Results:
From January 2000 to November 2012,
363 (6%) patients in our institution,
from a total of 6041 cases of malignant breast neoplasms,
had confirmed diagnosis of the above referred unusual malignant breast tumors by imaging guided biopsy (see Fig.
1).
362 patients underwent both mammography and ultrasonography; 1 patient was studied only by ultrasonography; and the majority of the patients performed MR imaging.

Fig. 1: Table 1 and Graphic 1 - Malignant Breast Tumors
Of these,
- 2,7% (n=163) were due to well-differentiated subtypes of Invasive Ductal Carcinoma (1,4% (n=85) Mucinous Carcinoma; 1,1% (n=68) Papillary Carcinoma; 0,15% (n=10) Tubular Carcinoma) (see Fig.
2);
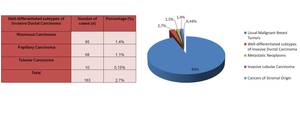
Fig. 2: Table 2 and Graphic 2 - Unusual Malignant Breast Tumors: Well-differentiated subtypes of Invasive Ductal Carcinoma
- 1,5% (n=90) to Metastatic neoplasms (0,65% (n=38) Metastatic Carcinoma; 0,61% (n=36) Metastatic Melanoma; 0,17% (n=10) Non-Hodgkin Lymphoma; 0,08% (n=5) Rhabdomyosarcoma and 0,02% (n=1) Metastatic Myeloma) (see Fig.
3);

Fig. 3: Table 3 and Graphic 3 - Unusual Malignant Breast Tumors: Metastatic Neoplasms
- 1,4% (n=84) to Invasive Lobular Carcinoma (see Fig.
1);
- 0,44% (n=26) to Cancers of stromal origin (0,28% (n= 16) Phyllodes Tumor; 0,13% (n=8) Adenoid Cystic Carcinoma and 0,03% (n=2) Angiosarcomas) (see Fig.
4).
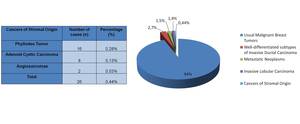
Fig. 4: Table 4 and Graphic 4 - Unusual Malignant Breast Tumors: Cancers of Stromal Origin
Discussion of the results:
The results of our study are globally concordant with the available literature,
either in terms of the type of tumors described and of its respective prevalence.
Literature review:
Unusual malignant breast tumors may be widely divided into well-differentiated subtypes of invasive ductal carcinoma,
metastatic neoplasms,
invasive lobular carcinoma and cancers of stromal origin.
Well-differentiated Subtypes of Invasive Ductal Carcinoma
Most breast cancers are of ductal origin,
with IDC not otherwise specified with lack of distinctive morphologic features being the most common type.
However,
several subtypes of ductal carcinoma,
exhibit differentiation by producing tubules,
mucin,
and cetera.
These subtypes of ductal carcinomas are then named according to their morphologic differentiating features.
Because these cancers are well differentiated,
they grow slowly (with the exception of medullary carcinoma) and are often relatively circumscribed at imaging (with the exception of tubular carcinoma).
These well-differentiated tumors usually have an excellent prognosis.
However,
the prognosis can be worse in the mixed-type tumors (less than 90% of the differentiated type),
since it is determined by the least differentiated portion of the tumor.
Mucinous Carcinoma
Mucinous carcinoma accounts for 1%-7% of all breast cancers [1].
This cancer usually occurs in women over 60 years old.
Mucin is a dominant feature at histologic examination of these tumors and they can be divided into pure or mixed types depending on the concentration of the mucin content.
Pure mucinous carcinoma is associated with a better prognosis and a lower incidence of axillary lymph node metastases.
If palpable,
these cancers tend to manifest as soft masses.
At mammography,
tumors with high mucin content typically manifest an oval shape with low density,
relatively well-defined or microlobulated or lobular margins/contour,
because of the richness in mucin (see Fig.
5).
An irregular margin is identified in the tumors with lower mucin content.
On US scans,
mucinous carcinomas are often heterogeneous in echogenicity,
and in more than one third of the cases,
they can have both solid and cystic components.
Posterior acoustic enhancement is common,
in opposite to posterior acoustic shadowing which is very uncommon (see Fig.
5).
