Multi-Detector Row CT -Technique and Data Manipulation
- The imaging parameters used are: 120 kV,
70-120 mAs (variable values according caredose ®),
rotation time of 0.42 s,
0.75 mm collimation and "pitch" of 0.85.
The thickness of image reconstruction is 1 mm,
with an interval of 0.7 mm.
- The acquisition is performed with the patient in the supine position at maximal inspiration during a single breath-hold,
in a craniocaudal direction from the base of the neck to the middle third of the kidneys (renal arteries level),
to include the supraaortic great vessels and the infradiaphragmatic arteries.
- 100 ml of nonionic iodinated contrast was administered intravenously at a rate of 4 mL/ s
- The axial sections will be useful for detecting the origin of the systemic arteries.
Reconstructions in maximum intensity projection (MIP) will be essential to display the tortuous course of these arteries.
- The reconstructions in the coronal plane are better suited for analysis of the intercostal and internal mammary arteries;
- The axial reconstructions are ideal for demonstrating inferior phrenic arteries and branches from the celiac axis.
- The reconstructions in the oblique sagittal plane are also useful for demonstrating the mammary and phrenic arteries.
- The degree of obliquity and the thickness of the reconstructions we fit in each case.
The MIP reconstructions of the pulmonary arteries in different planes,
also will be necessary in suspected pathology of them.
Assessment of the lung parenchyma
-
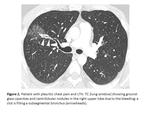
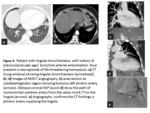
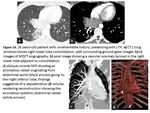
The most common signs of bleeding in the lung parenchyma are centrilobular nodules,
ground-glass opacities and / or condensation (Fig.
1.
b,
2). Clots can also be observe inside the bronchi,
which may secondarily produce atelectasis (Fig.
2).
If the damage is extensive and bilateral,
the multiplanar reconstructions are useful in assessing bleeding zonal predominance.
-
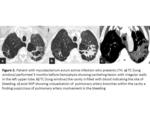
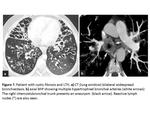
When there is cavities,
can be filled with blood (Fig.
3) and occasionally may identify hyperdense areas by the clots.
-
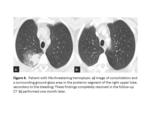
The blood clots may simulate nodules or masses,
which is why it is sometimes advisable to perform follow-up CT scan several weeks after the episode of hemoptysis,
to see the evolution of suspicious images (Fig.
4). Rarely extravasation of contrast can be seen in the bronchial lumen.
Assessment of the vascularization
Bronchial arteries
-
In 90% of cases,
the bronchial systemic arteries are the source of hemoptysis.
-
Bronchial arteries are considered those that go to the lungs through the pulmonary hilum,
along the bronchial tree.
The evaluation of bronchial arteries must detect:
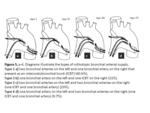
a) The site of the ostium of the bronchial artery (or arteries), to assess whether they are orthotopic or ectopic.
b) Description of the output in the aortic wall (anterior,
psterior,
left or right side).
It is also useful to report the presence of atherosclerotic plaques and the angle made by the vessel with the aorta,
if is very acute may difficult the catheterization
c) The bronchial artery diameter.
d) The total number of pathological bronchial arteries on each side.
-
The orthotopic bronchial arteries are those having an origin in the descending thoracic aorta at the level of the vertebral bodies T5-T6 (approximately in the region of the carina).
-
The bronchial arteries that origin outside the area between T5-T6 are called ectopic bronchial arteries.
-
The systemic arteries that do not reach the lung parenchyma through the pulmonary hilum are called nonbronchial systemic arteries.
-
The orthotopic bronchial arteries are highly variable in their anatomical origin,
branching pattern and course (Fig.
5).
-
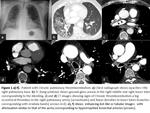
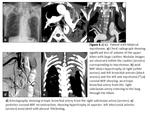
The most constant vessel is the right intercostobronchial trunk (ICBT),
present in 90% of cases (Fig.
1.
g-i).
-
In MDCT angiography the bronchial arteries are identified in the posterior mediastinum as dots or lines of increased attenuation (almost imperceptible if not hipertrofidas),
around the main bronchi,
esophagus and aortopulmonary window (Fig.
1.
e,f); MIP reconstructions are essential in different planes to represent its origin and courses (Fig.
1.
g).
-
A diameter of more than 2 mm in origin of bronchial artery is considered pathological and oriented toward the artery to be embolized; unfortunately there is a poor correlation between the size of the artery and the risk of bleeding.
-
Another important aspect is the traceability of bronchial artery causing hemoptysis.
- The bronchial artery aneurysms are rare entities that may arise within the mediastinum or from the intrapulmonary portion of the artery.
MDCT-angiography can show them (Fig.
7).
-
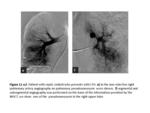
The ectopic bronchial arteries have a prevalence between 8.3% and 35%.
-
The most frequent ectopic origins are: the concavity of the aortic arch (74%), the subclavian artery ipsi-or contralateral (10.5%) (Fig.
6),
the abdominal aorta (8.5%), the ipsilateral brachiocephalic trunk (2%),
the ipsilateral internal mammary artery (2.5%) and the trunk thyrocervical ipsilateral (2.5%).
