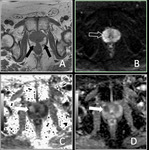
Basic concepts for the biologic basis of DWI and techniques of prostate 3-T DWI.
DWI depends on the microscopic mobility of water.
This mobility,
classically called Brownian motion,
is due to thermal agitation and is dependent on the cellular environment of water (2).
Bulky fluids in the body (such as the urinary bladder or gallbladder) show free and isotropic diffusion.
In biologic tissues,
however,
water diffusivity is restricted by different components,
such as tissue cellularity,
tissue organization,
extracellular space tortuosity,
and the integrity of cell membranes.
Thus,
DW MR imaging is unique in its ability to provide information that reflects tissue cellularity and the integrity of cellular membranes.
Restriction in the diffusion of water molecules is directly proportional to the degree of cellularity of the tissue (3,4,5).
This restricted diffusion is observed primarily in malignancies,
hypercellular metastases,
and fibrosis,
which contain a greater number of cells with intact cell walls than does healthy tissue.
In contrast,
in a microenvironment with fewer cells and a defective cell membrane (eg,
the necrotic center of a large mass),
water molecules are able to move freely (ie,
diffusion is less restricted) (3,5).
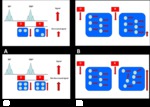
The most common method used for diffusion-weighted imaging is to incorporate two symmetric motion-probing gradient pulses into a single-shot spin-echo (SE) T2-weighted sequence,
one on either side of the 180° refocusing pulse (Stejskal-Tanner sequence),
which made possible direct measurement of water molecules diffusion (6).
Moving water protons acquire a phase shift from the first diffusion-sensitizing gradient,
which,
as a consequence of motion,
is not entirely rephased by the second gradient,
resulting in attenuation of the measured signal intensity.
Hence,
the presence of water diffusion is observed as signal loss on DW MR images.
As stated previously,
the phenomenon of diffusion resembles a three-dimensional distribution; however,
in a single diffusion-encoded image of the diffusion-encoding gradients is measured (7) .
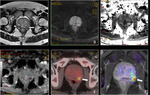
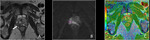
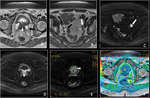
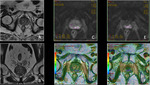
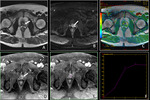
MR diffusion images should be assessed qualitatively or quantitatively using the Apparent Diffusion Coefficient (ADC).
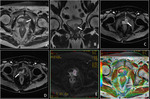
Fig. 1 Fig. 2
Qualitative Analysis of DWI and b value
The sensitivity of the imaging sequence to water diffusion can be altered by changing the b value,
or b factor,
which is dependent in a specific mathematical way on the diffusion-encoding gradient waveforms.
The sensitivity of diffusion-weighted imaging to diffusion can be incrementally increased by increasing the amplitude,
duration,
and temporal spacing of the two motion-probing gradients (10).
These gradient properties determine the b value expressed in seconds per square millimeter (s/mm2),
an index of the degree of diffusion weighting.
In clinical practice,
multiple b values are used to reduce the error in ADC calculation for improved characterization of tissue (Traditionally,
a set of images with a b value of 0 and another set with a b value of up to 1000sec/mm2 are acquired) (8).
Logically,
the maximum b value is limited by the power of the gradients (10).
For each selected b value a sequence of images are acquired (9,10).
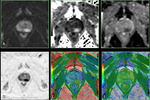
There are several alternatives to present these images usually are represented in grayscale (sometimes inverted) or on scales with arbitrary colors (color map ADC) (6).
Also,
the images can be processed with modern software programs,
obtaining images with multiplanar reconstruction,
maximum intensity projection (MIP and MPR) and even volume rendering.
Diffusion images can also be fused with the images obtained with "conventional" sequences.
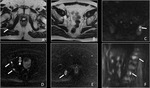
Fig. 3
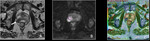
The images acquired with low b values (eg,
50 s/mm2) are characterized by good signal / noise (S / N),
but low diffusion weighting.
As the b value increases,
the sensitivity of the diffusion sequence increases,
although at the expense of a reduction in S / N ratio,
greater distortion of the image and an increase time of echo (3).
