Patients and methods:
This is a retrospective study of 10 surgically-proven cases of rectus abdominis muscle endometriosis,
seen over a 5-year period from 2007 to 2012 that were collected from our institution (Nejran Armed Forces Hospital,
Saudi Arabia).
All cases were reviewed as regards history,
clinical examinations,
investigations and management employed.
Sonographic examination was performed in all patients using 3.5- and 5.0-MHz convex-array and 7.5-MHz and 12-15 MHz linear-array transducers (Antares,
Siemens Medical Solutions and E9 General electric medical system,
GE).
Power Doppler sonography was used to assess the vascularity of all lesions.
In addition to sonography,
a CT examination that included intravenous (IV) contrast material was performed in 6 patients on a multi-detector computerized tomography (MDCT) 64 slices scanner (Lightspeed,
General electric medical system,
GE).
Four other patients also underwent MRI on a 1.5-T scanner (GE); those examinations included spin-echo T1-,
fast spin-echo fat-saturated T2-,
STIR sequence and fast spin-echo fat-saturated gadolinium-enhanced T1-weighted sequences.
Pathological material was obtained pre-operatively by FNA in 2 patients and per-operatively by crush smear in 3 cases.
Surgical excision with or without mesh reinforcement was the way of treatment in all patients.
The clinical data were analysed and the imaging studies were reviewed.
Histopathological confirmation of endometriosis was the definite way of diagnosis.
All patients were followed-up for post-operative complications or recurrence of the disease,
for a period varying from 6 months to 2 years.
Results:
Our study comprises 10 patients originating from four different nationalities (Saudi Arabia (4),
Egypt (4),
Pakistan (1),
and Romania (1).
All patients were in the reproductive period with age groups ranging from 16 to 46 (mean 33.9 years).
All except 1 were fertile with parity ranging from 3 to 8 deliveries.
One patient had primary infertility,
bicornuate uterus and ovarian cyst.
All our patients had positive personal history for previous abdominal surgery and 9/10 were subjected to cesarean section (CS).
The lapse interval between CS and appearance of symptoms ranged from 6 months to 6 years.
All our patients presented with moderate to severe lower abdominal pain which was cyclic associated with menses in 3 cases (30%) and non cyclic pain recorded in the remaining 7 cases (70%).
A palpable tender abdominal wall mass was noted only in 3 patients (30%).
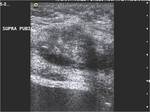
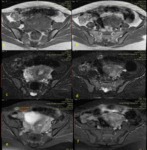
Sonography depicted the lesion in all patients,
except in 1 who had two endometriomas located within the right rectus abdominis and only the lesion close to the midline was depicted,
the lateral one being missed.
The size of the lesions ranged from 15 to 52 mm with a mean of 28 mm.
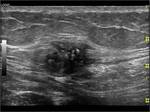
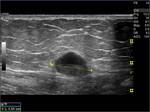
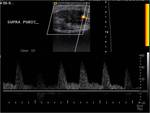
All masses were hypo-echoic and heterogeneous with scattered internal echoes (Figs.1 and 2).
Ten masses were completely solid on sonography,
whereas one lesion also contained some cystic changes and turbid echoic sediment (Figs.3).
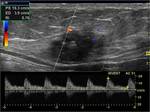
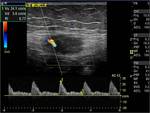
Internal vascularity was noted in all cases with resistance indices varying from 0.70 to 0.77.
The vascularity was peripheral and minimal for 2 lesions having a size less than 1.8 cm and was prominent,
central and peripheral for the other 9 lesions which had sizes superior to 2cm (Figs.
4,
5 and 6).
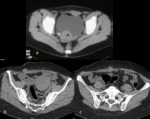
At CT examination,
all lesions appeared as solid,
ill-defined,
isodense nodular thickening of the rectus abdominis muscle,
with slight enhancement after contrast injection (Fig.
7).
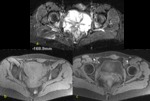
MRI was performed in 4 patients to further establish the nature and extent.
It demonstrated in all cases the lesions as iso-intense to muscle on the T1-weighted sequence,
as inhomogeneous hyper intensity on fat-saturated T2-weighted images and with marked enhancement following contrast administration (Fig.
