INTRODUCTION
Endometriosis is defined as the presence of functional endometrial glands and stroma outside the endometrial cavity and myometrium.
This ectopic tissue may appear as microscopic implants to large heme containing cysts (endometriomas).
Due to their hormone-dependence they can cause bleeding,
inflammation,
fibrosis and adhesions,
causing pelvic pain and infertility.
It is difficult to determine precisely the prevalence of endometriosis,
but it is estimated to affect 5-10% of the female population,
with or without symptoms (1).
The prevalence is higherin women withinfertilityor pelvic pain.
Among woman with endometriosis,
the prevalence of the deeply infiltrating type ranges from 5% to 25% (2).
For practical purposes,
endometriosis is classified into three categories in terms of symptoms,
management and evolution: superficialperitoneal endometriosis,ovarian endometriosis,
and deep pelvic endometriosis (infiltrating).
The first form is superficial peritoneal endometriosis,
or non invasive implants,
which are well recognized at laparoscopy.
The second form of pelvic endometriosis is ovarian endometrioma.
An endometrioma is endometriotic cyst that generally occurs within the ovaries and are the result of repeated cyclic haemorrhage within a deep implant.
They may completely replace normal ovarian tissue.
Cyst walls are generally thick and fibrotic and commonly have areas of discoloration and dense fibrous adhesions.
The third form,
deep pelvic endometriosis is defined by the presence of endometriotic lesions penetrating into de retroperitoneal space or infiltrating the wall of adjacent organs at the depth of more than 5 mm from the peritoneal surface (3).
In clinical practice,
deep pelvic endometriosis and deeply infiltrating endometriosis are often used interchangeably,
but rigorously,
the term deep pelvic endometriosis should only be used for the retroperitoneal tissue affecting lesions (3) (referred by other authors like subperitoneal or extraperitoneal endometriosis) (4) and the term infiltrating endometriosis for those involving visceral structures such as intestine,
ureters and the bladder.
Peritoneal endometriotic implants are classically described as “powder burns”,
blue-gray on visual inspection.
This colour is attributed to the encapsulation of menstrual blood by fibrotic tissue that is then haemolysed (3).
Ectopic endometrium may also appear as non-pigmented vesicular lesions,
white patches or red petechiae. Endometriomas typically are composed of thick,
dark,
degenerated blood products.
This appearance has been called “chocolate cyst”.
Endometriomasare bilateral in up to half of the cases and may be large,
although they seldom exceed 15 cm in diameter.
Large lesions and lesions with wall nodularity should be carefully sampled to rule out malignancy.
At microscopic analysis,
both superficial and deep pelvic endometriotic lesions contain glands and stroma and may be accompanied by hemorrhage or adjacent fibrosis.
As with ectopic endometrium,
the ectopic endometrial foci respond to circulating hormones and may show secretory changes during the second half of the menstrual cycle and stromal decidualization during pregnancy.
Haemorrhage within these foci results in an inflammatory response,
with infiltration of histiocytes that become pigment laden with hemosiderin and hemofuscin.
However,
deeply infiltrating endometriotic lesions are typically characterized by generating fibromuscular hyperplasia surrounding the endometriotic foci,
leading to solid nodules formation (solid infiltrating endometriosis).
In visceral infiltrating endometriosis,
the implants adhere to the serosal surface and can invade the muscular layer causing smooth muscle proliferation,
with consequent stenosis and obstruction.
The cause of endometriosis is controversial.
Different theories have been proposed such as metastatic implantation due to retrograde menstruation or the metaplastic differentiation of the serosal surface of the peritoneum or müllerian duct remnant.
Another alternative theory is the induction theory,
which proposes that the ectopic endometrium induces undifferentiated mesenchyme differentiation into endometriotic tissue (1).
Alternatively,
some experts suggest that the rectovaginal endometriosis represents adenomyotic nodules originated from de müllerian remnants due to metaplasia (2).
Treatment for endometriosis is complex,
especially for the deep infiltrating.
Although medical treatment induces temporary quiescence of some active deep pelvis lesions,
it does not act with fibrosis and adhesions,
so that surgical excision is usually the final solution.
Preoperative diagnosis is crucial in these patients to establish a clear division of deep infiltrating lesions,
which can be missed in surgery,
in order to properly plan the surgery and try to ensure complete removal,
what determines a successful treatment (3).