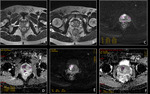
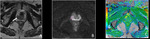
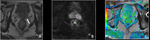
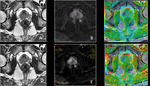
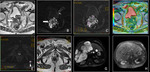
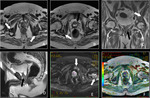
Quantitative Analysis with Diffusion-weighted Imaging: ADC Mapping
The use of DWI enables the calculation of the apparent diffusion coefficient (ADC),
which is a value that measures water diffusion in tissues (3,4).
The final image with different ADC values calculated for each pixel of an image is referred to as an ADC map.
By drawing regions of interest within a lesion,
the ADC value for the lesion can be ascertained. The ADC maps have poor anatomic detail and should be analyzed in conjunction with other MR images,
including different b-value diffusion-weighted images,
higher resolution anatomic images,
and,
if available,
contrast material–enhanced images.
The equation used to calculate the ADC value is as follows:
ADC = −ln (S / S0) / b,
where S0 is the signal intensity of no diffusion gradients and b is the b factor.
The ADC is usually presented as a parametric grayscale image (visual assessment of ADC map) reflecting differences in diffusion of water molecules in an area.
Areas of interest can be also selected (ROI inside the ADC map) to determine the average (or mean ADC) of a tissue or specific area that is expressed in mm2 / s (10) .
The regions with more restricted diffusion and,
therefore,
higher diffusion-weighted signal show lower ADC value,
whereas a large ADC value decreases signal intensity at DWI.
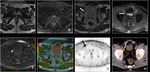
Fig. 4 Fig. 5 Fig. 6
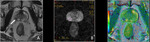
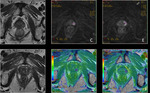
The changes in ADC are inversely correlated with the changes in tissue cellularity: a low ADC indicates areas of restricted diffusion in highly cellular areas,
whereas a high ADC value indicates areas of diffusion in less cellular areas.
Techniques of Prostate 3-T DWI
An echoplanar sequence is used for diffusion-weighted image acquisition and is especially susceptible to magnetic field heterogeneities.
The use of an echoplanar sequence in abdominal imaging is particularly challenging due to the presence of air within the gastrointestinal tract and lung bases.
Other sources of susceptibility artifact include medical devices (eg,
metallic stents,
surgical clips) and grafts.
One way to reduce susceptibility artifact is to shorten the echo time and increase the bandwidth.
To reduce such artifacts 3T equipment apply parallel acquisition image techniques that can reduce the echo time (TE) and the number of echoes and increase bandwidth.
3T MR system increase the signal / noise ratio (SNR) and lets you use high values of b (> 1,000 s/mm2) and smaller slice thickness (# 3mm).
At our institution,
prostate MRI is performed using a 3-T MR system (3 T HDXT,
General Electric equipped with a eight-channel TORSO PA coil). DWI is part of the routine prostatic MRI protocol.
DW images are obtained by applying a single- shot echo-planar imaging sequence with the same slice location as the T2 sequences.
Diffusion-encoding gradients are applied as bipolar pairs at b values of 0 and 1000s/mm2 along all three directions in the axial plane.
Parameters are as follows: TR 6275 ms,
TE 68,1ms,
BW 250 khz,
FOV 38x 26,6 cm,
slice 3/0mm,
matrix 128x128,
10 NEX.
b values 0 and 1,000 s/mm2,.
4m 17s.
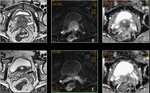
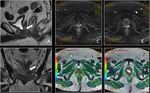
DWI and ADC have become powerful indicators for characterization of prostate tissue,
particularly in differentiation between benign and malignant lesions.
Healthy prostate tissue exhibits signal loss,
whereas areas of restricted molecular motion,
such as in densely packed tumor cells,
show less signal loss and are therefore bright on the raw DWI (11).
The signal intensity observed depends on both water diffusion and the T2 relaxation time.
Consequently,
an area with a very long T2 relaxation time may remain high signal at DWI and be mistaken for restricted diffusion.
This is known as the “T2 shinethrough” Effect. A simple solution to the problem of T2 shine-through can be found using the exponential image (3).
The exponential image is formed by taking the ratio of a DW image divided by an unweighted image (b = 0 s/mm2) from the same image series and slice position.
An echoplanar sequence is used for diffusion-weighted image acquisition and is especially susceptible to magnetic field heterogeneities.
The use of an echoplanar sequence in abdominal imaging is particularly challenging due to the presence of air within the gastrointestinal tract (12).
