INTRODUCTION
With the introduction of multi–detector row CT,
CT angiography has gained increasing acceptance for initial evaluation of patients who sustain major trauma and in suspected arterial injury.
Recently,
the growing widespread use of 64-row multidetector CT technology has offered considerable benefits in CT angiography in the trauma setting.
Sixty-four–row multidetector CT enhances imaging speed and allows submillimeter isotropic data sets.
CT angiography aids in the decision-making process to determine the appropriate treatment for each patient and injur.
Contrast material–enhanced computed tomography (CT) is the imaging technique of choice for the evaluation of abdominal trauma,
since it is widely available in emergency units and can quickly and accurately demonstrate damage to abdominal or retroperitoneal organs.
Active bleeding is easily depicted at multidetector CT.
Recognizing the imaging characteristics of these injuries is vital for optimization of patient treatment in emergencies.
In addition,
CT can help detect active hemorrhage,
urine leakage and delineating preexisting disease entities that may predispose to posttraumatic hemorrhage.
The main advantages of multidetector CT over single-detector CT are faster scanning times,
increased volume coverage,
and improved spatial and temporal resolution.
CT protocol for major trauma,
and routine CT usually includes a portal venous phase; which allows identification of parenchymal injuries.
Whenever vascular examination is necessary,
bolus-tracking multiphase CT can be performed during the arterial phase.
Because the imaging acquisition speeds have increased considerably with 64-row multidetector CT,
the margin for error in contrast bolus timing is minimal and accurate timing to ensure proper arterial phase imaging is paramount.
Another marked benefit of current multidetector CT technology is the advanced postprocessing features,
several of which are particularly relevant in CT angiography.The acquisition of isotropic voxel data sets has transformed CT into a fully multiplanar modality with the benefits of more accurate and confident diagnosis.
We currently employ the “direct MPR [multiplanar reformation]” function to routinely produce orthogonal reformations for every angiographic acquisition.
The ability to visualize vessels in any nonorthogonal plane is occasionally useful given their tortuous nature.
Not uncommonly,
further postprocessing options are performed by using picture archiving and communication system (PACS) integrated software.
Curved planar reformation at times can be an ideal postprocessing technique for vascular evaluation,
allowing visualization of the entire length of a vessel in one image.
Finally,
maximum intensity projection (MIP) images are useful in evaluating vascular injuries and,
in conjunction with a limited number of volume-rendered images,
can be of use when communicating with the clinical teams.
The use of the advanced postprocessing options including MIP,
volume rendering,
and curved planar reformation is employed on a case-by-case basis at the discretion of the interpreting radiologist by using the PACS incorporated software.
TECHNIQUE
It is used a standard delay or a timing bolus technique to ensure adequate arterial phase imaging,
depending on the type of CT studies requested.
Portal venous phase abdominopelvic imaging is performed by using a 70-second delay.
A 20-mL injection of intravenous contrast material is administered,
followed by a 30-mL saline “chaser,” by using a dual-syringe power injector with an injection rate of 5 mL/sec.
The timing of contrast material arrival is determined at the appropriately chosen level,
and 12 CT images are then acquired.The first image is acquired 5 seconds after the beginning of the timing bolus injection,
and the remaining 11 images are acquired every 2 seconds thereafter.
For the subsequent CT angiography,
image acquisition is started 5 seconds after the time to peak arterial enhancement shown on the time-attenuation curve of the test bolus series.This 5-second delay is added to the peak arterial enhancement time when a test injection technique is used to account for differences in time to peak enhancement between the test bolus and the subsequent contrast material injection,
which uses a larger volume of contrast material.
We routinely use a dose of 60 mL of intravenous contrast material (ioversol; Optiray (iodine concentration,
350 mg/mL)) at an injection rate of 5 mL/sec.
A 30-mL saline chaser is then administered,
also at a rate of 5 mL/sec.