MRI ANATOMY
After de ovaries and the pelvic peritoneum,
in descending order of frequency,
endometriotic lesions are located in the subperitoneal pelvic space,
gastrointestinal or genitourinary tract.
The most common locations of deep pelvic endometriosis are,
in descending ordertorus uterinus and uterosacral ligaments,
pouch of Douglas,
fallopian tubes,
vagina,
bladder and rectosigmoid colon.
The frequency of involvement of the posterior cul-de-sac can reach 54-56% in some series (5).
According to the anatomical distribution,
pelvic endometriosis is classified into anterior,
middle and posterior compartment.
ANTERIOR COMPARTMENT
The anterior pelvic compartment includes the bladder,
distal ureters,
round ligaments and urethra.
These structures are located in the minor pelvis,
anterior to the uterus and vagina and posterior to the symphysis pubis.
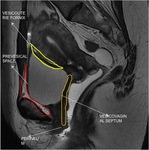
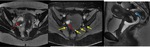
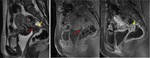
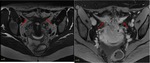
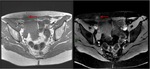
The fat planes between them are called vesicovaginal septum and prevesical space (figure 1) (4).
The dome of the bladder is covered by peritoneum,
creating with the pelvic wall an anterior fold,
the prevesical space,
and with the uterus a posterior fold,
known as anterior or vesicouterine fornix,
the latter being a common location of deeply infiltrating endometriosis.
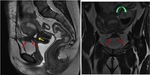
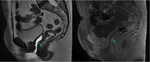
The lesions are usually confined to the serosal surface,
but may infiltrate the detrusor muscle,
generating solid wall nodules projecting into the bladder’s lumen (figure 2).
Bladder endometriosis may clinically present as suprapubic pressure,
urgency and dysuria.
Menstrual hematuria is seen in 20-30% of patients.
In surgery,
the uterus is usually positioned in anteflexion and the anterior fornix is obliterated by extensive peritoneal adhesions between the bladder and the uterus.
The infiltration of the detrusor muscleis generally nodular,
remaining adhered to the anterior wall of the corpus uterinus,
either at the isthmus,
trigone or vesicovaginal septum.
POSTERIOR COMPARTMENT
The rectum and the surrounding connective tissues dominate the posterior compartment.
The rectal fascia forms the morphological demarcation of this compartment,
which is visible con MRI as a thin hypointense structure surrounding the perirectal compartment (4).
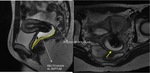
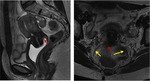
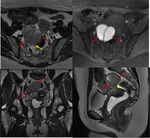
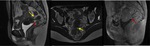
The rectovaginal septum (figure 3) is a thin membrane that contains fat and is located between the posterior wall of the vagina and the anterior wall of the rectum,
stretching from the deepest portion of the pouch of Douglas to the ceiling of the perineum.
In the absence of this fat,
rectal and vaginal walls are indistinguishable.
Vaginal distension with gel allows better assessment of the wall,
with a thickness of 3 mm (4).
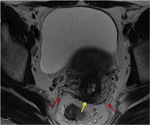
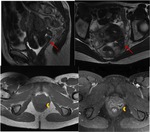
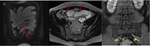
Rectouterine folds contain abundant fibrous tissue and non-striated muscle fibres that are anchored to the sacrum,
forming the uterosacral ligaments (figure 4).
On MRI,
these structures are seen as thin hypointense lines originating from the lateral margin of the cervix and posterior vaginal vault to target,
back and cranially,
the sacrum.
The torus uterinus is a small transverse thickening linking the original insertion of the uterosacral ligaments,
behind the posterior cervix,
even though it is a structure that is rarely seen in normal on MRI,
unless thickened (4).
The retrocervical space is an extraperitoneal virtual space behind the cervix,
located in the same plane as the pouch of Douglas and cranial to the rectovaginal septum.
The vaginal fornices are the deepest portions of the vagina resulting from the recesses created by the extension of the cervix into the vaginal area.
The major recess is the posterior vaginal fornix and is located posterior to the cervix.
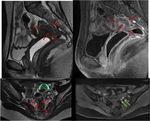
The cul-de-sac or pouch of Douglas (figure 3) is a deep peritoneal sac located between the two rectouterine folds.
It is the lowest portion of the peritoneal cavity,
covering part of the vagina and rectum,
being the upper limit of the rectovaginal septum its baseline.
