Pre-transplant evaluation
The evaluation of the living donor kidney is very important to identify vascular and pielo-ureteral variations,
whose presence affects the pre-transplant surgical planning.
Recipient assessment is carried out during the pre-transplant and post-transplant,
the latter in order to identify early and late complications.
Anatomical variations
Renal arteries and veins show numerous anatomical variations,
characterized by one or more vessels with different morphologies and branch level; consequently,
vascular anatomy plays a decisive role in the choice of the kidney for the transplantation,
especially if a laparoscopic nephrectomy is carried out [7].
Since there are no intra-renal arterial anastomoses,
each branch represents a terminal artery and their interruption causes ischemic damage; that's why the surgeon must reconstruct each branch of the renal vascular pedicle,
including anatomic variants [7].
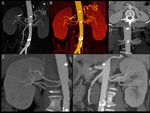
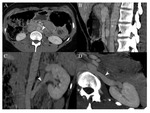
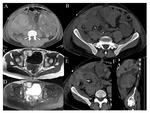
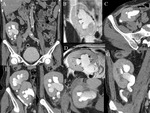
We describe the most common variations in renal arteries and renal veins (polar renal arteries,
double renal arteries,
tributaries to the left renal vein,
retroaortic left renal vein and circumaortic left renal vein) identified with TCMD and/or RM in more than 50 renal transplant donors.
About 70% of the general population has a single artery to the kidney,
while in the remaining 30% of cases anatomical variations may occur [8].
These vascular abnormalities depend on the persistence of fetal vessels,
which usually regress when renal blood vessels are formed.
In recent studies on living kidney donors investigated by MDCT and MRA,
accessory arteries were found in 40-46% of cases.
Accessory arteries (usually 2 or 3) are twice as common as accessory veins; in addition,
variants of number and position of renal arteries - without venous anomalies - are commonly encountered Fig. 3.
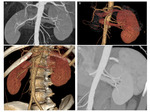
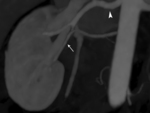
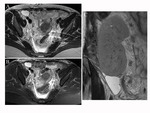
The extra-hilar renal artery - commonly indicated as polar artery - is a variation type where the artery divides before reaching the renal parenchyma Fig. 4, Fig. 5.
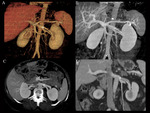
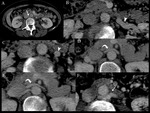
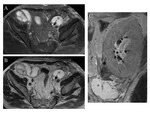
The most common vein variations are: accessory veins Fig. 6 ,
tributaries to the left renal vein,
retroaortic left renal vein Fig. 7 and circumaortic left renal vein Fig. 8 .
About 85% of the population has a single right renal vein,
often without other tributary vessels from other organs or structures; for this reason the right kidney is frequently chosen for laparoscopic nephrectomy procedures.
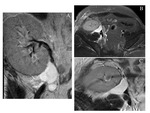
The anatomy of the left renal vein and its tributaries is often very complex; the most common variant of the left renal vein is the circum-aortic,
which occurs in 5-7% of cases Fig. 8 .
A single retroaortic left renal vein can be identified in 2-3% of individuals Fig. 7 .
In addition to vascular variants,
pre-transplant evaluation must identify any alterations of morphology and course of the urinary tract; duplication of upper urinary tract - especially the duplication of the abdominal and pelvic ureter - is one of the most common variations; such anomalies result from the division of the ureteric bud.
Ureteral duplication extension depends on the completeness of the diverticulum division [7].
Vascular complications
Several studies have shown the significant role of vascular complications in terms of morbidity and mortality: they occur in 4-8% of patients [1].
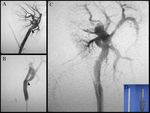
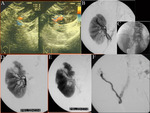
These complications are represented by the renal artery stenosis of the transplanted kidney (TRAS),
the renal vein thrombosis (TVR) and the arteriovenous fistula.