To reduce these artifacts in pelvis our studies were performed with prior cleansing enema.
CONSIDERATIONS CLINICAL
Prostate cancer is the most frequently diagnosed cancer in males,
accounting for 25% of all cancers in males (192,280 of 766,130 newly diagnosed cancers in males in 2009),
compared with 15% for lung cancer.
It is the second leading cause of cancer-related death in men,
making up 9% of cancer deaths in males (27,360 of 292,540 cancer-related deaths in males in 2009),
a value exceeded only by the death rate from lung cancer in males (30%).
Sixteen percent of males (one in six) will develop prostate cancer during their Lifetime.
The prevalence of prostate cancer increases with age; 34% of men in the 5th decade of life and up to 70% aged 80 years or older have histologic evidence of prostate cancer.
The anticipated demographic change in an aging population is expected to increase the incidence of prostate cancer.
Over the past 25 years,
the 5-year survival rate for all stages of prostate cancer combined has increased from 69% to almost 99%.
The corresponding 10-year survival rate is 93%,
and the 15-year survival rate is 79% (13) .
The notable improvements in survival,
particularly at 5 years,
are commonly attributed to earlier diagnosis and improvements in treatment.
Prostate cancer screening is based on assessment of the level of PSA elevation and results of digital rectal examination (DRE).
Both markers have suboptimal accuracy for the diagnosis of prostate cancer.
At least 15% and up to 44% of biopsy – proved prostate cancers occur in patients with PSA levels in the acceded normal range below 4ng/ml.
In 15.2% of prostate cancer patients with a normal PSA level,
the tumor is not palpable and therefore is clinically silent(14).
In the setting of a high normal total PSA level or in the intermediate range,
data suggest that a free PSA level above 18% of the total PSA is associated with a risk of less 10% for prostate cancer; conversely a free PSA level below 10% is associated with a 30% risk for prostate cancer
Accordingly,
in the setting of a low to intermediate total PSA level,
the clinical decision to perform a prostate biopsy is often made if the free PSA level is found to be below 20%–25% (15).
Prostate cancer detection (tumor localitation and prognostication)
Diffusion weight imaging may be helpful in localizing tumor in patients with previous failed transrectal ultrasound (TRUS) biopsy,
with the potential to improve rebiopsy success rates. DWI at 3T combined with T2WI had improved sensitivity of 81–84% compared with 54–73% for T2WI alone (16).
Sensitivity of ADC values for PC in the peripheral zone is 98%,
compared with 81% in the transitional zone although there were no significant differences in specificity(17).
ADC values obtained from DWI at 3T are significantly lower in prostate cancers with intermediate and high clinical risk scores (D’Amico Clinical Risk Scores) and higher Gleason scores.
A significant negative correlation exist,
the ADC can therefore predict the aggressiveness of CP(18).
The aim was to develop clinical guidelines for multi-parametric MRI of the prostate by a group of prostate MRI experts from the European Society of Urogenital Radiology (ESUR),
based on literature evidence and consensus expert opinion.
Also they developed a scoring system PIRADS similar to that employed successfully by breast radiologists (BIRADS) what includes a combination of high-resolution T2-weighted images (T2WI),
diffusion weighted imaging (DW) MR spectroscopic imaging (MRSI) ,dynamic contrast enhanced MRI (DCE-MRI)(19).
Diffusion weighted imaging (DWI) SCORE:
SCORE 1.
No reduction in ADC compared with normal glandular tissue.
SCORE 2.
No increase in SI on any high b-value image (≥b800).
SCORE 3.
Diffuse,
hyper SI on ≥b800 image with low ADC; no focal features,
however,
linear,
triangular or geographical features are allowed.
SCORE 4.
Intermediate appearances not in categories 1-2 or 4-5.
SCORE 5.Focal area(s) of reduced ADC but isointense SI on high b-value images (≥b800) Focal area/mass of hyper SI on the high b-value images (≥b800) with reduced ADC.
A recent consensus meeting of prostate cancer experts used the UCLA-RAND appropriateness method and recommended that a five-point scale be used for the PI-RADS scoring:
Score 1 = Clinically significant disease is highly unlikely to be present
Score 2 = Clinically significant cancer is unlikely to be present
Score 3 = Clinically significant cancer is equivocal
Score 4 = Clinically significant cancer is likely to be present.
Score 5 = Clinically significant cancer is highly likely to be present.
