==============================================
Anatomy of perforating arteries
==============================================
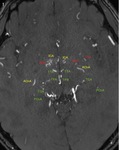
The basal perforating arteries are divided into three groups.
- the striate arterial group arising from ACA and MCA
- the thalamic arterial group
- the AChA and ICA branches
1. supply the basal ganglia.
- MSA: medial striate arteries
- LSA: lateral striate arteries
2. supply the diencephalon.
- TTA: thalamotuberal arteries
- TPA: thalamoperforate arteries
- TGA: thalamogeniculate arteries
- PChA: medial posterior choroidal arteries
- PChA: lateral posterior choroidal arteries
3. supply the area between 1 and 2.
- ICA: perforating arteries arising from internal carotid artery
- AChA: anterior choroidal artery
These basal perforating arteries are difficult to be shown on MRA at 1.5 T. Even if at 3.0 T,
visualization of the perforating arteries is not sufficuent.
However, they may developed to large calibers enough to be delineated on MRA in some pathological states,
such as moyamoya disease,
arteriovenous shunts,
and tumors.
< Perforating arteries in moyamoya disease on MRA >
SEE also Fig.
3-6 (sidebar)
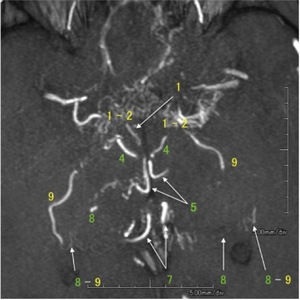
Fig. 1: Partial MIP image of MRA shows developed perforating arteries in moyamoya disease.
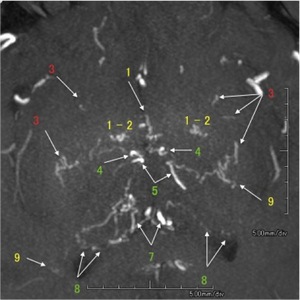
Fig. 2: Partial MIP image of MRA shows developed perforating arteries in moyamoya disease.
<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<
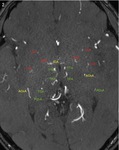
1) ICA: perforating arteries arising from internal carotid artery,
2) MSA: medial striate arteries,
3) LSA: lateral striate arteries,
4) TTA: thalamotuberal arteries,
5) TPA: thalamoperforate arteries,
6) TGA: thalamogeniculate arteries,
7) PChA: medial posterior choroidal arteries,
8) PChA: lateral posterior choroidal arteries,
9) AChA: anterior choroidal artery,
* : anastomosis,
MA : medullary artery
<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<<
< Distribution of perforating arteries >
SEE also Fig.
8 (sidebar)
CASE 1.
a 57-year-old male with moyamoya disease
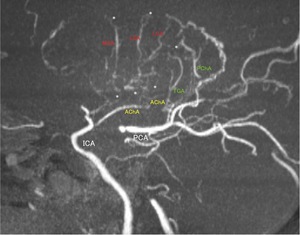
Fig. 7: A Partial MIP image of MRA (lateral projection) shows abnormally developed striate arteries, AChA, and thalamic arteries, which distribute in lentiform nucleus and thalamus. AChA is extremely large and anastomoses with perforators. These arteries depicted on MRA highlights the location of lentiform nucleus and thalamus.
==============================================
Anastomoses between perforating arteries in pathological states
==============================================
In the chronic ischemic state,
especially moyamoya disease,
it is likely to develop anastomoses at the terminal of the end arteries to supply hypoperfusion areas such as lentiform nucleus or cerebral hemisphere.
Therefore,
anastomoses are considered to be borderlines of supply of these perforating arteries.
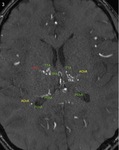
< Anastomoses between perforators and MAs>
SEE also Fig.