ACUTE ABDOMINAL BLEEDING: ROLE OF MDCT
- Trauma
- Nontraumatic conditions
- vascular: rupture of aneurysm
- nonvascular: rupture of neoplasm
- upper & lower gastrointestinal tract
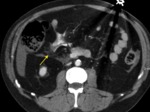
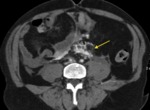
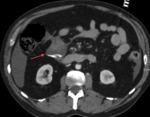
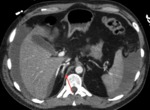
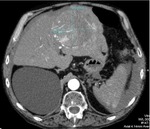
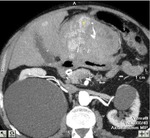
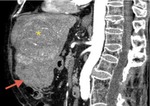
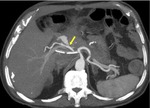
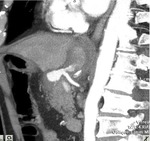
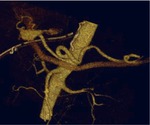
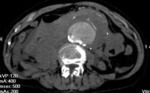
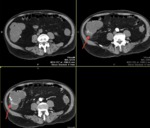
TRAUMA (FIG.1,2,3,4)
Patients in whom blunt abdominal and pelvic trauma is suspected require an accurate rapid assessment before treatment planning.
Important diagnostic findings include solid organ injuries such as laceration and infarction and associated active bleeding,
mesenteric and bowel injuries,
bladder rupture,
major vessel trauma,
diaphragmatic rupture,
and musculoskeletal injury to the lumbar spine and pelvis.
Trauma imaging studies are performed with contrast material enhancement.
Scan coverage is from the diaphragm to the inferior pelvis.
The major advantage of using multidetector CT for trauma cases is the ability to combine an arterial vascular phase study of the thorax (to look for traumatic aortic disruption) with a parenchymal phase examination of the abdomen.
In these combined thoracoabdominal studies,
contrast material is injected at 5 mL/sec.
If skeletal trauma is detected,
images can be reconstructed at 2.5mm thicknesses by using a bone algorithm with a limited field of view and 50% overlapped reconstructions.
This approach allows the generation of a three-dimensional volume data set for assessment of pelvic and thoracolumbar spinal fractures.
A) Bowel and Mesenteric Blunt Trauma
Numerous CT signs of bowel and mesenteric injuries secondary to blunt abdominal trauma have been described in the literature.
The main goal in evaluating these signs is to distinguish significant bowel and mesenteric injuries that require surgical intervention from those that can be managed nonsurgically.
Bowel and mesenteric injuries are detected in 5% of blunt abdominal trauma patients at laparotomy and are the third most common type of injury from blunt trauma to abdominal organs.
The common sites of blunt trauma injury in the small bowel are the proximal jejunum,
near the ligament of Treitz,
and the distal ileum,
near the ileocecal valve.
Delayed diagnosis of bowel and mesenteric injuries results in increased morbidity and mortality,
usually because of hemorrhage or peritonitis that leads to sepsis.
Although abdominal pain from peritoneal irritation may be a clinical manifestation of bowel and mesenteric blunt trauma injuries,
this symptom is nonspecific.
Patients in whom abdominal injury is suspected can be evaluated with various diagnostic tests,
including peritoneal lavage,
ultrasonography (US),
and computed tomography (CT).
Multidetector CT is more sensitive and specific than diagnostic peritoneal lavage,
abdominal US,
and clinical examination for the diagnosis of bowel and mesenteric injuries,
and it has become the diagnostic test of choice for the evaluation of blunt abdominal trauma in he modynamically stable patients.
The results of various studies show sensitivities of 69%–95% and specificities of 94%–100% for the diagnosis of bowel and mesenteric injuries with CT.
Findings Specific to Mesenteric Injury
Mesenteric Extravasation. This sign has a specificity of 100% for the diagnosis of significant mesenteric injury.
A finding of mesenteric extravasation is usually an indication for urgent laparotomy.
Mesenteric Vascular Beading.This feature appears on CT images as an irregularity in mesenteric vessels.
Like mesenteric extravasation of contrast material,
it is indicative of vascular injury.
It was observed more frequently than was active extravasation of contrast material.
Termination of Mesenteric Vessels. Abrupt termination of a mesenteric artery or vein is also an indication of vascular injury.