In 93% of woman it extends to the middle third of the vagina (4),
although,
in normal,
it is not visible on MRI,
unless there is peritoneal fluid.
The endometriotic lesions of the posterior compartment include peritoneal wounds located in the most dependent portion of the pouch of Douglas,
which generate adhesions between the peritoneal surface of the anterior and posterior wall of the rectum (figure 5),
and may infiltrate the muscular layers of these structures.
This could justify that the endometriotic lesions located on the rectovaginal septum are originated from massive disease in the deepest part of the pouch of Douglas,
with subsequent exclusion of the remaining pelvis by adhesions (2).
Rectovaginal septum lesions were classified according to their location in rectovaginal septum’s lesions (type I),
lesions of the posterior vaginal fornix (type II) and hourglass injuries (type III) (3).
- - Rectovaginal septum lesions (figures 6) are observed in 10% of cases and are usually small.
They are located on the rectovaginal septum,
between the posterior wall of the vaginal mucosa and the muscle layers of the anterior rectal wall.
These lesions are not related to the cervix and are placed in extraperitoneal location,
under the peritoneal fold of the pouch of Douglas (3).
- - The lesions of the posterior vaginal fornix (figures 7 and 8) are observed in 65% of cases.
These lesions are usually small and do rarely spread to the rectovaginal septum or rectal wall,
although they can cause fibrosis of the fat and mesorectal fascia
- - The hourglass lesions are observed in 25% of cases (figure 9) and occur when the posterior cervix lesionsextend cranially to the anterior rectal wall.
They tend to be larger (more than 3 cm) with systematically rectal wall invasion.
Bowel endometriotic involvement is usually seen in the rectosigmoid colon (figures 9 and 15) but can affect,
in descending order,
the appendix,
cecum and distal ileum (figures 15 and 16).
This has been estimated to occur in 12-37% of patients with endometriosis.
The implants are usually serous but can also erode and infiltrate subserosal layers,
with thickening and fibrosis of the muscularis propria (fibromuscular hyperplasia).
On MRI they behave like retractable nodular formations,
hypointense on T2-weighted sequences,
attached to the wall.
Serous implants or adhesion syndromes can only appear as parietal distortion or angulations,
while infiltrating implants may cause focal or circumferential wall thickening,
mucosal erosions,
leading to stenosis,
real sub-occlusive or obstructive processes,
and even the formation of rare intestinal endometriomas.
In a systematic review,
the microscopic extension of mural involvement in patients undergoing bowel resection for colorectal endometriosis,
95% showed serosal invasion,
95% invasion of the muscularis propria,
38% penetrated to the submucosa and only 6% had mucosal involvement (2).
MIDDLE COMPARTMENT
The middle compartment contains the female genital organs,
including the ovaries,
uterine tubes,
uterus,
and vagina.
The broad ligaments are peritoneal folds between the uterus and the lateral walls of the pelvis and are also a part of the rectouterine and vesicouterine folds.
The ovaries lie in the ovarian fossa and are suspended from a double fold of peritoneum known as the mesovarium.
Behind the ovarian fossa are extraperitoneal structures,
including the ureters and the internal iliac vessels.
DIAGNOSIS
Laparoscopy is considered the reference diagnostic method for endometriosis (1).
The presence of nodules or brown or black plaques on theperitoneal surface is a pathognomonic finding.
For staging of endometriosis (1),
the classification of the American Fertility Association (1975) is the most widely used.
It consists of three components: evaluation of the endometrial implants (location,
size and depth of penetration),
degree of cul-de-sac obliteration and the evaluation of adhesions (appearance and amount of surface involvement).
According to their score,
patients are categorized in 4 types: minimal,
mild,
moderate or severe disease.
This staying system offers some standardization for a better assessment with prognostic and therapeutic purposes.
Nevertheless,
laparoscopy shows limitations in atypical cases,
mainly in the assessment of extraperitoneal deeply infiltrating endometriosis,
often hidden by adjacent adhesions.
During surgery,
it requires palpation and the opening of the subperitoneal space to confirm and evaluate the extension of the lesions,
and thus,
a good radiologic evaluation facilitates the choice of surgical strategy.
Ultrasonography (US) is the first and most common imaging modality used to evaluate female patients with pelvic pain and infertility.
Transvaginal US is the most widely performed.
