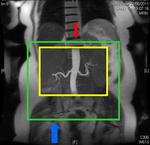
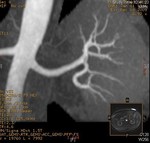
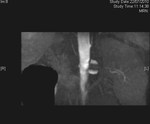
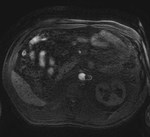
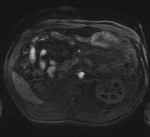
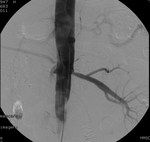
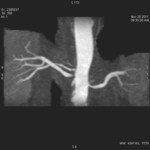
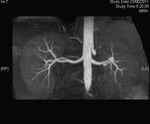
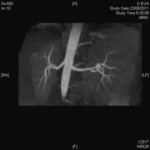
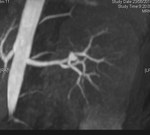
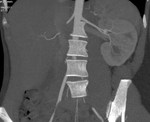
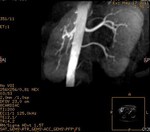
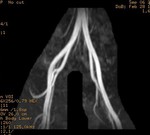
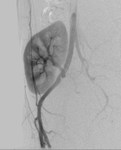
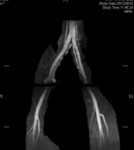
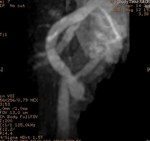
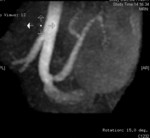
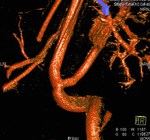
We evaluate the utility and diagnostic capability of 3D-sequence Inhance Inflow-IR (IFIR).
It is a technique that combines sequence ASL (arterial spin labeling) with SSFP (steady state free precession).
Use:
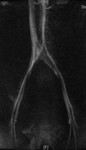
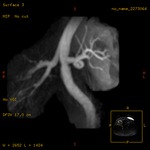
Selective inversion pulse (IR) was applied to suppress the signals from the renal vein blood and stationary tissue (green volume,
figure1).
Spectral fat saturation pulse that enhancing the suppression of the signal from fat and increases the visibility of arterial signal.
A saturation band inferior was used to suppress the signal of the venous flow,
inferior vena cava (blue arrow,
Fig.
1).
A blood suppression inversion time (TI) of 1400 to 1800 ms is used to allow the arterial fresh blood to flow into the renal artery from the superior side of the imaging volume (red arrow,
Fig.
1).
All this along with high blood signal observed in the 3D FIESTA helps to create an angiographic image with high signal suppression of stationary tissue and exquisite delineation of major and the peripheral vasculature in the study area (yellow volume,
Fig.
1).
Use respiratory triggering,
allowing free respiration of the patient,
which promotes patient comfort,
while minimizing respiratory artifacts.
Unlike sequences with respiratory apnea,
respiratory triggering sequence allows to acquire images with high spatial resolution (0.7 mm x 0.7 mm x 1 mm) and high signal/noise,
using parallel acquisition techniques (ASSET Factor acceleration) to reduce the acquisition time.
The usual duration is 3 to 5 minutes.
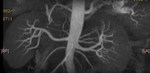
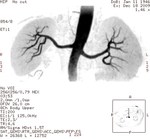
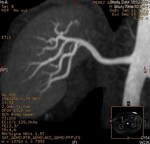
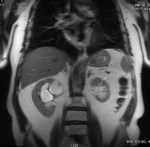
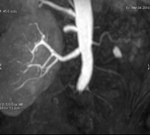
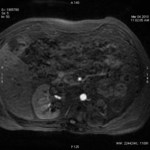
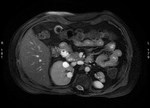
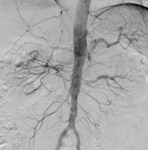
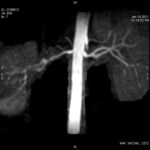
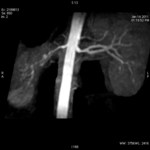
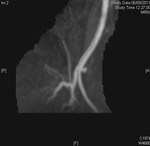
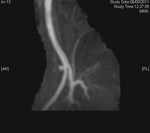
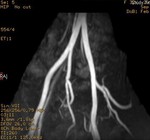
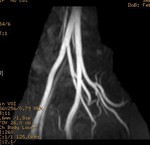
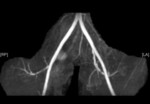
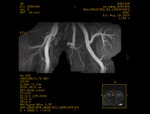
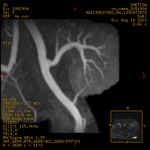
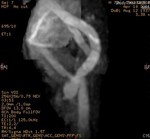
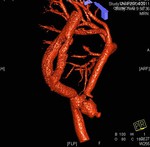
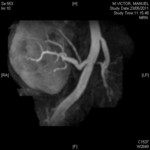
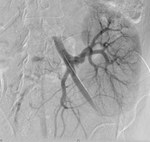
Case 1: 53-year-old man with prostate cancer and hypertension.