• Renal artery stenosis:
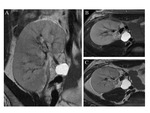
Renal artery stenosis ranges from 1% to 23%,
and represents an important cause of hypertension and/or deterioration of the function of the transplanted kidney Fig. 9.
Stenosis generally occurs in the first year after transplantation.
The definition of hemodynamic significant TRAS is not standardized and the referee value ranges from >50% to >80%.
In addition,
the introduction of cyclosporine,
a drug agent responsible for vascular damage,
seems to increase the prevalence of TRAS.
The stenosis is classified on the basis of its location:
- proximal stenosis: the stenosis is located before the anastomosis (due to atherosclerosis (donor vessel disease);
- anastomotic stenosis (due to altered vascular perfusion,
faulty suture technique or reaction to suture material)
- post-anastomotic or distal stenosis (caused by rejection or turbulent flow due to malposition of the kidney)
Doppler criteria for significant hemodynamic stenosis have been formulated by Akbar et al1:
a) a peak rate greater than 7.5 KHz (with 3 MHz probes) or a systolic peak velocity higher than 2 m/sec;
b) a velocity gradient between stenotic vessel segment and pre-stenotic tract > 2: 1;
c) and the presence of turbulence flow phenomena.
Downstream of stenosis vessels of the renal parenchyma show characteristic flow paths "tardus and parvus" [1].
• Thrombosis of the renal vein:
It is a rare complication (5% of patients or renal transplant recipients) usually encountered in the first week after transplantation (acute post-transplantation phase).
The increased prevalence of renal vein thrombosis in the lower left abdominal quadrant has been attributed to the compression of the left common iliac vein between the sacred and the common iliac artery (“silent iliac artery compression syndrome”) [1,2].
According to Sebastià et al9,
it is most frequently caused by "acute" or "hyperacute" rejection,
but it may be caused by inadequate surgical technique,
hypotension,
hypercoagulability,
cyclosporine therapy,
emboli.
Renal infarction is the consequence of vascular thrombosis,
and it appears as hypoechoic area bounded by a hyperechoic rim,
with variable enhancement [9].
• Acute renal failure:
Renal infarction is caused by the disparity between vascular supply and demand.
We distinguish an early infarction - due to damage of the renal artery of the donor or resulting from surgical complications – and a late infarction,
caused by embolic phenomena.
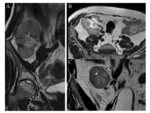
Patients show renal function impairment,
often with oligo-anuria; frequently they report pain due to the local inflammatory response Fig. 10
• Hematoma:
Frequently found during the early post-operative phase,
occurring spontaneously or following procedures (biopsy) or trauma.
Hematoma usually resolves spontaneously,
but if it increases in size it can lead to hydronephrosis and therefore require surgery.
Patients complain severe pain at the site of transplantation,
decreased haemoglobin or haematocrit,
and increases in serum creatinine Fig. 11, Fig. 12
Hematomas should be differentiated from organized urinomi [1].
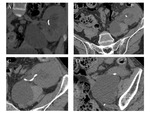
• Fistulas and arteriovenous pseudoaneurysms:
Among complications after biopsy,
fistulas and arteriovenous pseudoaneurysms have an incidence ranging from 6.3% to 10%,
depending on the techniques used for the biopsy and post-operative controls performed Fig. 13 .
According to a recent study by Furness et al10,
the incidence of arteriovenous fistula after 2127 biopsies of kidney transplant is 2.4%.
It should be noted that up to 95% of post-bioptic fistulas resolve spontaneously [10].
Pseudoaneurysm is frequently reported as complication after biopsy in 1 to 18% of cases.
It is generally asymptomatic,
but sometimes can occur with hypertension,
hematuria and deterioration of renal function.
Non-vascular complications
Perirenal transplant fluid collections can be associated with renal transplant in up to 50% of patients,
including urinomi,
hematomas,
lymphoceles,
and abscesses.
The clinical significance depends on their size,
location and the eventual growth.
Fluid collections can be distinguished by their clinical onset.