It has been saw more common than extravasation of contrast material.
Less Specific Findings
Mesenteric Infiltration. Haziness and fat stranding in the mesentery may indicate mesenteric injury with or without bowel wall injury.
This finding had the highest sensitivity for mesenteric injury,
but is nonspecific.
Mesenteric Hematoma. Well-defined mesenteric hematoma indicative of laceration of a mesenteric vessel.
Although specific to mesenteric injury,
mesenteric hematoma does not always indicate a need for surgery.
Bowel Features.
Bowel wall thickening and abnormal enhancement may be secondary to mesenteric injury,
indicating vascular compromise.
Bowel ischemia secondary to mesenteric injury may not be evident on initial CT images.
Common Features in Bowel and Mesenteric Injuries
Intraperitoneal and Retroperitoneal Fluid. The specificity is low for both intraperitoneal and retroperitoneal fluid because of other concomitant injuries.
The location of the fluid may indicate the location of injury.
Retroperitoneal fluid may indicate injury of a retroperitoneal segment of bowel.
Retroperitoneal blood tends to localize at the site of injury.
Hemoperitoneum in the absence of solid organ injury should raise the possibility of bowel or mesenteric injury.
Abdominal Wall Injury.
There is a significant association between abdominal wall injury (tear,
hematoma,
or “seat belt” sign [subcutaneous fat stranding along the course of the fastened seat belt]) and bowel and mesenteric injuries.
B) Acute Traumatic Aortic Injuries
Multidetector CT has been shown by several groups to have high accuracy in diagnosis and exclusion of acute aortic injuries.
CT findings include an aortic intimal flap or irregularity,
pseudoaneurysm,
aortic thrombus,
adventitial hematoma,
or aortic transection.
Diagnosis of minimal injury can be difficult,
particularly when only an intimal irregularity is seen.
Multiplanar reformatted images can be extremely helpful in further evaluation of the aorta and characterization of an injury and are helpful in therapy planning.
Management decisions are often based solely on the result of CT angiography and are greatly aided by the use of interactive advanced visualization workstations.
C) Active extravasation in blunt abdominal and pelvis trauma
The presence of active extravasation is an important indicator for morbidity and mortality in polytrauma patients because it denotes significant vessel or organ injury.
Active extravasation refers to administered contrast agent that has escaped from injured arteries,
veins,
bowel,
or urinary tract.
Active extravasation is seen in a minority of trauma patients in whom CT reveals a hematoma in the abdomen or pelvis.
Its detection with enhanced single phase CT,
however,
is limited when nonspecific areas of hyperattenuation are encountered; active extravasation is better distinguished with a dual phase CT protocol.
The classic pattern of active extravasation at dual phase CT is a jet or focal area of hyperattenuation within a hematoma on initial images that fades into an enlarged,
enhanced hematoma on delayed images.
This finding indicates significant bleeding and must be quickly communicated to the clinician,
since potentially lifesaving surgical or endovascular repair may be necessary.
Bleeding,
manifested as active extravasation at dual phase CT,
can be associated with other injuries to arteries,
such as a hematoma or a pseudoaneurysm.
Pelvic bleeding often occurs secondary to unstable pelvic fractures and is more often retroperitoneal compared with bleeding that occurs in cases of stable pelvic fractures.
Types of Vessel Injury
Signs of vascular injury seen at CT include hematoma and contrast material that surrounds the injured vessel.
Other clues to the location of vessel injury include lack of vascular enhancement (caused by occlusion or spasm),
vessel irregularity,
caliber change (such as occurs with pseudoaneurysm),
and an intimal flap (which signifies dissection).
For instance,
an intraluminal flap is pathognomonic of dissection,
but its presence could be occult because of occlusion,
spasm,
or dissection,
all of which would prevent contrast material from filling the occluded vessel.
The “contrast extravasation sign” seen at dual phase CT reportedly has greater than 95% accuracy,
negative predictive value,
and specificity with 80%–97% sensitivity and positive predictive value for identifying patients with pelvic fractures who require embolization to control bleeding.