Nonbronchial systemic arteries
-
The nonbronchial systemic arteries are involved in 41% -88% of cases of hemoptysis.
-
May constitute the primary cause of bleeding or be an additional cause of bronchial arteries bleeding.
-
They enter the lung parenchyma through the pleura or the inferior pulmonary ligament and its course is not parallel to the bronchi.
-
Can originate from the: intercostal arteries (most often involved),
branches of the supaaortic great vessels ( brachiocephalic artery,
subclavian arteries,
thyrocervical and costocervical trunks),
axillary arteries,
internal mammary arteries and aortic infradiaphragmatic branches (inferior phrenic arteries,
gastric arteries and the celiac trunk).
-
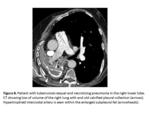
The presence of abnormally dilated and tortuous arteries within the extrapleural fat,
associated with pleural thickening (greater than 3 mm) and adjacent lung parenchyma abnormalities (bronchiectasis,
tuberculosis sequelae),
should make us suspect their involvement in hemoptysis (Fig.
6 ,
8).
-
Once the site of bleeding is located in MDCT-angiography,
a systematic search of the nonbronchial arteries that can potentially vascularize this area should be performed:
- the inferior phrenic artery (Fig.
9) (lower lobes and inferior segment of the lingula),
- the intercostal arteries (posterior pleura),
- the internal mammary artery (anterior segment of the upper lobes,
middle lobe and lingula) and
- branches of the subclavian and axillary arteries (the pulmonary apex).
Failure to recognize these systemic arteries can lead to early recurrence of hemoptysis after successful BAE.
-
We consider also congenital lung malformations with systemic irrigation,
rare entities that may cause LTH,
such as:
-
bronchopulmonary sequestration (mass of nonfunctioning lung tissue,
usually without connection to the normal bronchial tree) (Fig.
10) and
-
normal lung systemic irrigation (ISPN), a purely vascular anomaly,
usually a single systemic artery irrigates a normal lung portion.
-
In both cases,
the anomaly usually affects the lower lobes, the systemic artery often arise from the abdominal aorta (entering into the lung via inferior portion of the pulmonary ligament),
and the venous drainage is via the pulmonary veins.
- The possibility of congenital malformation as a cause of LTH may be suspected especially in young patients without known previous lung disease (Fig 10).
Pulmonary arteries
-
Assessment of the thoracic vasculature should always include the pulmonary arterial circulation.
-
Hemoptysis originating from the pulmonary artery represents 10% of cases of LTH.
-
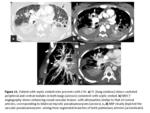
The aims of the evaluation is identifying aneurysms or pseudoaneurysms (PA,
artery dilation that not includes all layers of the wall) of the pulmonary arteries,
entities that are visualized on MDCT-angiography as saccular or fusiform dilatation of the pulmonary arteries filled with contrast,
simultaneously with the rest of the pulmonary arteries.
-
Visualization of a pulmonary artery branch in the inner aspect or within the wall cavity,
even without finding a PA,
suggests the possibility of a pulmonary artery origin of hemoptysis (Fig.
3).
-
MDCT-angiography with MIP and multiplanar reconstructions allow anatomic localization and the feeding artery of PA.
-
Sometimes the distal PA are not visible in the global or lobar pulmonary arteriography,
and are only displayed in superselective angiography of the pulmonary arteries (Fig.
11.
e,f).
-
Potential causes of pulmonary arterial origin hemoptysis are numerous and include:
- Pulmonary necrosis (Fig.
11)
-
Destructive or inflammatory lung process (infectious or cancer) destroy adjacent lung,
weaken the arterial wall,
or eroding any vessel in its vicinity,
causing a PA.
- Vasculitis
- Mainly associated with Behçet’s disease and Hughes-Stovin syndrome.
- Causing pulmonary artery aneurysms,
thrombosis and surrounding inflammation.
- Traumatic
- Iatrogenic causes,
mainly malpositioned Swan-Ganz catheter,
can developed in pulmonary artery pseudoaneurysm, that is contained in the adventitia and sometimes by thrombosis.
Penetrating trauma can cause it as well.
- Pulmonary Arteriovenous Malformations.
- Mainly congenital,
produce abnormal communication between pulmonary arteries and pulmonary veins.
Most of patients have hereditary Rendu -Osler Syndrome.
Rasmussen aneurysms is a pulmonary arterial pseudoaneurysms that develops in pulmonary vessels along the wall of tuberculous cavities.
- granulation tissue replaces external layers of the vessel wall,
with erosion of the artery and pseudoaneurysm formation.
- usually involves the upper lobes in the setting of reactivation tuberculosis
Causes of rebleeding
- After embolization immediate control of hemoptysis is achieved in 73-99% of patients.
- The recurrence is not uncommon,occurring in 10-53% of cases.
- Early recurrence,
within the first few weeks of embolization,
is caused by incomplete occlusion of the vessels involved,
which may be due to the extensive nature of the underlying cause,
or incomplete search for all abnormal vessels.
- Late rebleeding occurs due to recanalization of previously embolized vessels,
embolization of other vessels not involved,
or revascularization of collateral circulation secondary to persistence or progression of the underlying pathology.
For these reasons it is important to identify and embolize all vessels that may contribute to abnormal irrigation,
including any nonbronchial systemic or lung artery.