The urinomi and haematomas develop early after transplantation,
while the lymphoceles occur 4-8 weeks after surgery [1].
• Urine Extravasation and Urinomi:
These represent relatively rare complications in the post-operative period.
The urine extravasation arises from the renal pelvis,
the ureters and the ureteroneocistotomia.
Other causes of extraureterale extravasation include inadequate surgical technique and rejection [9].
Urinomi generally occur in the second or third week after transplantation,
and require surgery or percutaneous interventional approach.
The urinomi are usually located between the transplanted kidney and bladder Fig. 14; less frequently they are encountered in the scrotum and thighs.
Clinical features of these complications are: poor urine emission,
sense of weight in iliac fossa and scrotal edema [2].
The small leaks can be treated by percutaneous nefrostomia or stent placement; alternatively,
a new ureter reimplantation is recommended.
The leak’s location suggests the causes of urinoma: when located near ureteopelvic-vescical anastomosis,
it is most frequently due to ischemia or inappropriate surgical procedure; on the other hand,
diffused urinomi along the ureteral course is more suggestive of ischemic alteration [2].
• Abscesses:
Abscesses represent a rare complication that occurs usually in the first week after transplantation.
They may be caused by pyelonephritis,
and bacterial infection of a lymphocele,
hematoma or urinoma.
Clinically,
the patient may have symptoms of infection due to the state of immunosuppression: fever of unknown origin,
pain or symptoms related to the compression (in cases of large abscesses).
US or CT guided drainage is generally recommended for their treatment.
• Urinary obstruction:
This,
Fig. 15,
occurs in 2% of transplants,
and almost always in the first 6 months after surgery.
The most common site of obstruction is the site of implantation of the ureter into the bladder.
More than 90% of stenosis regards the distal end of the ureter.
In 50% of cases the obstruction is caused by kinking,
technical errors during the ureteral anastomosis,
or by post-ischemic fibrosis; rarely they are caused by pelvic fibrosis,
lithiasis or papillary necrosis.
The obstruction may be also due to compression by a large peri-renanl mass of fluid collection.
There are early or late obstructions: early forms are related to kinking,
clots,
edema or inflammation; late obstructions are caused by fibrosis,
ischemia and periureteral masses [9].
The patient does not report the typical pain (due to the denervation),
but there is an increase of creatinine levels.
The therapeutic approach - in cases of significant hydronephrosis - involves surgical reconstruction; in case of narrowing of the distal end of the ureter,
therapeutic options include balloon dilatation and /or stent placement.
• Infections:
More than 80% of transplant recipients have at least one episode of infection during the first year after transplantation.
In the first week after transplantation,
most infections are usually pneumonia,
surgical wound infections and urinary infections.
From 1 to 6 months after transplantation it is possible to find infections caused by opportunistic agents and CMV; 6 months after the transplant,
transplant recipients are commonly prone to catching the same kinds of infections that target the general population [1].
Infection is the most feared complication of transplantation,
and it should be recognized early and treated.
Pulmonary infections may be transmitted from an infected donor or blood transfusions; more often,
they are caused by the persistence of the infectious foci - existing in the recipient before transplantation.
The underlying disease,
the uremic state and the established immunosuppressive therapy after transplantation,
contribute to the decline of the immune system in the recipient.
• Lymphocele:
Represents the most frequent fluid collections responsible for hydronephrosis of the transplanted kidney,
with a prevalence ranging from 0.5% to 20%.
It may occur at any time,
from weeks to years after transplantation,
but in most cases they are encountered 1-2 months after transplantation.
The lymphoceles,
Fig. 16, Fig. 17, Fig. 18, are caused by extravasation of lymph from lymphatic vessels or iliac nodes - surgically destroyed in the transplantation.
Risk factors include: inadequate ligation of lymphatic vessels at the level of the iliac vessels and administration of heparin.
Generally they are located in the medial position between the transplanted kidney and bladder.
Clinically,
the patient may be asymptomatic or present edema of the lower extremities (due to ipsilateral compression of the femoral vein) or,
rarely,
swelling of the abdomen or scrotum [2].