Postbiopsy Hemorrhage
Hemorrhage in the prostate after a biopsy may decrease ADC values in benign tissues.
This can affect the tissue contrast on DWI between cancer and benign tissue,
which in turn may decrease the diagnostic accuracy of cancer prediction.
Furthermore,
this can increase imaging distortion because of susceptibility artifacts.
A recent study of 3-T DWI showed that although 65% of patients had prostate hemorrhage after biopsy at 3 weeks,
the overall image quality was still sufficient for prostate cancer localization (20).
However,
acute hemorrhage within 1 week after biopsy may hamper prostate cancer localization because of a T2 blackout artifact.
Thus,
we recommend performing prostate DWI at least 4–6 weeks after biopsy.
Prostate cancer staging,
recurrence detection and treatment
Diffusion weight Imaging has been evaluated for their ability to improve staging accuracy.
The most important aspect of local staging is differentiation between organ-confined disease (stage T1 or T2) and early advanced disease in the form of extracapsular extension or seminal vesicle invasion (stage T3).
Advanced MR imaging techniques have been repeatedly shown to be more accurate in differentiation between stage T2 and T3 prostate cancer than other imaging modalities and are preferred for local staging,
despite the lack of clearly specified clinical protocols for integration of MR imaging at this time.
In the work of Kim and colleagues,
they propose that the use of T2-weighted (T2WI) with diffusion-weighted imaging (DWI) as compared with T2WI alone for predicting seminal vesicle invasion (SVI) of prostate cancer.
MR imaging is the most suited for evaluation of the prostate,
as it has unparalleled ability to depict detail of the prostate owing to its exquisite soft-tissue contrast.
,
but MR is of limited value in the evaluation in pelvic lymphadenopathy,
with a diagnostic accuracy similar to scanner.
There is not a unified approach to determine the size of lymph,
but it is accepted a value of 8mm in redonds lymphadenopathy and 10mm in the oval lymphadenopathy.
Diffusion weighted Imaging allows the detection of regional or extraprostatic lymphadenopathy but does not improve diagnostic accuracy.
Use of ultrasmall paramagnetic iron oxide particles with MR imaging have been developed to improve the sensitivity and specificity in the detection of lymphadenopathy (22),
but this contrast is not approved in medical practice.
Beheshti M and colleagues reviewed the value of 18F Choline PET/CT to detect lymph node metastases of size less than 5mm in high-risk patients with a sensitivity,
specificity,
positive predictive value and negative predictive value of 66%,
96%,
82% and 92%,
respectively (23).
DWI of pelvis should be included in the protocol to improve the detection of bone lesions and a whole-body DWI examination can be performed.
The results indicate a higher diagnostic accuracy of MRI compared to bone scintigraphy (24).
TNM
Primary tumor (T)
- TX: Primary tumor cannot be assessed.
- T0: No evidence of primary tumor
- T1: Clinically inapparent tumor neither palpable nor visible by imaging.
-
- T1a:Tumor incidental histologic finding in ≤5% of tissue resected.
- T1b: Tumor incidental histologic finding in >5% of tissue resected.
- T1c: Tumor identified by needle biopsy (e.g.,
because of elevated PSA).
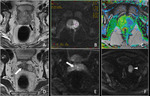
- T2: Tumor confined within prostate.
-
- T2a: Tumor involves ≤one-half of one lobe.
Fig. 7 Fig. 8
- T2b: Tumor involves >one-half of one lobe but not both lobes.
Fig. 9 Fig. 10
- T2c: Tumor involves both lobes Fig. 11
- T3: Tumor extends through the prostate capsule.
-
- T4: Tumor is fixed or invades adjacent structures other than seminal vesicles such as bladder neck,
external sphincter,
rectum,
bladder,
levator muscles,
and/or pelvic wall.
Fig. 16 Fig. 17
Regional lymph nodes (N)
- NX: Regional lymph nodes were not assessed.
- N0: No regional lymph node metastasis.
- N1: Metastasis in regional lymph node(s).
Distant metastasis (M)
- MX: No distant metastasis cannot be assessed
- M0: No distant metastasis.
-
- M1: Distant metastasis.
- M1a: Nonregional lymph node(s).
- M1b: Bone(s).
- M1c: Other site(s) with or without bone disease.
The importance of a correct staging lies on the value prognosis and treatment possibilities .
Prognosis value:
- Low recurrence risk: if PSA levels < 10ng/ml,
Gleason score 2-6 and T1 o T2A.