8 and 9).
All lesions were confined to the rectus muscle,
most of them were located midway between umbilicus and the CS scar,
and in 8 instances the right rectus muscle was affected.
All lesions were solitary except in 1 patient who had two concomitant lesions in the right rectus muscle.
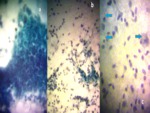
Pre-operative FNAC and intra-operative crush smear preparation was performed in 2 and 3 cases respectively.
They were all suggestive of endometriosis,
presenting at least two out of three criteria for diagnosis: endometrial,
glands,
endometrial stroma or haemosiderin-laden histiocytes (Figure 10 and 11).
Wide local excision with safety margin of 1 cm was done in all cases,
only 2 requiring reinforcement with polypropylene mesh.
One of the latter had two lesions in the right rectus muscle.
The excised specimens ranged from 1.5 to 5.2 cm (mean 2.8) in size and 2-18 gm in mass.
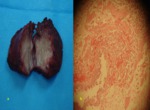
In all patients,
histological examination confirmed the diagnosis of endometriosis,
revealing at least two out of three characteristic features: simple columnar epithelium-lined glands,
vascular endometrial stroma and foci of haemosiderin-laden macrophages (Figure 12).
In all patients investigations revealed no pelvic endometriosis.
All cases went into uneventful post-operative course with no complications or recurrences during the follow-up period (6 months to 2years).
Discussion:
Endometriosis is classically defined as the presence of functional endometrial glands and stroma outside the uterine cavity (1).
This disease affects as many as 5–10% of women of childbearing age,
and in most cases,
is located within the pelvis (2 and 3).
Endometrial implants,
however,
have been reported in many unusual sites outside the pelvis including the abdominal wall especially after surgical operations such as Caesarean sections (2,
6 and 7).
Rectus abdominis endometrioma,
wherein the endometrial focus is solely confined to the body of the rectus abdominis muscle,
is rare and sparsely recorded in literature (4).
Until last year,
only 21 such cases had been reported in medical annals since its first description in 1984 by Amato and Levitt (11).
Although incidental cases of spontaneous endometriosis in a scarless abdominal wall has been reported (8,
12),
it is usually associated with operations in which the uterus is opened [17].
All our patients had positive personal history for previous abdominal surgery and 9/10 were subjected to cesarean section.
Our study comprises 10 patients of different nationalities gathered over a rather short period of time (6 years),
which delineates how undermined this disease possibly is.
Time lapse between the previous operation and the onset of symptoms can vary from 6 months to 20 years (9 and 10).
In our series,
the time interval between operation and clinical presentation ranged from 6 months to 6 years.
Several theories have been proposed for the development of extra-pelvic endometriosis including metaplasia,
retrograde menstruation,
venous or lymphatic metastasis and mechanical transplantation into scars at the time of surgery (13).
In our series,
the positive personal history for previous abdominal surgery noted in all our patients supports the etiologic concept of iatrogenic transport as a plausible mechanism.
Endometriosis of the abdominal wall may be difficult to diagnose.
It is often mistaken both clinically and radiologically for other abnormalities such as incisional and ventral hernias,
haematoma,
abscess and benign or malignant subcutaneous tumors (4).
In general,
the characteristic clinical symptom of endometriosis is cyclic pain associated with menses (9,
10),
which was the case of only three of our patients in contrast with the predominant non cyclic pain recorded in the remaining 7 cases (70%).
The noncyclic nature of pain in endometriosis of the abdominal wall has occasionally been reported by others (3,
5) but has generally been regarded as atypical,
which may explain why it is clinically often misdiagnosed.
Only three of our patients presented with a palpable mass at the site of maximum tenderness in the region of the surgical scar.
Endometriosis has no pathognomonic imaging findings on ultrasound,
CT or MRI,
as its appearance depends on the phase of the menstrual cycle,
the proportion of stromal and glandular elements,
the amount of bleeding,
and the degree of surrounding inflammatory and fibrotic response (14,15).
Ultrasonography is used routinely for assessing patients with abdominal pain.
However,
endometrial lesions in the abdominal wall may be missed during sonographic examination when the appropriate probe is not used,
particularly in obese patients.