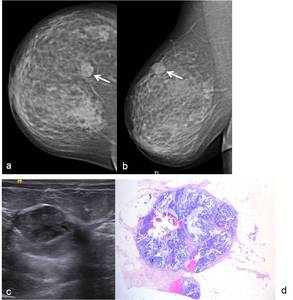
Fig. 5: 75-year-old woman. Right (a) CC (craniocaudal) and (b) MLO (mediolateral oblique) views show at 12 o’clock, a low-density circumbscribed mass with irregular margins (arrows). (c) On US scan this mass show heterogeneous echogenicity and posterior acoustic enhancement. Core needle biopsy revealed a mucinous carcinoma. (d) Hematoxylin-eosin, low-power view and with amplification of 200x shows a proliferation of clusters of generally uniform, round cells with minimal amounts of eosinophilic cytoplasm, floating in lakes of mucous. Delicate fibrous septae divide the mucous lake into compartments.
On MR images,
mucinous carcinomas are one of the few cancers that have high signal intensity on T2-weighted images,
due to the watery nature of mucin (see Fig.
6).
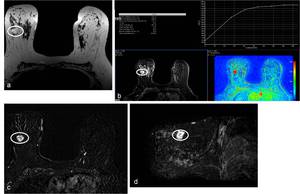
Fig. 6: 75-year-old woman with right breast mucinous carcinoma. (a) This lesion has high signal intensity on axial T2-weighted images (ellipse), and show progressive enhancement on (b) axial dynamic studies and on (c) axial and (d) sagittal subtraction images.
Histopathological examination demonstrates a great amount of mucin lakes presenting a pure type of carcinoma.
Mixed type tumors have usually lower mucin concentration rates and have more irregular margins due to infiltrative tumor cells at the margins (see Fig.
5).
Papillary Carcinoma
Papillary carcinoma accounts for 1%-2% of breast cancers [1].
It generally occurs in women over 60 years old.
This type of carcinoma frequently manifests as an intraductal or intracystic mass.
Patients with papillary carcinoma often present a palpable central mass or bloody nipple discharge.
It is characterized by slow growth and better prognosis compared to ductal carcinomas not otherwise specified.
The frequency of axillary nodal metastases is low.
The clinical and imaging findings of papillary carcinoma can be similar to those of benign intraductal papillomas,
since both of them represent most frequently as centrally located intraductal masses and clinically present with nipple discharge.
The mammographic appearance consists of a solitary round (see Fig.
7) or lobulated (see Fig.
8),
relatively circumscribed mass (see Fig.
7) or as a cluster of masses,
with equal- (see Fig.
7) or high-density (see Fig.
8).
Associated microcalcifications can also be identified (see Fig.
8).
This tumor can mimic a benign ductal papilloma at mammography.
On US,
papillary carcinoma can manifest as a single (see Fig.
7 and 8) or multiple circumscribed solid mass,
as a complex cystic mass,
or as a cyst with an intracystic mass (see Fig.
9).
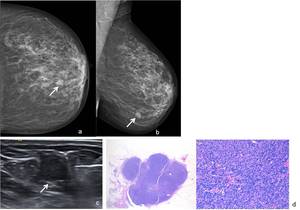
Fig. 7: 75-year-old woman. Left (a) CC and (b) MLO mammographic views show a round, relatively circumscribed mass, with isodensity on the transition of the lower quadrants. (c) On US scan the mass consisted of a round circumscribed solid mass. US-guided core needle biopsy was performed, revealing a solid papillary carcinoma.(d) Hematoxylin-eosin, low-power view and with amplification of 200x shows that there is solid cellular epithelial nodules arranged in geographic, jigsaw-like pattern within a fibrous, focaly desmoplastic background. At higher magnification one can observe fibrovascular cores within some of the solid nests.
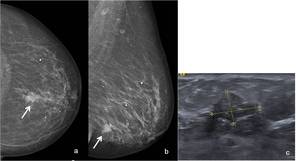
Fig. 8: 63-year-old woman. Left (a) CC and (b) MLO mammographic views show a nodular high-density lesion with irregular and spiculated contours (arrows) and with pleomorphic microcalcifications. It is located on the transition of the lower quadrants. (c) On US scan that lesion manifests as a circumscribed solid mass, with mixed echogenicity and lobulated contours. US-guided core needle biopsy revealed an invasive papillary carcinoma.
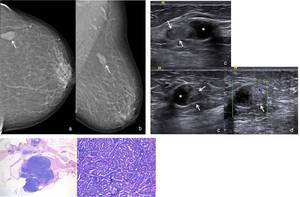
Fig. 9: 79-year-old woman. Left (a) CC and (b) MLO mammographic views show a lobulated circumscribed mass with equal-density in the superior outer quadrant. (c) On US scan this lesion consisted of a circumscribed cyst (*) with an echogenic component which had a solid nature, as demonstrated on Color Doppler US (arrows) (d). US-guided biopsy of the intra cystic mass was performed, revealing an Encapsulated Papillary Carcinoma (WHO 2012). (e) Hematoxylin-eosin, low-power view and with amplification of 400x. At scanning magnification one can observe an encapsulated papillary carcinoma. It is composed of papillary frond composed of a monotonous epithelial proliferation with absence of myoepithelial cells both within and at the periphery of the lesion.
