Keywords:
Metastases, Observer performance, Contrast agent-intravenous, CT, Oncology, Liver, Abdomen
Authors:
M. Gajic Dobrosavljevic, J. STEVANOVIC, P. P. Milenkovic; Belgrade/RS
DOI:
10.1594/ecr2013/C-0906
Methods and Materials
This study was carried out at Institute for Oncology and Radiology of Serbia.
The sample encompassed 25 patients who were initially diagnosed with colorectal adenocarcinoma and consequent liver metastatic disease.
All patients underwent eight to twelve cycles of chimeric anti-EGFR monoclonal antibody agent therapy combined with other chemotherapeutics.
The study data were comprised of CT examinations performed by Siemens SOMATOM Sensation Open involving contrast-enhanced 3mm-reconstructed images from 5mm thickness.
The study involved portal venous phase images of baseline CT examinations and follow-ups after averagely 25.6 weeks.
The analyses process was conducted on Carestream Health PACS Viewer 11.3.2 using liver windowing.
The measuring was performed in accordance with RECIST 1.1 criteria carried out by two experienced radiologists and one resident of the first year.
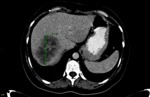
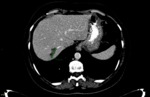
For the purpose of the current study only the largest solitary observed liver metastatic lesion was followed.
In order to estimate inter-observer reliability,
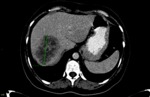
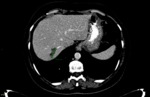
baseline and follow-up readings were initially performed by the first radiologist (reffer to Fig. 1 and Fig. 2,
respectively),
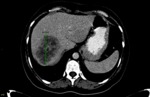
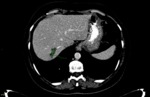
afterward the recordings were repeated by the second radiologist (reffer to Fig. 3 and Fig. 4,
respectively) and the first-year resident (reffer to Fig. 5 and Fig. 6,
respectively).
The intra-observer reliability was tested by conducting independent second series of observations with a month lapse.
The reassessments were carried out by all three observers.
The involved observers assessed the scans independently without knowledge of patient’s age and sex.
Statistical analyses were performed using SPSS for Windows,
version 15.
The intraclass correlation coefficient (ICC) was used for estimation of intra- and inter-observer reliability.