It is very helpful for the assessment of ovarian endometriomas and bladder endometriosis,
but is of limited value in assessing peritoneal endometriosis,
adhesions and deeply infiltrating endometriosis.
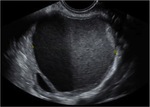
The classic sonographic appearance of an endometrioma is described as a well defined and homogeneous lesion with moderately echogenic content located within the ovary (figure 10)(1),
although ultrasonographic (US) features are variable and can mimic those of other benign and malignant ovarian lesions,
especially teratoma.
Rectal endoscopic US with high frequency transducers has been recommended for rectovaginal,
uterosacral ligaments and rectosigmoid endometriosis detections.
Its main advantage is that it provides a reliable mean to evaluate the degree of intestinal wall infiltration.
Preliminary results show that rectal endoscopic US is better than MRI for detecting intestinal wall infiltration (6).
MRI
Because of its multiplanar capability,
high sensitivity in detecting blood degradation products and ability to identify occult disease due to adhesions,
MRI is the non-invasive imaging modality of choice for the detection and staging of endometriosis.
It also provides a tremendous advantage over other diagnostic methods due to the possibility of a full panoramic assessment of both anterior and posterior compartments of the pelvis at the same time.
In a study by Togashi et al.
(1; 7),
MRI reaches a sensitivity,
specificity and accuracy,
for the diagnosis of endometriomas,
of 90%,
98% and 96% respectively.
However,
its value in demonstrating bladder endometriosis,
superficial lesions and ovarian foci is controversial and some authors have highlighted the limitations of MRI in detecting small lesions in these locations,
although recent studies have shown good sensitivity in identifying small peritoneal implants (6).
Considering all locations as a whole,
MRI has a sensitivity of over 90% (6;8).
Nevertheless,
the radiological evaluation may not be easy,
mainly when obliteration of the rectovaginal recess takes place or in cases of vaginal o rectovaginal septum involvement,
where the sensitivity of MRI may fall between 70-80%,
according to some authors (6).
Classically,
endometriosis was diagnosed with MRI by detecting ovarian endometriotic cysts (endometriomas) and heme-containing implants.
These lesions appear hyperintense on T1-weighted fat-suppressed sequences,
allowing their differentiation from fat containing lesions (teratomas).
A pathognomonic feature of endometriomas is “shading”: a hyperintense cyst on a T1-weighted image becomes hypointense on a T2-weighted image.
This shading reflects the chronic nature of an endometrioma as a result of repeated episodes of chronic bleeding,
accumulating extremely high concentrations of iron,
proteins and intracellular methemoglobin.
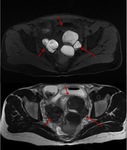
30% associates tubal abnormalities as hematosalpinx(1)(figures 11 and 12).
In deep pelvic endometriosis there is a wide variety of lesions finding rectovaginal septum lesions,
small infiltrating implants,
deep pelvic solidligamentous lesions and adhesions (typically located in the pouch of Douglas,
affecting the uterosacral ligaments or torus uterinus) or solid visceral infiltrating endometriosis (affecting the bladder or rectal wall).
Imaging findings represent directly the predominance of fibrotic or glandular component.
Thus we can find fibrotic,
glandular,
fibroglandular or fibromuscular lesions,
with or without the presence of hemorrhagic content.
In deeply infiltrating implants,
MRI may occasionally show small punctate hemorrhagic foci,
not being essential for the diagnosis of endometriosis.
The predominant and ubiquitous fibrotic component and the adhesions appear with low signal intensity on T2-weighted images (difficult to differentiate from vaginal and rectal walls) (figure 12).
In the absence of inflammatory disease,
these fibrotic lesions do not enhance after contrast administration.
Some endometriotic masses located on the round ligaments (figure 13),
uterosacral ligaments,
posterior vaginal fornix or pouch of Douglas,
may be composed of a large proportion of glandular materials with little fibrotic reaction,
resulting on high hypersignal on T2-weighted images (4) and can show punctate foci of high signal intensity on T1-weighted images representing petechial haemorrhage.
These solid glandular materials usually enhance intensely and early after intravenous contrast administration.
The involvement of the uterine ligaments,
specially the uterosacral ones,
with endometriotic nodules,
generates their thickening and,
in advanced stages,
fibrosis and adhesions with obliteration of the pouch of Douglas.
On MRI,
adhesions are usually hypointense,
shading the interfaces between the pelvic organs.