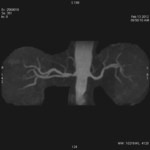
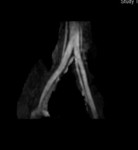
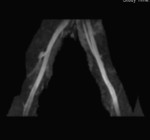
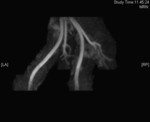
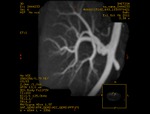
The unenhanced MRA shows normal bilateral renal arteries (Figs.
2,
3,
4,
5 and 6).
Advantages of the sequence:
- Free breathing,
therefore no breath hold problems.
- Sharp delineation of main and peripheral renal arteries due to suppression of respiratory motion (respiratory triggering).
- High contrast between blood and the surrounding tissues.
- High signal-to-noise ratio.
- Spatial resolution of 0.7 mm x 0.7 mm x 1.0 mm for each voxel.
- Independent of the direction of flow.
Limitations of the sequence:
- Maximum FOV of 40 cm.
- Acquisition of data only in the axial plane.
- Potential overestimation of stenosis.
- Low accessory arteries.
(Case 2)
- The saturation band inferior can saturate arterial blood flow in the opposite direction.
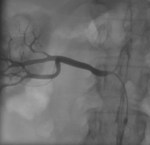
Case 2: 67-year-old male with hypertension,
diabetes mellitus and renal failure of unknown etiology.
In the first sequence (Fig.
7) does not study the hilar inferior right renal artery,
to be below the volume of study.
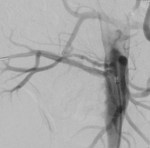
When viewed in the coronal sequence SS-FSE-T2 (Fig.
8) will increase the number of cuts and located more downwardly,
allowing proper study (Fig.
9). Left renal parenchymal atrophy with a single artery normal.
Right kidney with polilobular simple cyst and two hilar arteries without significant stenosis (fig.
10).
Case 3: 45-year-old woman with hypertension and renal ultrasonography asymmetry.
Left renal atrophy with a small renal artery without stenosis.
Right kidney with a normal artery (Figures 11,
12 and 13).
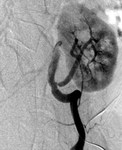
Case 4: 63-year-old male with right nephrectomy secondary to renal cancer. Poorly controlled hypertension and chronic renal insufficiency.
Stent in left renal artery stenosis.
The stent creates a local artifact that prevents assess its permeability (Figs.
14,
15 and 16).
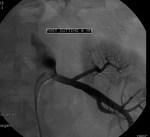
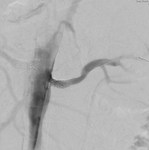
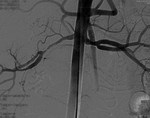
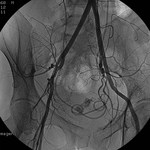
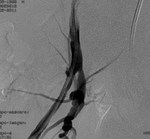
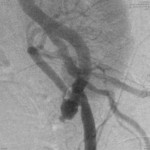
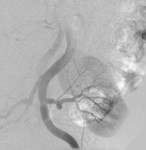
Arteriography demonstrated an intimal hyperplasia with a pressure gradient of 20 mmHg (Fig. 17).