Even in hemodynamically unstable patients with evidence of vessel injury at CT,
no bleeding will be found at angiography in 26%–44% of patients with abdominal and pelvic trauma.
In most cases,
the lack of angiographically evident bleeding can be attributed to the presence of venous or bone bleeding.
In some cases,
the findings vary because of inherent differences in the imaging modalities; the evaluation of vessel injury; and the timing of the performance of CT,
angiography,
and surgery.
Depending on the delay between examinations,
it is anticipated that some cases of active extravasation at CT may demonstrate other evidence of vascular injury but not contrast material extravasation at the time of angiography.
Given the nonselective nature of vessel enhancement and the relatively long injection time used in dual phase CT,
it is not possible to definitively diagnose the early draining vein seen in an arteriovenous fistula at CT.
Ultimately,
the role of CT may be to locate the bleeding vessel quickly in polytrauma patients.
Treatment decisions will be based on findings from subsequent angiography or surgery.
In the triage of polytrauma patients,
active extravasation represents current bleeding and is a better indicator of potential for continued bleeding or rebleeding than other forms of vessel injury that have not yet bled or have stopped bleeding.
The spleen is a frequently injured vascular organ with a large adjacent potential space in the intraperitoneal cavity.
Some treatment algorithms recommend use of follow-up CT to detect pseudoaneurysms not seen at initial imaging in patients with conservatively managed splenic injury.This follow-up may be deferred for younger individuals (<55 years old) with grade I injury.
If a pseudoaneurysm (or evidence of other vessel injury) is demonstrated at any point,
angiography is recommended.
Management options for active extravasation from splenic blunt trauma include arteriography and possible selective or subselective splenic artery embolization versus surgical splenectomy.
Active extravasation is a good predictor of the need for intervention in abdominal and pelvic trauma,
and it is even more indicative in cases of splenic injury.
Blood closest to the injury site has more time to retract,
forming higher-density clotted blood.
This phenomenon is known as the “sentinel clot sign”.
The sentinel clot sign is an important clue for locating the bleeding source when other more localizing findings of vessel injury are not present.The sentinel clot sign is present in every patient with hemoperitoneum,
whereas fewer injured patients demonstrate a positive sign of vascular injury,
such as active extravasation or pseudoaneurysm.
The earliest sign of decreased vascular volume affects the low-pressure vessel and can manifest as a flattened contour of the inferior vena cava.
A further decrease of vascular volume leads to development of a hypoperfusion complex,
which is characterized by decreased enhancement of the spleen and pancreas and increased enhancement of the bowel wall,
vasculature,
kidneys,
and possibly the adrenal glands.
Imaging findings can suggest a patient’s pending instability before it manifests clinically,
thus further expediting the performance of a lifesaving intervention.
D) solid organ injury
The spleen is the most frequently injured organ in blunt abdominal trauma; splenic injuries account for 40% of abdominal organ injuries.
Injuriesto the liver account for 20% of solid organ injuries related to blunt abdominal trauma.
However,
the liver is the single most commonly injured organ when blunt and penetrating trauma are combined.
There are numerous systems for classifying visceral injuries,
all of which include the following categories,
in some form: intraparenchymal contusion or hematoma,
subcapsular hematoma,
laceration,
fracture,
and vascular pedicle injury.
In the trauma setting,
CT can help accurately diagnose the type
and extent of the primary injury,
determine the presence and amount of hemoperitoneum,
and detect associated injuries.
The presence of small amounts of contrast material in the liver or spleen with adjacent hemoperitoneum may be indicative of arterial injury or pseudoaneurysm formation and should prompt surgical or angiographic intervention.
It has been reported that major vascular injury is more likely in the presence of high-grade liver lacerations that involve branches of the hepatic or portal veins or from which there is active bleeding,
and these findings should prompt angiography.
There is also a high likelihood of venous disruption and biliary injury in the presence of lacerations that extend into the region of the inferior vena cava or the major hepatic veins.
Fang et showed a significant correlation between the need for surgical intervention and the presence of hepatic and intraperitoneal bleeding or parenchymal contrast material extravasation in association with hemoperitoneum.
