic changes in the brains of five patients who were treated with intrathecal chemotherapy and cranial radiotherapy and who suffered a rapid neurologic deterioration ending in death.
Pathologic changes in those patients included multifocal areas of demyelination,
coagulative necrosis,
glial loss,
and axonal swelling in the centrum semiovale white matter [5].
Reversible posterior leukoencephalopathy syndrome (PRES). The term of PRES refers to reversible acute neurologic complications in patients under treatment for AL and myeloproliferative disorders,
occurring with immunosuppressor medication,
mainly cyclosporine A and L-Asparaginase [30-34].
The clinical syndrome was first described as acute neurologic changes in the setting of sudden or prolonged arterial hypertension that overcomes the autoregulatory capacity of the cerebral vasculature.
Symptoms include seizure,
altered mental status,cortical blindness,
and speech and motor disturbances,
within the first month of therapy.
With discontinuation of cyclosporine A,
the symptoms and radiologic changes are reversible [30-34].
CT and MRI show subcortical white matter edema,
predominantly in the posterior temporal,
parietal,
and occipital areas (Fig. 28),
although more severe cases can also affect the basal ganglia,
cerebellar hemispheres,
and brainstem.
On diffusion-weighted images,
these affected areas usually,
remained normal,
because of the absence of cytotoxic edema [35-36].
Failure to recognize this syndrome may lead to progressive,
irreversible neurologic deterioration with cerebral ischemia and infarction [35-36].
RADIATION THERAPY
White Matter Disease.Neurotoxic reactions to radiation therapy can be divided into acute (1–6 weeks after treatment),
early delayed injury (3 weeks to several months after irradiation),
and late delayed injury (months to years after treatment) [27-29,
40-46].
In acute injury,
there is increased capillary permeability and vasodilatation leading to vasogenic edema.
Early delayed injury includes vasogenic edema and demyelination.
The late delayed effects include white matter necrosis,
demyelination,
astrocytosis,
and vasculopathy [40].
CTand MRI.
Acute lesions are in general mild and reversible,
affecting the periventricular white matter; the delayed toxic effects are more severe and usually irreversible,
resulting from white matter vasculopathy and infarction.
Patients with this latter injury can manifest focal deficits or stupor,
but no direct association between mild or moderate changes at imaging and clinical symptoms has been found [41-46].
Mineralizing microangiopathy. Radiation therapy produces hyalinization and fibrinoid necrosis of small arteries and arterioles with endothelial proliferation and calcium deposition.
Dystrophic calcifications in the basal ganglia (Fig. 29,
Fig. 30) and subcortical white matter were a relatively common finding at cranial CT in children previously treated with radiation therapy and intrathecal methotrexate [47].
Radiation-induced cryptic vascular malformations. Children treated with cranial irradiation due to leukemia can develop white matter hemorrhagic lesions (Fig. 31 ) with heterogeneous signal intensity similar to that of cavernoma on MRI.
T2 GRE and SWI sequences [6] are particularly useful for their identification because of their greater magnetic susceptibility (Fig. 32).
The lesions can be isolated or multiple.
Patients are usually asymptomatic,
although some have headaches,
seizures,
or focal signs.
Parenchymal Brain Volume Loss. Patients wit AL receiving CNS prophylaxis with irradiation or intrathecal chemotherapy may posteriorly develop cerebral volume loss (Fig. 33).
This finding correlates with posterior neurocognitive deficits,
more severe in the youngest group of patients [41-43].
Although enlarged CSF spaces usually result from therapy (mainly,
corticotherapy).
31% of children with ALL have slightly enlarged ventricles before treatment,
probably related to hydrocephalus secondary to primary disease [27-28].
If the enlargement does not reverse after remission but instead persists or even increases,
it is probably related to cranial irradiation [45,
46].
SECOND PRIMARY INTRACRANIAL NEOPLASMS. Patients treated for AL,
especially children are at risk for second primary tumors,
such as glioma (70%),meningioma (20%) or sarcoma (10%) probably as a direct result ofthe antileukemic therapy.
Radiation is a well-known cause ofbrain tumors in survivons patients treated for leukemic disease in whom malignant glial neoplasms developed [4-6,
46].