Underwent balloon dilation and cutting balloon with improvement in caliber and resolution of the gradient (fig.
18).
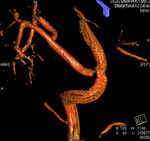
Just as in contrast- enhanced MR angiography,
MIP (maximum intensity projection) and VR (volume rendering) in combination with the axial slices are useful for diagnosis.
According to several authors,
the sensitivity and specificity of this technique in native kidneys is 93-100% and 88-99%,
respectively,
whereas in transplanted kidneys are 100 and 88%,
respectively.
However,
given the very recent development of these new techniques of nonenhanced MR angiography,
there are few published yet and requires further research to confirm these results.
His main clinical applications are:
• In renovascular hypertension study:
The renal artery stenosis is the most common cause of secondary arterial hypertension.
Atherosclerosis is the most common (80-90%),
affected,
usually males over 55,
smoking,
having atherosclerotic lesions in other vascular territories.
The lesion is usually located at the ostium of the renal artery or its proximal portion.
Case 5: 79-year-old male with coronary heart disease,
3-vessel injury and hypertension,
treatment with four drugs.
Admitted the emergency department of the hospital for a hypertensive crisis and left heart failure.
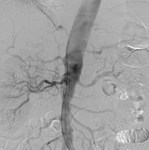
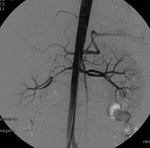
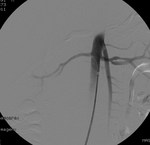
The noncontrast renal resonance angiography detected significant stenosis in the proximal third of the right renal artery (fig.
19).
Arteriography confirms the findings of noncontrast magnetic resonance angiography (fig.
20) and detects a pressure gradient of 20 mmHg.,
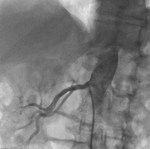
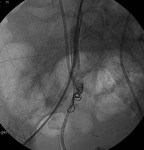
A stent is implanted (fig.
21),
with good results morphological (fig.
22).
Case 6: 70-years-man with type 2 diabetes mellitus,
hypertension and severe atheromatosis.
Presents CKD stage 3.
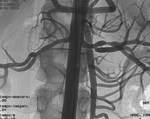
The non-contrast magnetic resonance angiography revealed significant stenosis in both renal arteries: 95% in the right and 80% in the left and a right hilar irregular accessory artery (fig.
23).
An arteriogram shows a 80 % stenosis in both renal arteries (Figs.
24 and 25).
Implanted stent of 6 mm.
by 20 mm.
and 6 mm. by 18 mm.,
with good morphological results (Figs.
26 and 27).
Right hiliar accessory artery show low caliber and irregular contours,
to assess with evolution.
Second cause is the fibromuscular dysplasia (10-15%),
mainly affects young women.
The lesion is usually located in the distal two thirds of the renal artery.
Case 7: 19-year- old woman with hypertension and recurrent episodes of dizziness and nausea.
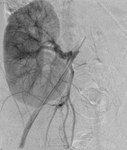
The unenhanced MRA shows irregularities in both renal arteries and long stenosis in the middle third of the right renal artery,
findings consistent with fibromuscular dysplasia.
Critical stenosis in the middle third of the right renal artery (Figs.
28 and 29).
Arteriography confirms the findings of the unenhanced MRA with good correlation (Figs.
30 and 31).
Arteriogram obtained after dilation with a 5 mm diameter angioplasty balloon in both renal arteries shows good results.
Other rarer causes include renal artery aneurysm (Case 8),
Takayasu arteritis and extrinsic compression of the renal artery.
Case 8: 26-year-old woman with primary hyperparathyroidism by right lower parathyroid adenoma.
On ultrasound practiced in the preoperative as incidental finding detected stenosis of the renal vein and a possible aneurysm in the left renal artery
The patient has no hypertension or hematuria.
The unenhanced MRA confirmed nutcracker syndrome: compression of the left renal vein by aorto-mesenteric clamp (fig.
34),
and the existence of an aneurysm in the left renal artery (fig.
35,
36 and 37).