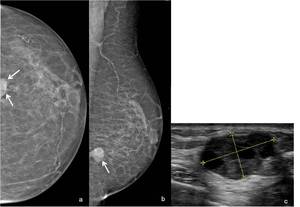
Fig. 10: 80-year-old woman. Left (a) CC and (b) MLO mammographic views show a high-density and relatively circumscribed mass, with a posterior location in the transition of the lower quadrants. (c) On US scan this lesion manifests as a partially solid and cystic mass with lobulated contours. US-guided core needle biopsy revealed an invasive intracystic papillary carcinoma.
Intracystic carcinomas (see Fig.
9 and 10) account for less than 1% of breast cancers and are usually papillary carcinomas.
Aspiration of these lesions often yields bloody fluid.
On histopathologic examination,
the tumor can have in situ (intracystic or intraductal) or invasive components (see Fig.
7).
In the in situ component,
the neoplastic epithelial cells are usually characteristic of low-grade ductal carcinoma in situ.
The absence of a uniform myoepithelial layer distinguishes papillary carcinomas from benign papillary lesions.
Tubular Carcinoma
Tubular carcinoma accounts for 1%–2% of all breast cancers [1].
It is most common in women aged 40 to 60 years.
The prognosis is excellent,
with 94% 5-year survival rate.
A minimum of 75% tubular elements is required for the diagnosis of tubular carcinoma.
Because of their small size,
these lesions are often nonpalpable and are usually detected incidentally through screening mammography.
On mammography,
this tumor appears as one or more small spiculated masses.
The spicules are often longer than the central mass (see Fig.
11).
This appearance mimics infiltrating ductal carcinoma and radial scars.
Tubular carcinoma should be considered in the differential diagnosis of one or more adjacent spiculated masses,
especially if the masses are nonpalpable and have long spicules.
On US,
tubular carcinoma is a hypoechoic solid mass with ill-defined margins and posterior acoustic shadowing (see Fig.
11),
mimicking IDC not otherwise specified.
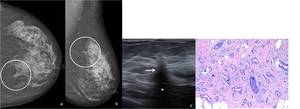
Fig. 11: 59-year-old woman. Left (a) CC and (b) MLO mammographic views show a small mass with very and long spiculations on the upper internal quadrant. (c) On US scan this lesion corresponded to a hypoechoic solid area with ill-defined margins (arrows) and posterior acoustic shadowing (*). Core needle biopsy revealed invasive tubular carcinoma. (d) Hematoxylin-eosin, with amplification of 200x shows a haphazard distribution of rounded and angulated tubules with open lumens, lined by only a single layer of epithelial cells separated by abundant reactive, fibroblastic stroma.
Histopathologic examination of tubular carcinomas reveals small round or oval tubules of ductal epithelium that infiltrate the stroma in a haphazard pattern.
The lumina of the tubules is lined by a single type of ductal epithelium cell that abuts directly onto the adjacent stroma with scarce underlying myoepithelial cells ( see Fig.
11).
Tubular carcinoma may occasionally be difficult to differentiate from radial sclerosing lesion (radial scar) at histologic examination.
Metastatic Neoplasms
Breast metastases account for 0.5%–2% of breast carcinomas [1].
Metastatic disease should be considered when there isbilateral axillary adenopathy or multiple breast masses.
These lesions tend to be superficially located in the upper outer quadrant.
They are much more likely to be multiple or bilateral compared to primary cancers.
The most common extramammary cancers that metastasize to the breast are lymphoma,
melanoma,
soft tissue sarcoma,
and lung,
ovarian,
renal cell and cervical carcinoma.
The mammographic appearance usually consists on a round mass with circumscribed or ill-defined margins.
On US,
circumscribed,
hypoechoic lesions are their usual features.
Axillary node involvement is frequently present.
Metastatic Carcinoma
The most common extramammary carcinomas that metastasize to the breast are lung,
ovarian,
renal cell and cervical carcinoma.
At clinical examination,
discrete masses are typically palpable,
although the breasts may become firmer without discrete palpable lumps.
Metastatic carcinomas in the breast typically manifest as multiple round or oval circumscribed masses (see Fig.
12 and 13).
The margins may appear circumscribed,
though often become ill-defined on spot compression views (obtained with or with-out magnification) or US scans.