13 (sidebar)
CASE 2.
a 63-year-old female with moyamoya disease
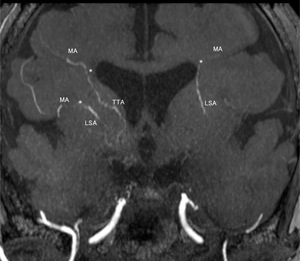
Fig. 11: A partial MIP image of MRA shows anastomoses between perforators (LSAs, TTA, and TPAs) and MAs.
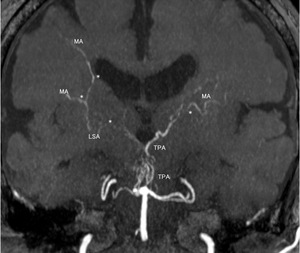
Fig. 12: A partial MIP image of MRA shows anastomoses between perforators (LSAs, TTA, and TPAs) and MAs.
Adults with moyamoya disease occasionally show intraventricular hemorrhage as a first symptom,
although ischemic symptoms but not hemorrhage are more common in children.
This is because that moyamoya vessels are gradually developed and anastomoses are formed in the teminals of end arteries,
where are often near the lateral ventricle.
Therefore,
it is considered that intraventricular hemorrhage is more common in adults than children.
< Hemorrhage at the anastomosis between LSA and MA >
CASE 3.
a 39-year-old female with moyamoya disease
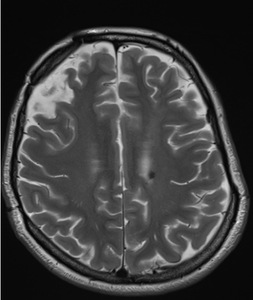
Fig. 9: T2WI shows a small hemorrhage (arrow) at the upper wall of the left lateral ventricle.
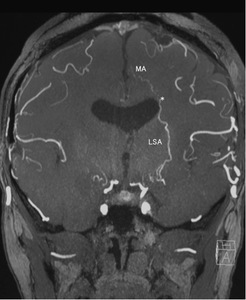
Fig. 10: A partial MIP image of MRA shows MA, which is supposed not to be shown normally. In this case, it is considered that anastomosis has developed at the end of LSA and MA. Therefore, hemorrhage has occurred at the fragile vessels in the anastomosis.
==============================================
Tumor related with perforating arteries
==============================================
Tumors,
especially malignant,
have a increased vascularity.
Feeding arteries may develop around or in the tumor.
In the basal ganglia,
perforating arteries may become markedly apparent on MRA.
< Perforating arteries in Glioblastoma >
CASE 4.
a 67-year-old male with glioblastoma
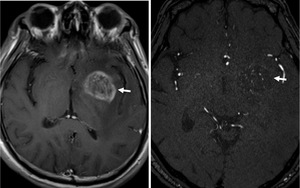
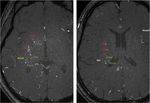
Fig. 14: T1WI with contrast enhancement (left) shows tumor (arrow) in the right lentiform nucleus.
An axial partial MIP image of MRA (right) shows low intensity in the tumor (arrow), which contains many hyperintense dots. They represent abnormally developed MSAs and LSA.
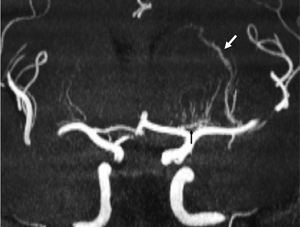
Fig. 15: A coronal MIP image of MRA shows MSAs (black arrow) arising from A1 segment of ACA and distributing mainly in the tumor. An abnormally developed LSA (white arrow) courses along the lateral part of the tumor and direct toward the caudate nucleus.In the distal part of the LSA, the caliber is kept as large as in the tumor.
==============================================
Vascular diseases related with perforating arteries
==============================================
Hemorrhages,
arteriovenous shunts,
infarcts,
and obstructive cerebrovascular diseases are sometimes associated with perforating arteries.