A multidetector computed tomography (MDCT) practiced with contrast showed good correlation with the unenhanced MRA (fig.
38).
Nutcracker syndrome: is the compression of the left renal vein by aorto-mesenteric clamp.
It may present with hematuria,
acute left flank pain and rarely varicocele.
• The study of the celiac axis,
superior mesenteric artery and aorto-iliac area.
It can identify accessory renal arteries causing stenosis of the ureteropelvic junction.
A hilar artery associated with hydronephrosis can be seen in 29%-46% of patients with ureteropelvic junction stenosis.
The vessel is most often located anterior to ureteropelvic obstruction,
however posterolateral vessel localization occurring in 5%-10%.
It is important to identify and locate the vessel prior to endoscopic surgery to prevent damage that can occur in up to 10% of cases (Case 9).
Case 9: 45-year-old woman,
was admitted to the emergency department for severe pain in the left flank.
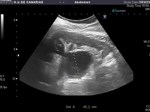
Ultrasound (fig.
39),
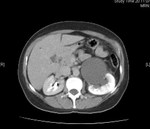
MDCT with contrast (fig.
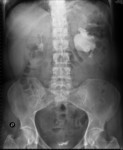
40) and the abdominal radiography after abdominal CT (Figure 41) detected a severe left pelvicalyceal dilatation.
He practices a nephrostomy.
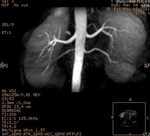
In the unenhanced MRA (Figs.
42 and 43) is evidence of the existence of lower polar accessory arteries in both kidneys,
being left in charge of the ureteropelvic junction syndrome.
A laparoscopic pyeloplasty was performed that confirming the existence of a left polar vessel as responsible of proximal ureter obstruction.
Identify stenosis and pseudoaneurysms aortoiliac sector (Case 10).
Case 10: 43-year-old woman with ovarian cancer operated and treated with chemotherapy and radiotherapy.
Development a frozen pelvis with an intestinal adherence syndrome requiring multiple bowel resections,
enterocutaneous fistula formation and bilateral ureteral stenosis secondary to retroperitoneal fibrosis.
She went several times to the hospital emergency department for hematuria.
In the last entry has a gross hematuria with some clots.
Because of her Cr was 2.3,
it was decided to perform a non-contrast MRA in order to locate the cause of hematuria.
The non-contrast MRA detect a pseudoaneurysm in the beginning of the right external iliac artery that contacted with the ureter (fig.
44,
45 and 46).
Arteriography confirmed the existence of a pseudoaneurysm with fistula to ureter (Figs.
47 and 48).
The proximal segment of the hypogastric artery was embolized with coils to prevent reentry and two coated coaxial stents were implanted (fig.
49) with good final results (fig.
50).
The hematuria is resolved.
• Assessment of the vascular tree of recipient before the kidney transplant.
Kidney transplantation is usually positioned in the pelvis by anastomosis of the artery and vein graft to the receptor external iliac vessels.
It must evaluate the state of the iliac vessels to discard wall calcification,
aneurysms and stenosis preventing or leading to a change in the location of the graft.
Calcifications are not properly evaluated with MRI,
so that an x-ray of the abdomen,
when they exist,
the study is completed with unenhanced CT.
Case 11: 32-year-old woman with type 1 diabetes mellitus since age of 16 with secondary chronic complications: retinopathy and chronic renal failure due to diabetic nephropathy.
He underwent a pelvic unenhanced MRA before the kidney-pancreas transplant to assess the vascular tree,
showing no significant abnormalities.
The source images should be checked to rule out abnormalities that may be missed on MIP reconstructions (figs.
51,
52,
53 and 54).
The transplant was performed.
An arteriography was performed for suspected vascular complications,
no significant changes were detected (Fig.
55).
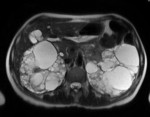
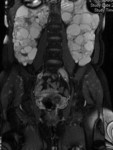
Case 12: 44-year-old woman with adult polycystic kidney disease,
study prior to living donor kidney transplantation.
The aortoiliac sector unenhanced MRA showed no abnormalities (figs.
56,
57,
58 and 59).