Sonographic examination overlooked one of the two concomitant right rectus muscle endometriomas in one of our patients,
possibly because of failure to focus on the near field and to the small size of the lesion.
High-resolution superficial transducers should be used for better assessment of the superficial rectus abdominis muscle lesion.
The rectus muscle endometrioma have been described as solid,
irregular,
elongated and poorly-defined hypo-echoic masse (4,
14,
and 16).
Infrequently a small fluid collection is found in the centre of the mass.
In view of the wide morphological spectrum of endometriosis,
varying from purely cystic lesions to solid deposits or fibrosis (10),
the almost invariably solid aspect of abdominal wall endometriosis in our study is noteworthy.
The imaging appearance might be expected to be more heterogeneous,
with frequent cystic changes owing to intralesional bleeding associated with menstruation.
Our study,
however,
concurs with some other imaging reports (4,
5).
At color Doppler examination,
as noted in our series intralesional vascularisation is abundantly seen if the lesion has a diameter of greater than 3 cm,
whereas hardly any such sign is detected in lesions smaller than 15 mm.
In lesions measuring between 2 and 3 cm,
a few vascular pedicles can be seen,
entering the mass at the periphery and displaying arterial and venous flow pattern.
CT scan is non-specific and demonstrates focal nodular tissue stranding and thickening of the rectus abdominal muscle,
with mild contrast enhancement after intra venous contrast injection as noted in 6 of our patients.
Classical MRI findings,
comprising multiple cystic structures which appear hyper-intense on T1-weighted images and have different signal intensity patterns on T2-weighted scans (18),
were not observed in our patients,
which could be explained by the low number of patients subjected to MRI in or series.
The mass was iso-intense to muscle in T1 images and hyper-intense in STIR sequence with contrast enhancement after injection.
MRI,
though non-specific,
may be useful in distinguishing endometrial tissue from surrounding structures.
MRI is highly sensitive in detecting very small masses and offers excellent differentiation of endometriomas from neighboring tissue (3).
The multitude of signal patterns seen in endometriomas is due,
in part,
to the different stages of hemorrhage found within these implants.
In our cases CT and MR imaging did not reveal the presence of hemorrhage in the mass.
Because of this non-specific presentation,
a broad radiological differential diagnosis remains,
including endometriosis,
abscesses,
hematomas,
hernia,
benign tumors (e.g.
haemangioma,
desmoid tumor) and malignancies such as lymphomas and sarcomas (5,
14 and 19).
The high proportion of glandular tissue might also explain the moderate-to-high contrast enhancement on CT as well as on MRI.
A certain diagnosis can only be accomplished by microscopic examination of the lesion.
Pre-operative assessment can be made by fine-needle aspiration cytology (FNAC) or core-needle biopsy under stereotactic measures (14).
In equivocal cases,
frozen section or crush-smear cytological examination offers a rapid per-operative diagnosis.
In two of our patients,
the provisional diagnosis was preoperatively made by FNA.
It is a rapid and accurate diagnostic procedure in women with abdominal wall endometriosis,
enabling malignancy to be excluded and a definite treatment to be defined.
Koger(9),
Patterson (7) and Calabrese (20) recommend FNAC.
If the FNA results are inconclusive,
as may occur because endometriomas are often fibrous in composition (22),
an additional histological biopsy may be considered.
After complete excision of the mass,
histological assessment of the excised mass imparts the definite diagnosis after examination of formalin-fixed,
paraffin-embedded sections.
The morphological criterion set for a diagnosis is the detection of at least two of the salient features: haemosiderin-carrying macrophages,
endometrial stroma or endometrial glands identifiable by their simple columnar epithelium.
According to Han et al,
malignant transformation of endometriosis is rare,
but cannot be ruled out (21).
The treatment for endometriotic foci in the rectus abdominis muscle can be surgical (4) or expectant,
depending on the severity of symptoms.
In our series surgical treatment was the mainstay of management after confirmation of diagnosis.
We expect an increasing incidence of extra pelvic endometriosis in this era of increasing requests for caesarean delivery.
Hence,
preventive measures should be taken during CS to minimize the chances of endometrial tissue implantation in the abdominal wall.