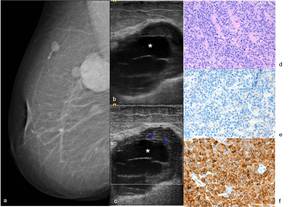
Fig. 12: 53-year-old woman with history of neuroendocrine lung cancer. Right (a) MLO mammographic view show a high-density circumscribed mass located posteriorly in the upper quadrants. Other adjacent smaller lesions with the same mammographic appearance are observed. (b) On US scan this lesion manifests as a hipoechogenic and heterogeneous mass with a peripheral solid component and central cystic areas (*). (c) On Color Doppler US scan the peripheral solid component of the mass shows blood flow. US-guided core needle biopsy was performed in the solid portion of the biggest lesion. (d) Hematoxylin-eosin, with amplification of 400x – Metastatic Neuroendocrine carcinoma to the breast showing negativity to GCDFP-15 (e) and immunoreactivity to synaptophysin (f).
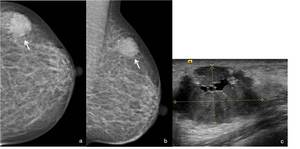
Fig. 13: 44-year-old woman with history of cervical carcinoma. Left (a) CC and (b) MLO mammographic views show a oval mass, with isodensity and microlobulated contours located on the upper outer quadrant. (c) On US scan this lesion corresponded to a hipoehogenic mass with small internal cystic areas and microlobulated contours. US-guided core needle biopsy was performed, revealing a metastatic cervical carcinoma.
Metastatic Melanoma
Metastatic melanoma may manifest as multiple round or oval circumscribed masses similar to other metastatic carcinomas (see Fig.
14).
It should also be considered when there is unilateral adenopathy or enlargement of an intramammary lymph node,
particularly when the primary lesion was on the back or upper abdomen.
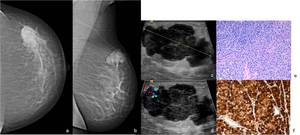
Fig. 14: 44-year-old woman with previous history of melanoma and recent complain of a lump in the left breast. Left (a) craniocaudal and (b) MLO mammographic views show a high-density oval lesion with lobulated contours in the upper outer quadrant. (c) On US scan this lesion corresponded to an heterogeneous solid mass with internal and peripheral blood flow on Color Doppler imaging (d). US-guided biopsy was performed. (e) Hematoxylin-eosin, with amplification of 400x – Metastatic Malignant melanoma to the breast showing immunoreactivity to HMB-45 (f).
Non-Hodgkin Lymphoma
Breast lymphoma is a rare disease representing only 0.1%–0.5% of all breast cancers [1].
Secondary involvement is more common and is almost always of non-Hodgkin’s type.
Non-Hodgkin Lymphoma is most frequently of concern when bilateral axillary adenopathy is seen on the mammogram.
The most common mammographic manifestation of non-Hodgkin’s lymphoma,
whether primary or secondary,
is one or more spherical or ovoid noncalcified masses with circumscribed,
relatively circumscribed or indistinctly margins (see Fig.
15).
Diffuse increased parenchymal density in combination with skin thickening can be seen.
Bilateral axillary adenopathy is usually present.
At ultrasonography it manifests as a hypoechoic mass with circumscribed or irregular margins.
Mixed echogenicity also may be seen.
Posterior acoustic enhancement is a common feature noted at US,
and an echogenic rim may be seen around the lesions (see Fig.
15).
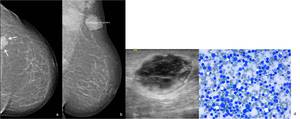
Fig. 15: 77-year-old woman with history of Non-Hodgkin Lymphoma (NHL). Left (a) CC and (b) MLO mammographic views show a high-density mass posteriorly located in the upper outer quadrant. Homolateral axillary adenopathies can also be seen. (c) On US scan the lesion corresponded to a hypoechogenic and heterogeneous mass with partial cystic component and an outer echogenic rim. US-guided core needle biopsy was performed. (d) May-Grunwald Giemsa, with amplification of 400x – Metastatic Non-Hogkin Lymphoma to the breast.
At histopathologic examination,
the most common type is a diffuse large cell (histiocytic) lymphoma (see Fig.
15).
Core biopsy rather than fine-needle aspiration biopsy should be performed,
as lymph node architecture is needed to make a definitive diagnosis of lymphoma.
Rhabdomyosarcoma
Breast involvement by rhabdomyosarcoma is rare.
It typically occurs in the context of advanced metastatic disease,
although it may also be the first sign of recurrent disease ( see Fig.
16).
The appearance on a mammogram is nonspecific,
manifesting as a focal or global asymmetry.
Rhabdomyosarcoma infiltrate appear as ill-defined areas of shadowing at US.
