Intraxial Vascular Malformations:
A.
CVMs with arteriovenous shunting:
1.
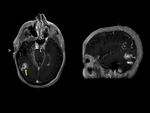
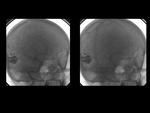
ARTERIOVENOUS MALFORMATION:
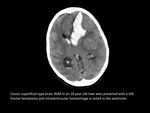
Brain AVMs,
or (more speci’¼ücally) pial AVMs,
are abnormal connections between arteries that would normally supply the brain tissue (pial vessels) and veins that normally drain the brain,
resulting in arteriovenous shunting with an intervening network of vessels within the brain parenchyma and lack of a true capillary bed.The transition between artery and vein can take place via a so-called nidus (a tangle of abnormal vessels located in the brain parenchyma) or can be direct (’¼üstulous) without any intervening network.
In the latter case,
the term brain arterivenous fistula or pial arteriovenous fistula is used.
Although brains AVMs are congenital lesions,
patients tend to present later in life,
most commonly with intracranial hemorrhage or seizures.
The imaging features of a nidus type brain AVM are consistent with its de’¼ünition.
The diagnostic criteria include
(a) The presence of a nidus embedded within the brain parenchyma,
identi’¼üed at either cross-sectional imaging (computed tomography [CT],
magnetic resonance [MR] imaging) or conventional angiography;
(b) Early venous drainage,
which is best seen on dynamic studies,
the standard of reference being convencional catheter angiography.
If a nidus is present,
two subtypes of abnormal Networks of vessels can be encountered.The tipical type is the glomerular or compact type nidus,
which consists of abnormal vessels without any interspersed normal brain tissue The more rarely seen second type is the so-called diffuse or proliferative type nidus,
in which normal brain parenchyma is interspesed thoroughout the tangle of vessels.
Arteriovenous malformation is the most common symtomatic cerebral vascular malformation with a mean age of preentation of 20 to 40 years.
The main symptoms are bleeding and headache.
All brain AVMs are potencially dangerous.
The risk of first hemorrhage is lifelong,
rises with age (2-4%/year,
cumulative)
Imaging findings:
Usually AVMs,
Ocur anywhere in brain and spinal cord but 85 % are supraentorial and 15% are in posterior fossa.
CT may be normal with very small AVMs,
but usually we show iso/ hiperdense serpentine vessels and contrast enhanced CT scans show intensely enhancing vascular structures.
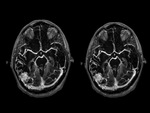
In MR,
signal varies with flow rate,
direction,
presence of hamorrhage.
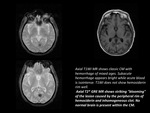
The classic Imaging finding is tightly packed mass loks like ŌĆ£honeycombŌĆØ of ŌĆ£flow voidsŌĆØ.
MRA is helpul for gross depictaion of flow,
post ŌĆōembo/XTR,
and MRV may be useful for delineating presence.
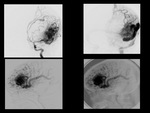
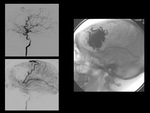
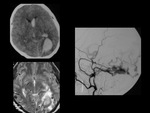
DSA is the best Imaging tool that delineates internal angioarchitecture and despicts three components of AVMs:
1.
Enlarged arteries.
2.
Nidus of tightly packed vessels.
3.
Draining veins (AV shuting with early appearance of contrast in enlaged veins).
DSA is very important for diagnosis of assciated anomalies such us:
1.
Flow ŌĆō related aneurisma of feeding artery.
2.
Intranidal ŌĆ£aneurismaŌĆØ
3.
Vascular ŌĆ£stealŌĆØ may cause ischemia in adjacent brain
We have several treatment options: embolization,
sterotaxic radiosurgery and microvascular surgery.
2.
PIAL FISTULAE ARTERIOVENOUS:
Pial AVFs represent a speci’¼üc subgroup of brain arteriovenous shunts and account for approximately 5% of all brain AVMs.
They constitute true pial arteriovenous shunts and consist of a direct ’¼üstulous communication between a pial artery and a vein without any intervening nidus.
Pial AVFs are located on the surface of the brain,
are often high ŌĆō flow lesions,
and in most instantes are associated with dilated venous pouches.
Pial AVFs are more commonly encountered in children and are frequently associated with hereditary hemorrhagic telangiectasia.