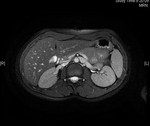
Multiple enlarged cysts were see in both kidneys and in the liver parenchyma (Figs.
60 and 61).
Case 13: 47-year-old woman with type 1 diabetes mellitus diagnosed at 7 years of age with chronic renal failure.
We study the vascular tree prior to renal transplantation.
Unenhanced MRA was performed and phase contrast sequence detected no abnormalities (figs.
62,
63 and 64).
• Assessment of renal graft artery:
Renal transplantation has established itself as the treatment of choice for most patients with endstage renal disease.
The vascular complications most common are: renal artery stenosis,
the renal vein stenosis,
arteriovenous fistula and pseudoaneurysm.
Stenosis of the renal graft artery is the most common vascular complication,
can cause hypertension and renal graft dysfunction.
It can occur in up to 23% of allografts.
Most problems are related to the stenosis surgical anastomosis of the graft renal artery.
Also can be located in the iliac artery proximal to the anastomosis or even in multiple locations within the artery.
This complication is detected mainly in the first and second year after transplantation.
The early detection and proper treatment are crucial to preserve renal graft function.
Thus it is important to have a screening method,
noninvasive,
accurate and safe to assess renal vasculature after transplantation accurately detecting vascular complications,
to decide the most appropriate treatment.
Cases 14,
15,
16,
17 and 18.
Case 14: 59-year-old woman with polycystic kidney disease.
First transplant in 1991 lost due to acute rejection and calcineurin nephrotoxicity.
Second cadaveric kidney transplant prior to elective right nephrectomy in 2008.
Presented hypertension and volume retention.
Unenhanced MRA is requested to rule out artery stenosis renal graft,
no alterations were found (Figs.
65 and 66).
Case 15: 74-year-old male with second kidney transplant for diabetic nephropathy.
Have high blood pressure and Doppler ultrasound suggestive of stenosis of the renal artery graft.
The Unenhanced MRA shows a significant stenosis and a pseudoaneurysm in the left iliac artery (Figs.
67 and 68).
Arteriography confirms the existence of a wide neck pseudoaneurysm in the arterial anastomosis transplantation,
not susceptible of endovascular treatment and stenosis in the origin of the main artery transplantation (Figs.
69 and 70).
The patient underwent surgery but was unable to correct the false aneurysm with graft loss.
Case 16: 58-year-old male with atheromatosis disease,
dyslipidemia and hypertension.
Kidney transplant on 04/06/2009.
Presents increased arterial hypertension,
retention volume and deterioration of glomerular filtration.
Unenhanced MRA no anastomotic stricture exists,
although there is a prominent loop in the middle third of the renal transplant artery (Figs.
71 and 72),
recommending an arteriography to assess its hemodynamic consequences.
Arteriography confirmed in the loop and take pressure,
with no pathological pressure gradient (fig.
73).
Case 17: 18-year-old male with living donor kidney transplant for chronic renal failure secondary to bilateral renal cortical necrosis multifactorial: prematurity,
hypoxia and fetal distress.
Background of two previous artery stenosis renal graft angioplasties in 2006 and 2008.
Now progressive deterioration of renal function is suspected of restenosis.
The Unenhanced MRA shows significant stenosis and irregularity in the onset of renal graft artery (fig.
74).
Arteriography confirms the findings of unenhanced MRA ,
showing a stenosis in the anastomosis and first millimeters,
in a short segment before the main branch arrangement tortuous and irregular edges (Fig.
75) stent is implanted with acceptable final image (Fig.
76).
Following the procedure presented improved renal function,
stability of hemoglobin,
platelet and LDH.
Case 18: 59-year-old male with polycystic kidney transplanted graft body on 23/12/2007.
As history has hypertension and hypertrophic cardiomyopathy.
Presents uncontrolled blood pressure and impairment of glomerular filtration.
An unenhanced MRA is requested to rule transplant renal artery stenosis .
It shows a significant stenosis (fig.
77).
Arteriography evidence an aorto-iliac sector without significant lesions.
90% stenosis at the ostium of the renal artery graft (Figs.
78 and 79).
Angiogram after stenting (fig.
80).