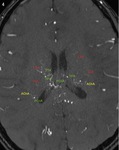
< Hemorrhage caused by the disruption of developed TPAs >
CASE 5.
a 46-year-old female with stenoses of bilateral ICAs after radiation therapy for chordoma of the clivus.
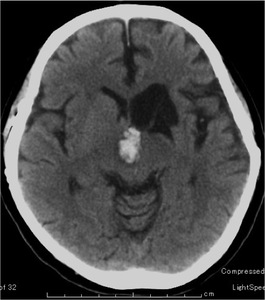
Fig. 16: CT shows hemorrhage in the interpeduncular cistern and midbrain. In the midbrain, there is a low density area adjacent to the hemorrhage, which represents edema.
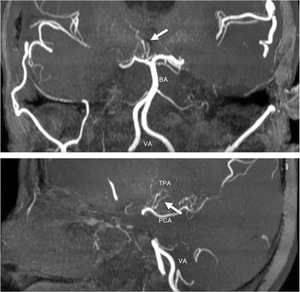
Fig. 17: A partial coronal (upper) and sagittal (lower) MIP images of MRA show several large TPAs arising from the top of BA. TPAs course along the medial side of the right thalamus.
In the cases of arteriovenous malformation (AVM),
peroforating arteries can be a part of feeding arteries to the nidus.
Perforators as feeding arteries may develop to be large enough to maintain hemodynamics of the nidus.
CASE 6.
a 17-year-old female with AVM in insular and operculum
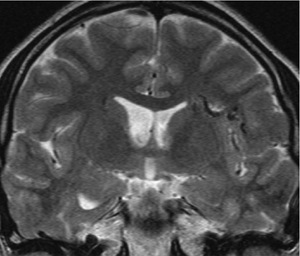
Fig. 18: A coronal T2WI shows arteriovenous malformation in the left insula and operculum.
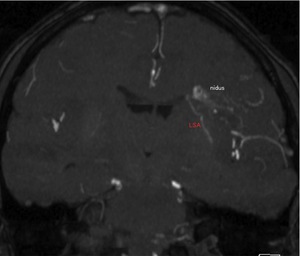
Fig. 19: A coronal partial MIP image of MRA shows the nidus of AVM which is corresponding to flow voids on T2WI. A larger LSA than normal right LSAs is depicted. The end of the LSA is at the upper lateral wall of the left lateral ventricle and anastomoses with the nidus.
Lacunar infarcts are caused by obstruction of the end arteries.
LSAs are common arteries associated with lacuna infarcts.
LSAs are difficult to be delineated on MRA.
However,
MRA sometimes depicts obstruction of the LSA which made a lacunar infarct.
CASE 7. Old lacuna infarct in the corona radiata
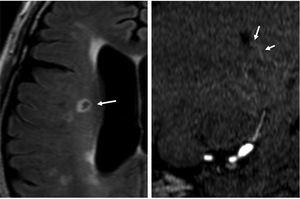
Fig. 20: FLAIR (left) image shows a lacunar infarct (arrow) in the corona radiata.
A coronal partial MIP image of MRA (right) shows the disruption of the LSA (arrow) at the lower part of the infarct.
Hypertensive cerebral hemorrhage is common in the putamen and thalamus.
In patients with hypertension,
LSAs in the putamen are barely demonstrated on MRA, although perforators in the thalamus are not.
Within the cavity of the old putaminal hemorrhage,
MRA occasionally delineate preserved LSA associated with hemorrhage.
CASE 8. Old putaminal hemorrhage
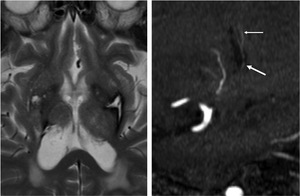
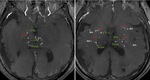
Fig. 21: T2WI (left) shows old putaminal hemorrhage related with hypertension.
A sagittal partial MIP image of MRA (right) shows preserved LSA in the cavity of hemorrhage (arrow). LSA distal to the lesion is also preserved (thin arrow).