Pial AVFs can be congenital or result from a traumatic injury.
Little is known about their pathophysiological mechanisms.
Abnormal angiogenesis may play a role in the formation of pial AVFs,
and it is also possible that an embryological misstep could produce these lesions.
Pial AVFs can present as hemorrhage,
seizure,
neurological deficit,
cardiac failure in neonates and infants,
headache,
bruit,
symptoms of increased intracranial pressure in infants,
giant varices presenting as a palpable mass,
skull erosion and macrocephaly.
Congenital pial AVFs usually present during childhood along with syndromes such as Rendu-Osler-Weber disease and Klippel-Trenaunay-Weber syndrome.
Clues to the diagnosis of pial AVFs at cross-sectional imaging include the presence of dilated vessels,
mainly at the brain surface; and asymetric dilation of the pial feeding artery (the MCA,
anterior cerebral artery or PCA),
which is the best seen at the level of the circle of Willis.
These findings can be used to diferenciatiate pial AVFs fron dural AVFs and may be accompained by dilated Venus pouches outsider the brain parenchyma.
DSA is the best Imaging tool that delineates internal angioarchitecture.
Pial fistulas can be treated by neurointerventional techniques or by microneurosurgey.
Endovascular treatment offers a simple and safe option.
Recent advances in micro-catheter technology allow rapid,
safe and more distal intravascular navigation,
thus improving the results,
safety and reliability of endovascular treatment.
As endovascular techniques and technologies progress and the experience of the treating neuro-vascular team grow,
the microneurosurgical form of treatment will probably become uncommon.
B.
CMVs without arteriovenous shunting:
1.
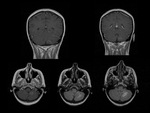
Capillary malformation (telangiectasia).
Capillary telangiectasiae(s) are small,
asymptomatic low flow vascular lesions of the brain. As these lesions are asymptomatic,
diagnosis usually matches the age of first imaging with MRI,
and as such are most frequently found in middle aged and elderly adults.
Their incidence varies according to the series.
They can account for up to 20 % of all intracerebral vascular malformations intracerebral vascular on autospy studies and are considered the second most common vascular anomaly alter venous angiomas (developmental venous anomaly) on imaging.
The majorities of capillary telangiectasias are completely asymptomatic and discovered incidentally on MRI,
when the brain is imaged for other reasons.
Occasionally there may be associated intracerebral haemorrhage although a direct causative relationship has not been established beyond a doubt.
They are comprised of dilated capillaries and are interspersed with normal brain parenchyma with a thin endothelial lining but no vascular smooth muscle of elastic fiber lining.
This is in contrast to cavenous malformations (cavernous haemangiomas) which have no normal brain within their confines.
Capillary telangiectasias are most are located within the brainstem and pontine regions,
and are more commonly multiple.
They have only become widely recognised in the radiology community following introduction of MRI,
as they are invisible both on CT and DSA.
Although CTSs are occasionally visible on computed tomography (CT) scans,
the ideal modality for detecting and imaging the lesions is contrast-enhanced MRI,
which should include a gradient-echo sequence (fast low-angle shot,
gradient-recalled echo [GRE]).
GENERAL FEATURES:
CT Findings
ŌĆó NECT: Usually normal (occasionally may have Ca++)
ŌĆó CECT: Usually normal
MR Findings
ŌĆó TIWI: Usually normal.
ŌĆó T2WI: 50% normal; 50% stippled foci of hyperintensity
ŌĆó FLAIR: Usually normal; may show hyperintense foci
ŌĆó T2* GRE : Lesion moderately but not profoundly hypointense
ŌĆó DWI: Usually normal
ŌĆó Tl C+
-Faint stippled or speckled "brush-like" enhancement
-May show punctate,
linear/branching vecollecting vein (if mixed with DVA)
As these lesions are almost always asymptomatic,
have interspersed normal brain tissue and are most frequently located in the pons,
makes treatment impractical and unwarranted.
No followup is thus required if the imaging appearances are characteristic.
2.
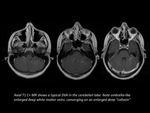
Developmental venous anomalies.
A developmental venous anomaly (DVA) also known as cerebral venous angioma,
is a congenital malformation of veins which drain normal brain.
They were thought to be rare pre cross sectional imaging,
however are now recognised as being the most common cerebral vascular malformatio accounting for 50 - 63% of all such lesions .