– Intermediate risk: PSA levels between 10-20ng/ml,
Gleason score 7 and T2B or T2C.
– High risk: PSA levels > 20ng/ml,
Gleason score 8-10 or T3A.
– Locally advanced: T3B or T4.
– Metastatic: N1 with any T or M1 with any T.
Treatment:
- Low recurrence risk: if PSA levels < 10ng/ml,
Gleason score 2-6 and T1 o T2A.
Life expectancy < 10 years: monitoring or radiotherapy (RT).
Life expectancy>10 years: monitoring,
RT within or without hormone therapy (4-6 months) or radical prostatectomy.
– Intermediate risk: PSA levels between 10-20ng/ml,
Gleason score 7 and T2B or T2C: Life expectancy < 10 years: monitoring,
radiotherapy or surgery.
Life expectancy >10 years: RT,
RT within hormone therapy (4-6 months) or radical prostatectomy.
– High risk: PSA levels > 20ng/ml,
Gleason score 8-10 or T3A: Hormone therapy 2-3 years and RT or RT within hormone therapy (4-6 months).
Radical prostatectomy in particularly cases.
– Locally advanced: T3B or T4: Hormone therapy 2-3 year and RT or RT within hormone therapy (4-6 months).
Radical prostatectomy in particularly cases.
– Metastatic: N1 with any T or M1 with any T: N1 with any T: Hormone therapy 2-3 year and RT or RT within hormone therapy (4-6 months).
M1 with any T: Hormone therapy 2-3 year.
The radiotherapy planning consequences
Two of the most extendly used diagnosis methods are: the digital rectal examination (DRE) and the transrectal ultrasound (US).
Both markers have a poor sensitivity and specificity,
what brings up an incorrect staging of prostate cancer and currently establish a wrong radiation management.
MRI at 3T has the ability to increase the sensitivity and specificity of the diagnosis,
and it could change the radiotherapy treatment.
Radiotherapy planning is usually issued by analysing the recurrence risk group,
based on the prostate- specific antigen (PSA) levels,
the Gleason score of the biopsy and the staging by transrectal US or TC.
The MR imaging within the Diffusion- Weighted Imaging identifies morphologically and functionally the prostate tumor,
which leads to a modification on the T-stage,
mostly of the T1c are not visible by US,
restaged to T2A or even T2B and T2C,
turning up into risk group.
Also,
diffusion-weighted imaging allows us to detecting the extracapsular tumor extension and the invasion seminal vesicles,
which modifies the radiotherapy planning and the radiation doses.
Kajihara and collaborators (25) pointed out that the combined use of diffusion weighted imaging and T2 weighted imaging increases sensitivity (from 26-51% to 47-72%) and the predictive value of a positive test (from 47-73% to 58-80%) in the localization of prostate tumor and suggests that this can improve the radiation planning.
At our centre,
the total radiation dose administrated in the low risk patients groups will be 78 Gy and 80 Gy in the rest of patients.
The intermediate-high risk group includes in the treatment field seminale vesicles.
It is used fractional dose 2 Gy each session,
5 sessions per week.
In patients with lymph nodes invasion we use boost technique: 33 sessions: pelvis 1.8 Gy/per session,
59.4 Gy total.
Prostate + seminale vesicles and lymph nodes affected: 72.6 Gy to 2.2 Gy/ per session.
If there is an elevation in the serum levels of PSA after surgery and there is not an apparently recurrence demonstrate in MR imaging,
the radiotherapy treatment administrated to the patient should be 70 Gy in the prostatectomy area; and in recurrence lesions seen in MR imaging,
use 76 Gy in the surgical area and in the recurrence area.
Monitoring the treatment response
Follow-up after radical prostatectomy,
radiation therapy,
hormonal treatment,
high intensity focused ultrasound (HIFU),
or cryoablation of prostate cancer is performed by periodic measurement of PSA levels and digital rectal examination (DRE).
Prostate imaging workup is commonly performed only if the patient has a positive DRE,
elevated PSA levels (biochemical failure),
or symptomatic findings such as bone pain.
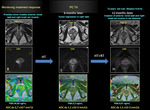
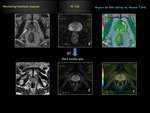
However, DWI may be a useful tool to monitor the therapeutic effects of prostate cancer after hormonal or radiation therapy.