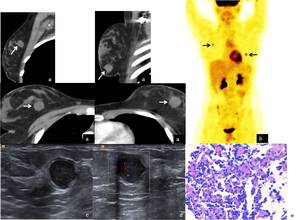
Fig. 16: 17-year-old girl with history of perineal rhabdomyosarcoma. (a) On chest CT, one lesion was detected in each breast. (b)At positron emission tomography (PET) scan these lesions showed hypermetabolic behavior. (c) On US scan, these lesions were solid, homogeneous and well – circumscribed. (d) Color Doppler US scan shows blood flow within the lesions. US-guided biopsy was performed. (e) Hematoxylin-eosin, with amplification of 200x – Metastatic Rhabdomyosarcoma to the breast.
Metastatic Myeloma
Extramedullary plasmacytomas most commonly occur in patients with a previous diagnosis of multiple myeloma.
Plasmacytomas of the breast are extremely rare.
The mean age ofoccurrence of breast plasmacytomas is 53 years.
It may present as a solitary finding or in association with disseminated multiple myeloma.
More than half of the lesions are unilateral,
with the majority of the cases occurring in the setting of multiple myeloma.
Plasmacytoma in the breast may manifest as multiple round or oval circumscribed masses similar to metastatic carcinomas.
The mammographic findings may consist of single or multiple well-defined masses without spiculations and microcalcifications (see Fig.
17).
It may also present as a relatively well-circumscribed mass without calcifications,
as an ill-defined focal asymmetry on a mammogram or rarely as a mass with partially spiculated margins.
Sometimes these lesions may have partial,
lucent halo signs.
On US,
the masses may be either well-defined or ill-defined.
They are round and hypoechoic with posterior acoustic shadowing or enhancement or hypoechoic masses containing central and peripheral hyperechoic rims (see Fig.
17).
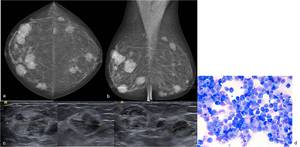
Fig. 17: 69-year-old woman with history of multiple myeloma. (a) CC and (b) MLO right (left) and left (right) mammographic views show multiple and bilateral high-density masses. (c) On US scan these lesions corresponded to relatively ill-defined and heterogeneous masses. US-guided biopsy of the largest masses was performed. (d) May-Grunwald Giemsa, with amplification of 400x – Metastatic Multiple Myeloma to the breast.
Histologically,
these tumors shows a monotonous population of cells with hyperchromatic eccentric nuclei and clumped chromatin,
confirming myeloma infiltration (see Fig.
17).
Invasive Lobular Carcinoma
Invasive Lobular Carcinoma (ILC) accounts for 6%–9% of breast cancers [2].
ILC typically spreads as sheets of a single-cell layer along Cooper ligaments and other structures in the breast,
somewhat like a spider web,
rather than as a mass.
Because of this infiltrative growth pattern,
resulting in decreased compliance of the tissue,
ILC is more difficult to detect at clinical examination and mammography than is IDC.
Therefore,
ILC usually is larger and multifocal at diagnosis.
The most common clinical findings of ILC are palpable thickening and skin or nipple retraction.
When large,
a firm palpable mass may become evident at clinical examination.
Usually,
the clinical examination findings are more suspicious for breast carcinoma than the imaging findings.
ILC also has a propensity for metastatic spread to the peritoneum,
retroperitoneum,
and gynecologic organs and therefore,
the diagnosis of metastatic involvement should be considered in women presenting with ascites,
hydronephrosis,
or pelvic masses.
Mammography often grossly underestimates the size of ILC relative to the physical examination findings and histologic examination.
At mammograhy,
the most frequent manifestation of ILC is indeed architectural distortion with or without a central mass or a focal asymmetry (see Fig.
18,
20 and 22).
Calcifications are a very uncommon feature.
Unlike IDC,
ILC is more frequently seen in only one view,
most commonly in the craniocaudal view,
which typically has better compression than the MLO view,
turning the spider web lines more obvious.
When ILC is large,
the affected breast may appear to be decreasing in size on the mammogram; this has been termed the “shrinking breast”.
Such shrinkage is probably due to the sheets of tumor cells causing decreased compressibility of the breast.
At clinical inspection,
the physical size of the breast is not different,
so the shrinking breast is a mammographic and not a clinical finding of ILC.
At US,
the numerous sheets of tumor cells will frequently cause architectural distortion,
with hypoechoic and heterogeneous internal echoes,
ill-defined or spiculated margins,
and posterior acoustic shadowing,
often without a discrete mass (see Fig.
18,
20 and 22).