It is characterised by a caput medusa sign of veins draining into a single larger collecting vein,
which in turn drains into either a dural sinus or into a deep ependymal vein.
The appearance has also been likened to a palm tree.
Pathology
The etiology of DVAs remains uncertain,
but may relate to arrested development of venous structures.
Histologically they consist of a number of abnormally thickened veins with normal feeding arteries and capillaries
Location and classification
The most common locations are frontoparietal region (36 - 64%) ,
usually draining towards the frontal horn of the lateral ventricle and cerebellar hemisphere (14 - 27%) draining towards the fourth ventricle.
DVAs however can be seen anywhere,
draining both superficially or deep.
Associations
Lesions are usually solitary (75%),
except in blue rubber bled naevous syndrome blue.
There is also an association for venous malformations of the head and neck and in 8 - 33% of cases,
they are associated with cavernous malformations and are referred to as mixed vascular malformations.
Radiographic features
DVAs are seen on both CT and MRI as a leash of vessels draining towards a central vein.
CT
If large then the draining vein may be seen on non-contrast CT,
and is confirmed with contrast administration.
MRI
May be visible on most sequences,
but is most easily seen on post contrast T1 sequences.
If there is an associated cavernous haemangioma then susceptibility weighted sequences will be most sensitive for this component.
DSA - angiography
Angiographically the caput medusa appearance is seen only in the venous phase.
No shunting is present.
Treatment and prognosis
If isolated,
then no treatment is necessary.
If part of an MVM then treatment will be predicated by other component.
Informing the surgeon of the presence of a DVA is however essential as cautery of the the collecting vein can lead to venous infarcion
When isolated,
a DVA has a very low complication rate (0.15) mainly from spontaneous thrombosis of the collecting vein leading to venous infartaction and haemorrhage.
DVA's are however commonly associated (15 - 20%) with other vascular malformations,
most commonly a cavernous malformaation n which case they should be referred to as mixed vascular malformation.
Their risk of complication is then elevated to that of the associated lesion.
There is an association with cortical dysplasia.
3.
Cavernous Malformation:
Cavernous malformations are dilated blood vessels that are characterized by multiple distended "caverns" of blood-filled vessels through which the blood flows very slowly.
The vessels of a cavernous malformation lesion have a tendency to leak because they lack the necessary structural support - smooth muscle and stretchable material (elastin).
Leakage (bleeding) from these vascular lesions is the underlying cause of clinical symptoms associated with the illness,
cavernous malformation.
Cavernous malformations are primarily located in the brain,
but can also be found in the spinal cord,
on the skin,
and more rarely in the retina.
Cerebral cavernous malformations are present in up to 0.5% of the general population,
and they account for a large proportion (8-15%) of all brain and spinal vascular malformations.
While the prevalence of individuals with at least one CCM lesion is quite high,
as many as 40% of affected individual may never experience symptoms or become diagnosed with cavernous malformation
Associations
At times,
cavernous malformations may be associated with venous malformations.
It is increasingly frequent to recognize a venous anomaly adjacent to a cavernous malformation.
When these patients present with hemorrhage,
it is often the cavernous malformation that has bled and not the venous anomaly.
In fact,
it is exceedingly rare for a venous anomaly alone to hemorrhage.
Clinical Issues:
Most common signs/symptoms
o Seizure 50%
o Neurologic deficit 25% (may be progressive)
o 20% asymptomatic
Radiographic features
Given the fact that cavernous malformations are vascular malformations with low flow,
they are "occult" to angiography.
On CT scan one may see evidence of hemorrhage of various ages and with contrast administration the lesion itself may enhance.
MRI offers the most sensitive means of suspecting a diagnosis of cavernous malformation.
Blood products of varying ages may be present throughout the lesion.
This is usually mixed with the small 'caverns' of blood vessels themselves.
Often the lesion can be distinguished from adjacent hemorrhage.
It is now thought that hemorrhage can occur within the lesion or into surrounding brain parenchyma.
Certain individuals may have multiple lesions and careful inspection of the MRI study must be performed to rule out the possibility of small lesions that have not yet hemorrhaged.
In addition,
families have been noted with a history of cavernous malformation,
and multiple lesions are often present in family members.
Clinical Issues:
When patients present with recurrent hemorrhage,
progressive neurologic deterioration,
or intractable epilepsy,
then treatment in the form of surgery should be considered.