Diffusion MRI measurements are sensitive and can be used to detect and quantify tissue–water diffusion values,
which have been proposed to be related to the ratio of intracellular water to extracellular water; thus,
changes in ADC are inversely correlated with changes in cellularity.
Anticancer therapy leads to tumor lysis,
loss of cell membrane integrity,
increased extracellular space,
and thus an increase in ADC values for the tumors.
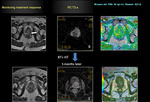
ADC values may be useful as an imaging biomarker for monitoring therapeutic response of prostate cancer to radiotherapy.
After treatment,
there was no significant difference in the ADC values of tumors and benign tissues (26).
In those patients with limited response to treatment,
ADC values do not increase significantly in relation to ADC values prior the treatment.
Fig. 18 Fig. 19 Fig. 20 Fig. 21 Fig. 22 Fig. 23
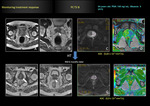
Detecting local recurrence after radiation,
hormonal therapy o radical prostatectomy
MR is able to identify local or systematic recurrence in patients with biochemical recurrence after the first treatment.
Biochemical recurrence after surgery is defined as three consecutive rises of the PSA>0.2ng/ml separated by one month.
The Phoenix Criteria definition of prostate cancer biochemical failure after radiotherapy >2ng/ml levels PSA or more beyond the nadir,
independently if the patients have been treated with hormonal therapy or not (27).
After different modalities of treatment,
prostate gland may present imaging changes as a marked low-signal-intensity in peripheral zone as in the central gland tissue that difficulties the valuation of recurrence.
Nevertheless,
in recurrence cases,
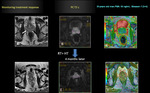
DWI show the decrease in ADC values in prostate cancer after radiotherapy or hormonal treatment.
Kim and collaborators (28) described combination of diffuse-weighted imaging and T2 weighted imaging at 3T for detecting local recurrence after radiation in 36 patients; sensibility and specificity of T2 weighted imaging vs combination T2 weighted imaging and DWI was 25% and 57% vs 62% and 91% respectively (p<0.05).
The mean ADC values in recurrence prostate cancer was 0.98 x 10-3 mm2/s meanwhile mean ADC values in benign prostate tissue was 1,60 x 10 -3 mm2/s.Fig. 24 Fig. 25 Fig. 26
18 months after RT,
some patients (12-33%) undergo a transient rise in serum prostate specific antigen (PSA) levels and subsequent decline in levels without any treatment.
This is known as a "PSA bump”.
(29).
The “PSA bump” is not a failure biochemical predictor and it should not be used as unique recurrence parameter although it is known that patients with oscillation PSA levels may develop higher rates of recurrences (30).
Diffusion-weighted imaging helps to differentiate the “PSA bump” levels after radiotherapy treatment from local recurrence.
Some patients monitored with DWI during the “PSA bump” levels,
show areas with decreased ADC values that normalize in future controls without treatment.
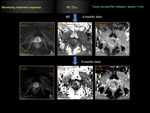
In cases of radical prostatectomy DWI helps to establish the presence of local recurrence and / or distant recurrence (lymph node or hematogenous metastasis).
Local redidiva after surgery is usually located next to the bladder neck or periurethral area.
In recent surgery cases can appreciate postsurgical-fibrous inflammatory changes at this location showing high ADC values.
In exceptional cases,
postsurgical granulation tissue presents low ADC values that can simulate tumor recurrence but these ADC values increase in successive controls without treatment.
Fig. 27 Fig. 28 Fig. 29 Fig. 30
The used of choline as a parameter,
although in minor series,
has promising results in the re-staging prostate cancer.
Recently,
though with a low volume of patients,
Schmid and colleagues (31) used 18 F-choline and equipment PET / CT in patients with suspected biochemical recurrence.
They showed that 18 F-choline CT-PET is a promising imaging modality for detecting local recurrence and/or lymph node and osseous metastases even with low levels of psa(< 5 ng/ml) (31) .
Fig. 31
Limitations of the diffusion-weighted imaging for detecting prostate cancer.
Exists many benign procedures that can decrease ADC values as acute prostatitis,
benign hyperplasia with dense fibromuscular stroma (32,
33) or hemorrhage areas (34) that can simulate prostate carcinoma Fig. 32 .
Other limitation of this sequence includes susceptibility artifacts produced by rectal gas or visceral movement artifactsn (35).
Well-defined tumors with glandular predominance present higher ADC values similar to benign prostate tissue (36).