Occasionally,
it may be difficult to distinguish the mild posterior acoustic shadowing that can be seen with fibrocystic changes from that of ILC.
The application of firm pressure with a transducer will often remove the mild shadowing in benign fibrocystic changes.
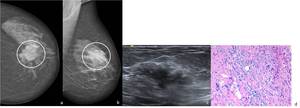
Fig. 18: 67-year-old woman. Left (a) CC and (b) MLO mammographic views show an ill-defined iso to high-density opacity in the transition of the superior quadrants. (c) On US scan this lesion manifested as an area of architectural distortion surrounding a lobulated mass with an hypoechoic center and an echoic rim at the periphery. Core-needle biopsy revealed a ILC. (d) Hematoxylin-eosin, with amplification of 200x shows a proliferation of small cells, which lack cohesion and individually dispersed through a fibrous connective tissue appear arranged in single file linear cords that invade the stroma with little host reaction or disturbance of the background architecture. The neoplastic cells have round or notched ovoid nuclei and a thin rim of cytoplasm.
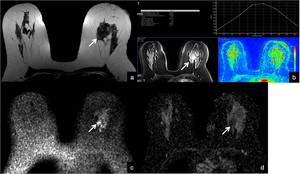
Fig. 19: 67-year-old woman with left breast ILC. (a) This lesion as irregular margins and low signal intensity on axial T2-weighted images (arrow), and (b) show enhancement on axial dynamic studies. (c) In Diffusion weighted sequence (b=1000), water restriction produces hiperintensity in the tumor area , matching the low signal in the ADC map (d).
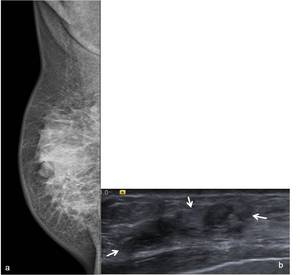
Fig. 20: Images in a 65-year-old woman that refers densification of the right breast and nipple retraction. (a) Right MLO mammographic view shows a breast with small dimensions and a retroareolar architectural distortion and densification. (b) On US scan it corresponded to an heterogeneous solid mass, with irregular margins and discrete posterior acoustic shadowing. Core-needle biopsy revealed a ILC.
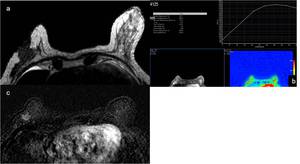
Fig. 21: Images in a 65-year-old woman that refers densification of the right breast and nipple retraction and diagnosis of lest breast ILC. (a) On MRI, the ILC corresponds to a mass with irregular margins and low signal intensity septa on axial T2-weighted images (arrows), (b) showing enhancement on axial dynamic studies and (c) axial subtraction images.
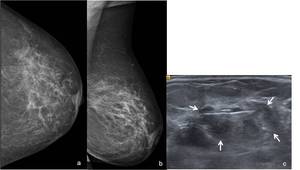
Fig. 22: 81-year-old woman complaining of left breast tenderness, skin thickening and nipple retraction. Left (a) CC and (b) MLO (right) mammographic views show an architectural distortion in the retroareolar area, as well as nipple retraction and adjacent skin thickening. (c) On US scan it corresponds to a ill-defined solid heterogeneous mass (arrows). Core-needle biopsy revealed a ILC.
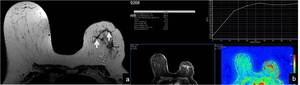
Fig. 23: 81-year-old woman complaining of left breast tenderness, skin thickening and nipple retraction, with the diagnosis of a left breast ILC. (a) On MRI, this lesion corresponds to multiple low signal intensity septa on axial T2-weighted images (arrows), (b) showing enhancement on axial dynamic studies.
On MR images,
ILC may manifest as an enhancing solitary mass with irregular or ill-defined margins,
multiple enhancing lesions,
or only enhancing septa (see Fig.
19,
21 and 23).
ILC has a characteristic histopathologic appearance consisting of small,
uniform tumor cells with round nuclei and scanty cytoplasm arranged in a classic single-file pattern (see Fig.
18).
It has a tendency to spread diffusely or between the collagen fibers of the breast and produces little desmoplastic response.
Cancers of Stromal Origin
Breast tumors may originate from the supporting tissue of the breast,
including fibromuscular tissue,
blood vessels,
and the lymphatics.
These tumors have diverse appearances but clinical history and imaging features often raise the suspicion.
Phyllodes Tumor
Phyllodes tumor accounts for less than 1% of all breast cancers [2].
The average age at presentation is 40–50 years.
In Asian countries,
they occur at a younger age (average,
25–30 years) [3].
Typically manifests as a rapidly enlarging palpable unilateral breast mass.