E) traumatic retroperitoneal injuries
Retroperitoneal injuries are known to occur in a significant minority of abdominal trauma cases.
Retroperitoneal injuries include duodenal,
pancreatic,
vascular,
renal,
and adrenal injuries.
Abnormal blood,
fluid,
or air within the retroperitoneal spaces may be isolated findings but can also occur in association with these injuries,
and their recognition is the key to correctly identifying the injury.
Retroperitoneal Hemorrhage
Retroperitoneal hemorrhage can be the source of significant but clinically occult blood loss in the trauma patient.
Retroperitoneal hemorrhage may arise from injuries to major vascular structures,
hollow viscera,
solid organs,
or musculoskeletal structures or a combination thereof .
Clinical management depends on the presence of active contrast material extravasation,
hematoma size and stability,
and the hemodynamic status of the patient.
Options include surgical intervention,
angiographic embolization,
and observation with fluid support.The goals of imaging are to identify the retroperitoneal hemorrhage,
its location,
and its possible source and to assess its relative stability on the basis of the size and presence (or absence) of active extravasation of intravascular contrast material.
NON TRAUMATIC CONDITIONS
A) acute gastrointestinal bleeding (FIG.12)
Acute gastrointestinal (GI) bleeding is a common major medical emergency.
Accurate and prompt diagnosis of the bleeding source is crucial because mortality can be as high as 40% if there is hemodynamic instability in patients.
Because of the length of the GI tract,
the multitude of pathologic processes that can result in GI bleeding,
and the often intermittent nature of GI bleeding,
imaging plays primary role in the diagnosis.
Upper GI bleeding occurs proximal to the ligament of Treitz and arises from the esophagus,
stomach,
or duodenum.
Lower GI bleeding involves the small bowel,
colon,
or rectum.
Depending on the amount of blood loss and location,
the clinical presentation varies considerably.
In many cases,
acute GI bleeding occurs intermittently or ceases spontaneously,
presenting a major diagnostic and treatment dilemma.
Even massive acute GI bleeding can be intermittent from minute to minute,
and failure to demonstrate active bleeding may therefore not prove cessation of bleeding in all cases.
Delayed follow-up examinations may then localize the bleeding.
Therefore,
to maximize detection capabilities,
it is crucial that CT angiography should begin as soon as possible while the patient is actively bleeding.
Multidetector CT angiography should be performed without prior oral administration of water or contrast material.
Active contrast material extravasation within the bowel lumen is obscured by oral contrast material,
leading to false-negative results.
Intraluminal water may be helpful in finding the cause of acute GI bleeding,
but it might also lead to dilution of extravasated contrast agent,
leading to false-negative results.
The diagnosis of acute GI bleeding is made when extravasated contrast material with a focal area of high attenuation (>90 HU) is seen within the bowel lumen during the arterial phase and increases during the portal-venous phase.The extravasation of contrast material may demonstrate a linear,
jetlike,
swirled,
ellipsoid,
or pooled configuration,
or may fill the entire bowel lumen,
resulting in a hyperattenuating loop.
To avoid pitfalls,
an unenhanced scan must be performed before intravenous contrast material injection.
False-positive multidetector CT results due to cone beam artifacts with hyperattenuating areas at the interface between normal bowel fluid content and air can also be obtained.
MPR and MIP images are useful for localizing the bleeding bowel segment and for determining the artery responsible for the bleeding in patients with an arterial source of bleeding.
CT angiography allows evaluation of the GI vasculature for preinterventional planning.
In cases of transcatheter treatment,
knowledge of the iliac and proximal femoral vasculature is useful for choosing the arterial approach.
Acute GI bleeding can have arterial and venous sources.
Venous bleeding within the upper GI tract is typically due to gastric or esophageal varices in the setting of portal hypertension.
However,
nearly 30% of patients with portal hypertension who have upper GI bleeding will have an arterial source of bleeding.The most common causes of upper GI bleeding are erosion or peptic ulcer disease.