Indirect signs suggestive of adhesions are ovarian displacement,
uterine retroflexion,
bowel angulations,
posterior vaginal fornix elevation or the presence of loculated fluid (figures 14)(1).
Solid visceral lesions correspond to fibroglandular or fibromyomatous implants and appear with low or intermediate-signal-intensity and punctate hyperintense foci on T1-weighted images,
showing a uniform and low-signal-intensity on T2-weighted images.
After the administration of intravenous contrast,
enhancement has been described corresponding to abundant fibrotic tissue located on these lesions on histological analysis,
although in our experience these deep fibrotic implants as well as the visceral infiltrating implants (by proliferation of smooth muscle tissue) appear in dynamic studies as hypointense lesions relative to the intestinal wall in the early stages with progressive and intense enhancement in late phases (figures 15).
MRI-PROTOCOL
1.
- In our institution we perform pelvic high-resolution MRI on 3Timaging system with an 8-channel cardiac coil,
without prior preparation.
The anatomical high-resolution images have been obtained with FRSET 2 sequences in all 3 standard projections (axial,
sagittal and coronal).
2.
- Performing T1-weighted sequences (SET 1) is mandatory to improve the conspicuity of heme containing lesions,
which is a cardinal feature of endometriotic implants (figures 5,
6,
11,
12 ).
3.
- Per protocol,
we perform 3D LAVA dynamic contrast-enhanced MRI in axial and sagittal projections.
The usefulness of intravenous contrast to define the extent of active inflammatory processes associated with endometriosis has been described (1)(figures 9,
13,
15).
Enhancement of the peritoneal surface can show the underlying implants location,
localized or diffuse,
in case of inflammatory reaction,
induced by deeply infiltrating implants situated in peritoneal reflections on uterosacral ligaments and the pouch of Douglas.
Post contrast sequences have also been recommended to identify enhancing solid nodules in endometriotic cysts when malignancy is suspected (1)
4.
- MRI is performed regardless of the timing in the patient’s menstrual cycle since there is no evidence that performing MRI scan during menstruation has higher diagnostic accuracy.
However,
the date of the last menstrual period is documented.
5.
- For better assessment of rectovaginal endometriosis we obtain images after vaginal distension,
and depending on the suspect,
also rectal distension,
using sterile ultrasonographic gel or rectal enemas with mild saline solution (figure 9) (4).
Opacification and distension of the vagina and rectum with gel improves significantly the sensitivity of MRI for detecting deep pelvic endometriosis,
with better delimitation of pelvic structures.
(Sterile ultrasonographic gel has a similar signal intensity to the water,
hyperintense on T2-weighted sequences,
what contrasts with the relative low signal intensity of the vaginal and rectal walls).
Moreover,
due to its viscous nature,
the loss during the examination is prevented.
It is,
therefore,
particularly effective in detecting vaginal and rectovaginal septum lesions.
6.
- Although bowel cleansing is not routinely performed,
the patients must fast for at least 6 hours before the examination.
Sometimes an antispasmodic drug is administered in order to reduce artefacts caused by intestinal peristalsis and uterine contractions.
20 mg butylescopolamine (Buscopan) is injected intravenously immediately before the start of the examination.
7.
- We try to ensure the bladder is half filled,
inasmuch as an empty bladder or a very filled one obliterates adjacent recesses,
impeding an adequate assessment of small lesions.
8.
- In case of suspected intestinal involvement,
MRI enterography is performed (9) which can be done in a single study time or in two separated sessions.
It is carried out on a 1.5T MRI with a surface antenna that allows studying the entire abdomen and pelvis in the same scan.
We include FIESTA sequences in axial and coronal projections,
fluoroscopic MRI or SSFSE multiphase (cinema mode) and SSFSE FAT-SAT in axial and coronal projections and dynamic LAVA study in axial and coronal projections after the administration of gadolinium.
Furthermore,
in the same scan we can study the urinary tract,
thanks to the coronal FIESTA sequences,
the dynamic sequences (they allow dynamic assessment of both ureters) and the post contrast uro-MRI (figure 16)
9.
- Endometriosis can also occur in nongynecologic sites.
Endometriosis has been described in chest (thoracic endometriosis síndrome),
skull and cutaneous scars.
All of them are associated with pelvic endometriosis except cutaneous endometriosis.
In these cases it is necessary to further examination based on clinical suspicion.
(Figure 17).