These tumors may develop de novo or from existing fibroadenoma.
Phyllodes tumor was previously known as cystosarcoma phyllodes which has been considered an inappropriate designation,
since most of these tumors are benign.
They can be categorized as benign,
borderline,
or malignant.
Malignant phyllodes tumors may contain sarcomatous elements such as chondrosarcoma,
osteosarcoma,
and leiomyosarcoma.
The margins of these lesions may become less defined.
Prognosis is poor for lesions containing sarcomatous elements as they tend to follow the behavior of sarcoma.
They usually metastasize hematogenously to the lungs or bones.
Mammographically,
phyllodes tumors usually manifest as a very large,
relatively circumscribed,
round or oval mass (see Fig.
24 and 25).
The mass may become extremely large.
At close inspection,
the margins are often ill-defined rather than sharply circumscribed.
When detected at a screening examination,
the appearance may mimic a fibroadenoma.
At US,
this tumor may resemble a fibroadenoma,
though phyllodes tumor more frequently contains cystic spaces (see Fig.
24 and 25) than does a benign fibroadenoma.
The margins of a benign fibroadenoma are sharply defined on US scans,
whereas the margins of a phyllodes tumor may be ill-defined (see Fig.
25).
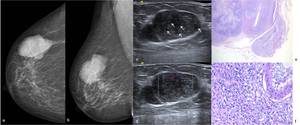
Fig. 24: 77-year-old woman not using menopausal hormone therapy, that complains of a rapidly increasing lesion on her right breast. Right (a) CC and (b) MLO mammographic views show a large lobulated and high-density mass on the in the upper outer quadrant . (c) On US scan, this hypoechoic lesion is heterogeneous and several cleftlike cystic spaces are identified within it (arrows) (d) On Color Doppler US scan the mass shows blood flow. US-guided core needle biopsy was performed. (e) Hematoxylin-eosin, low-power view and with amplification of 400x – Phyllodes tumor. The tumor exhibits a predominantly pushing margin with the characteristic leaf-like pattern and a well defined interface with the surrounding normal tissue. (f) At higher magnification shows stromal cellularity with severe stromal atypia and mitoses.
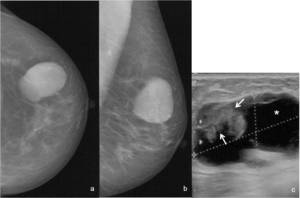
Fig. 25: 72-year-old woman that refers a rapidly increasing nodule on the left breast. Left (a) CC and (b) MLO mammographic views show a large and relatively circumscribed ovalar lesion in the upper outer quadrant, with high-density. (c) On US scan it consists on a complex cyst (*) with a parietal ill-defined mass (arrows). US-guided core needle biopsy of the intracystic mass revealed a malignant phyllodes tumor .
Small phyllodes tumors may be difficult to differentiate from benign fibroadenomas at mammography and US.
Patient age is useful as new or enlarging fibroadenomas are very uncommon in postmenopausal women,
though they are occasionally seen in women using menopausal hormone therapy.
Therefore,
any new or enlarging oval,
relatively circumscribed mass that is hypoechoic at US and resembles a fibroadenoma should be viewed with suspicion in a postmenopausal woman not using menopausal hormone therapy.
MR imaging findings of cystic changes with irregular walls and tumor signal intensity lower than that of breast tissue on T2-weighted images are suggestive of malignancy.
Histologically,
phyllodes tumors are composed of epithelial and stromal elements and typically exhibit an intracanalicular growth pattern,
with leaflike projections.
The tumors generally contain a large number of fusiform fibroblasts,
but the stromal component varies significantly from one tumor to another.
Malignant tumors show marked cellular pleomorphism,
infiltrative margins,
and frankly sarcomatous stroma (see Fig.
24).
Local recurrence is seen in both benign and malignant phyllodes tumors.
The local recurrence rate strongly depends on the width of the excision margin.
Consequently,
a wide surgical excision is recommended.
Adenoid Cystic Carcinoma
Adenoid cystic carcinoma (ACC) is a variant of adenocarcinoma typically seen in the salivary glands,
but has also been reported in other organs,
namely in the breast.
ACC accounts for 0,1%-0,4% of all breast cancers [3].
It predominantly affects women in the sixth decade of life.
The tumor is morphologically heterogeneous and shows three different architectural patterns: tubular,
cribriform,
and solid.
Like most of the slow-growing breast cancers,
it is typically round and has ill-defined margins.
It has generally a favorable prognosis,
as it rarely metastasizes to the axillary lymph nodes.