The decision to operate on a patient with a cavernous malformation must be made based on the exact location of the lesion and its surgical accessibility.
By and large surgery offers an excellent option in terms of complete excision of lesion with stabilization of symptoms.
Even seemingly deep- seated lesions can be reached using currently available stereotactic techniques.
Thus,
a relatively small lesion deep in the hemisphere can be reached through a small (1 cm or less) cortisectomy.
Another alternative is that of stereotactic radiosurgery.
While this has been used for patients with cavernous angiomas thought to be inoperable and associated with progressive worsening of neurologic symptoms,
the results are difficult to analyze.
The endpoint of treatment is difficult to assess in that cavernous malformations that have hemorrhaged will decrease in size simply by reabsorption of the blood around the lesion.
The only true test of whether radiosurgery (focused radiation) will offer protection from subsequent hemorrhage will be results obtained after following patients for long intervals of time.
This type of follow-up is currently in process and hopefully will shed light on this very issue.
The risks of focused radiation for cavernous malformations appear to be somewhat higher than true arteriovenous malformations of the same size.
The exact reason for this difference in complication rate remains unknown but is thought to relate possibly to the lower flow present in cavernous malformations.
In addition,
as with large true AVMs,
the risk of radiosurgery increases with larger lesions.
Extraxial Vascular Malformations:
DURAL A-V FISTULA:
Intracranial dural arteriovenous fistulas (AVFs) are abnormal arteriovenous shunts localised to the intracranial dura mater.
Some mechanisms that have been proposed for the development of dural AVFs are based on the theory of the opening up of pre-existing arteriovenous communications and the angiogenesis theory,
which suggests that angiogenesis is a result of intracranial venous hypertension.
In both theories,
cerebral sinus thrombosis (CST) may play an important role.
The shunt between the artery and sinus induces turbulent flow into the venous sinus that causes intimal injury leading to luminal thrombosis.
In addition,
secondary CST resulting from dural AVF may amplify venous hypertension and induce retrograde flow in the sinusthe groups for some features (including sex,
presence of CST,
location,
and type of AVF).
Dural AVFs with cortical venous re’¼éux will manifest with abnormal vessels that are present outside the brain parenchyma and must be differentiated from other vascular shunts (described later).
CT and MR imaging ’¼ündings include dilated cortical veins (a condition referred to as a pseudophlebitic pattern by some authors),
which manifest as abnormal enhancing tubular structures or ’¼éow voids within the cortical sulci with no true nidus within the brain parenchyma
Radiographic features
Hypoattenuation of the white matter at CT or hyperintense T2 signal at MR imaging indicates venous congestion or infarction,
which may eventually lead to venous hemorrhage.
Focal enhancement of these areas may also be observed as a sign of chronic venous ischemia.
Curvilinear subcortical calci’¼ücations can be seen at CT in patients with long-standing cortical venous re’¼éux,
possibly due to chronic venous congestion.
However,
cross-sectional imaging alone is often insuf’¼ücient to pinpoint the location of a dural AVF.
Dynamic studies with MR angiography,
CT angiography,
or digital subtraction angiography (the standard of reference) will demonstrate early venous ’¼ülling,
the contribution from external carotid artery branches (rather than pial vessels),
and shunt location.
Conventional angiography remains the most accurate method for detection and classification of DAVFs.
Clinical Issues:
The symptoms depend on their location and the pattern of the venous drainage.
Patients with cavernous sinus DAVFs often present with ocular manifestations such as exophthalmos,
chemosis and diplopia.
Patients with transverse or sigmoid sinus DAVFs frequently experience headache and tinnitus on the affected side.
DAVFs with anterograde sinus or cortical venous drainage (CVD) have been clinically regarded as benign,
whereas DAVFs with retrograde CVD are considered aggressive in behavior.
Natural History & Prognosis
ŌĆó Prognosis,
clinical course depends on location,
venous drainage pattern:
Anterior fossa,
tentorial,
SSS or SPS location correlated with aggressive course (hemorrhage,
encephalopathy,
progressive neurologic deterioration)
98% of dAVFs without RLVD have benign course
dAVF with RLVD have aggressive clinical course
ŌĆó Spontaneous closure rare
Treatment
ŌĆó Observation (benign dAVF carries only 2% risk of developing CVD)
ŌĆó Treatment options: Endovascular,
Surgical resection o Stereotaxic radiosurgery