Multidetector CT angiography can also help diagnose rare bleeding related to interventional endoscopic procedures such as endoscopic sphincterotomy of the ampulla.
The advantage of CT over strictly endoluminal procedures such as endoscopy is the ability to evaluate the condition precisely with regard to extraluminal abnormalities,
feeding and draining vessels,
and the anatomic region and its relationship to surrounding structures.This point is particularly important in cases of GI bleeding related to vascular lesions such as visceral artery aneurysm or erosion,
vascular enteric fistulas,
and vascular malformations.
Tumor,
inflammation,
and diverticular disease can be sources of both upper and lower GI bleeding.
Multidetector CT angiography is also helpful for the diagnosis of benign anorectal lesion bleeding.
B) Rupture of aneurysm (FIG.11)
A retroperitoneal hematoma adjacent to an abdominal aortic aneurysm is the most common imaging finding of abdominal aortic aneurysm rupture.
Periaortic blood may extend into the perirenal space,
pararenal space,
or the psoas muscles.
Intraperitoneal extension may be an immediate or a delayed finding.
These findings are readily visible on unenhanced CT images,
which may have been obtained for another indication or as part of an aneurysm evaluation protocol.
On contrast-enhanced CT images,
active extravasa- tion of contrast material is frequently demonstrated.
C) Tumor-associated Hemorrhage (5,6,7)
Spontaneous hemoperitoneum rarely occurs in the absence of trauma,
a surgical or interventional procedure,
or anticoagulation therapy.
In such cases,
the possibility of the rupture of an un- identified neoplasm must be excluded.
Although the occurrence is uncommon,
any primary or metastatic tumor can rupture and bleed into the peritoneal cavity.
The most common cause of nontraumatic hepatic hemorrhage is a hypervascular neoplasm such as hepatocellular carcinoma or hepatocellular adenoma.
Patients with large or peripherally located tumors devoid of normal overlying tissue are at a higher risk for rupture.
A wide spectrum of imaging findings can be seen,
ranging from minor intrahepatic bleeding to subcapsular hemorrhage and rupture through the hepatic capsule into the peritoneum.
Blood-tinged ascites is present in almost all cases.
Necrosis and hemorrhage also may occur in less common hypervascular hepatic tumors,
such as hepatic angiosarcoma.
Hemangiomas,
though common,
are not associated with nontraumatic hemoperitoneum.
Lung carcinoma,
renal cell carcinoma,
and melanoma are the metastatic lesions that most often cause hemoperitoneum.
Hemoperitoneum associated with a benign neoplasm most often occurs with hepatic adenoma.
These tumors are strongly associated with oral contraceptive use and estrogen steroid therapy; they most often are found in women of childbearing age who have a history of prolonged oral contraceptive use.
Large adenomas are prone to bleed,
and because the tumor capsule is incomplete,
a hemorrhage may spread into the liver or the abdominal cavity.
Pathologic splenic rupture may occur as a complication of a viral infection,
including infection by cytomegalovirus,
malaria,
or Epstein-Barr virus; a congenital disease; a metabolic abnormality such as Gaucher disease or amyloidosis; and,
rarely,
a neoplastic process such as hemangiomatosis,
angiosarcoma,
leukemia,
or lymphoma.
The prevalence of bleeding has been correlated directly with increased splenic weight.
At times,
even seemingly minor trauma,
such as that caused by lying prone,
can induce hemorrhage in patients with splenomegaly.
IATROGENIC CAUSES (8,9,10)
Even minimally invasive percutaneous or endovascular procedures occasionally lead to intraperitoneal hemorrhage.
Bleeding diatheses are common in patients who are undergoing anticoagulation therapy with heparin or coumadin.
In most cases,
the risk of bleeding is directly related to the intensity of the anticoagulant effect.
Anticoagulation therapy most commonly causes hemorrhage into the psoas or rectus muscles,
but it also occasionally results in hemoperitoneum.
Hemoperitoneum due to rupture of the gallbladder or spleen has been reported in patients undergoing anticoagulation therapy.
Like anticoagulation therapy,
blood dyscrasias may alter coagulation status and lead to bleeding.