At mammography ACC of the breast can manifest as a benign-appearing smooth or lobulated mass,
as an irregular mass or as an asymmetric developing density (see Fig.
26).
Sonographically,
ACC appears as a hypoechoic or heterogeneous mass with irregular contours (see Fig.
26).
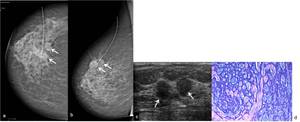
Fig. 26: 70-year-old woman. Right (a) CC and (b) MLO mammographic views shows in the superior outer quadrant, two adjacent rounded relatively circumscribed masses, with equal- to low-density. (c) On US scan these two adjacent lesions corresponded to relatively circumscribed homogeneously hypoechoic masses (arrows), with irregular contours. Core-needle biopsy revealed an adenoid cystic carcinoma. (d) Hematoxylin-eosin, with amplification of 200x shows typical fenestrated nests composed of two cell types (dominant basaloid and few eosinophilic) are shown.
MR imaging appearance of breast ACC may correspond to a well-circumscribed,
round lesion with rapid enhancement (but no washout) extending from the periphery centrally over time.
The solid variant of ACC often show high signal intensity on T2-weighted images (see Fig.
).
At pathologic analysis,
it is characterized by the presence of mucin within pseudocysts found in the tumor (see Fig.
26).
Angiosarcomas
Angiosarcomas are rare malignant tumors that arise from endothelial cells lining the vascular channels.
Angiosarcoma is rare as a primary breast tumor,
accounting for less than 1% of breast tumors [2].
Secondary angiosarcomas occur most frequently,
generally after breast conservation therapy with radiation therapy; the average latency period is at least 5–6 years.
Both may manifest as skin discoloration plaques or nodules as a palpable mass,
or as a combination of these signs.
Discoloration may be mistaken for bruising,
thereby delaying diagnosis.
Metastases are usually hematogenous and the disease most commonly spreads to the lungs.
The prognosis is poor in these situations.
Mammographic findings are nonspecific for angiosarcoma.
An ill-defined,
noncalcified mass or a focal area of asymmetry are common findings (see Fig.
27).
Sonographically,
angiosarcomas typically present as predominantly hypoechoic solitary or multiple masses.
Disruption of the normal breast architecture is generally associated (see Fig.
27).
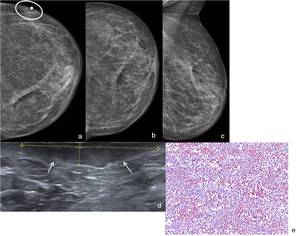
Fig. 27: 80-year-old woman who underwent left lumpectomy and radio therapy for IDC 8 years earlier. Patient had a new onset of redness and swelling in the left breast. A bulge in the contour of the lower outer quadrant of the left breast (ellipse) associated with mild skin thickening are observed in additional view with BB marker (a) findings not identified in conventional views CC (craniocaudal) (b) and MLO (mediolateral oblique) (c). (d) On US scan this lesion corresponded to a subcutaneous relatively circumscribed hypoechoic area (arrows), associated to cutaneous edema. Biopsy of the architectural distortion revealed an angiosarcoma. (e) Hematoxylin-eosin, with amplification of 200x shows that the tumor is composed of interanastomosing channels containing red blood cells. The endothelial nuclei are prominent and hyperchromatic, but mitotic figures are absent.
The MRI findings of angiosarcoma depict a heterogeneous mass with low signal intensity on T1-weighted images,
but signal intensity is high in images that are heavily T2-weighted (see Fig.
28).
Irregular areas of high T1 signal may be seen in the higher-grade lesions; these represent hemorrhage or venous lakes.
Enhancement of the mass depends on the tumor grade.
Low-grade angiosarcomas show progressive enhancement.
High-grade angiosarcomas show rapid enhancement and washout,
and large draining vessels can also be visualized (see Fig.
28).
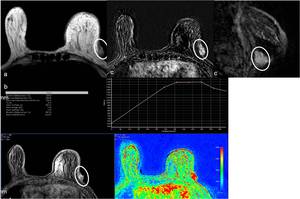
Fig. 28: 80-year-old woman who underwent left lumpectomy and radio therapy for IDC 8 years earlier. Patient had a new onset of redness and swelling in the left breast with the diagnosis of angiosarcoma. (a) At MR T2-weighted image, a superficial mass bulging (ellipse) the contour of the outer left quadrant is seen, as well as skin thickening. (b) After injection of paramagnetic contrast it shows wash-in and late wash-out (Type 3 curve). (c) In subtraction, the enhancing mass is confirmed in both axial and sagital reformatation image.





























