ANATOMY
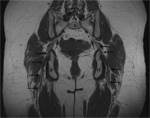
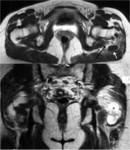
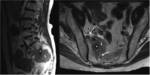
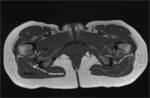
Being familiar with the anatomy of the sacral plexus,
sciatic nerve and subgluteal space is essential for the evaluation of deep gluteal syndrome.
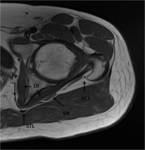
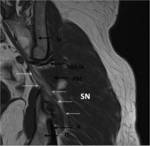
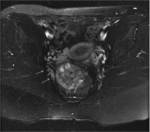
Subgluteal space (SS) is the cellular and fatty tissue situated between the middle and deep gluteal aponeurosis layers.
Its posterior limit is formed by the gluteus maximus muscle,
which is inserted medially in the posterior fourth of the iliac crest,
behind the posterior semicircular line of the external iliac fossa,
sacral crest and sacrotuberous ligament and laterally on the lateral ridge of the linea aspera.
The upper limit is the greater sciatic notch (medial) and major-minor gluteal muscles (superior and medial).
Downwardly SS continues with the posterior thigh.
Laterally is limited by the linea aspera and the lateral fusion of middle and deep gluteal aponeurosis layers reaching the posterior aspect of tensor fascia lata muscle.
The anterior limit is complex because of multiple anatomical structures that frame it,
being the most important top to down the minor and medius gluteus muscles,
superior gemellus,
obturator internus,
inferior gemellus and quadratus femoris.
Most authors include these structures within the space and set the posterior border of the femoral neck,
and greater and lesser trochanters as the anterior limit.
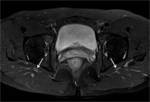
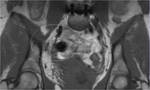
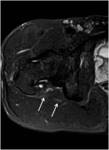
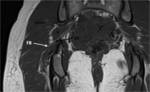
In its lower and medial margin SS is framed by the posterior surface of the ischium and the insertion of the hamstrings and in its lower and lateral margin by the posterior aspect of the greater trochanter (Fig.
1,
4,
6).
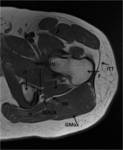
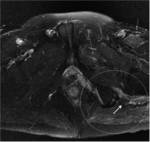
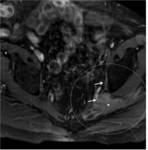
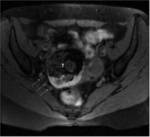
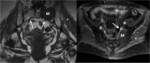
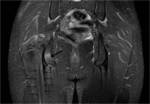
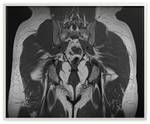
The medial limit is formed by the greater and minor sciatic foramina (Fig.
8).
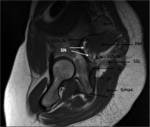
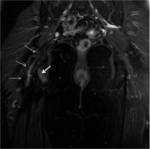
The greater sciatic fomaren is formed by the outer edge of the sacrum and the greater sciatic notch (top) and the sacrospinous ligament (bottom).
This ligament is inserted into the lateral margin of the sacrum and coccyx and extends laterally and anteriorly to the ischial spine (Fig.
7).
The limits of the lesser sciatic foramen are the confluence of tuberous and sacrospinous ligaments (internal),
the lesser sciatic notch (external),
sacrospinous lower border (top) and the upper edge of the sacrotuberous ligament (bottom).
The sacrotuberous is a strong support ligament with an insert similar to the sacrospinous medially,
that reaches distal and inferiorly to the ischial tuberosity.
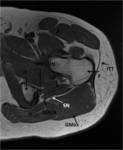
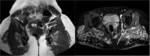
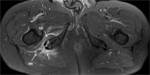
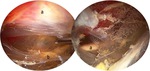
The falciform process is an expansion that extends off this ligament and fuses with the fascia of the obturator internus [1](Fig.1).
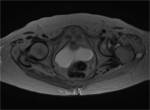
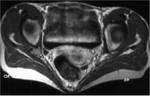
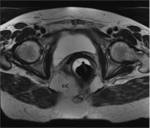
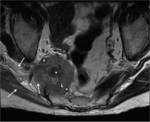
The greater sciatic foramen connects the pelvis to the gluteal region and contains the piriformis muscle as satellite structure,
which is inserted into the ventrolateral aspect of the sacrum (S2-S4) and into the medial aspect of the greater trochanter of the femur.
Thus,
divides it into two parts.
Within the suprapiriforme space runs the superior gluteal artery and nerve and within the infrapiriforme the inferior gluteal artery/nerve,
sciatic,
posterior femoral cutaneous,
obturator internus/gemellus superior and quadratus femoris/gemellus inferior nerves.
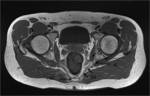
The lesser sciatic foramen contains the obturator internus muscl (Fig 8).
Within both infrapirimormis space and lesser sciatic foramen runs the pudendal artery and nerve,
before reaching the perineum.
The medial one-third of the piniform muscle is usually seen on a single axial plane just below the sacroiliac joint, the middle-third below the grater sciatic notch and the lateral-third of the muscle can be seen on the next lower contiguous images near the insertion on the greater trochanter [2].
Distinct fascial planes separate the piriform muscle from the gluteal group of muscles posteriorly and from the retropenitoneal structures anteriorly.
Identification of these fascial planes is important when searching pathology in the SS originating from parasacral and retropenitoneal spaces.
The fascial plane anterior to the piriform muscle communicates with the presacral space and thus with the opposite side.
Unlike the anterior fascial plane,
the fascial plane posterior to the piriform does not cross the midline and ends medially at the lateral border of the sacrum.
The sacrospinous ligament is also surrounded anteriorly and posteriorly by prominent fascial planes.
The space anterior to the ligament is the same space that contains the sacral plexus more superiorly.
The space posterior to the ligament containing the sciatic nerve is limited medially by the sacrum (Fig.
7).
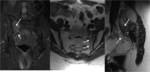
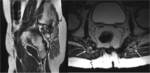
The lumbar plexus is composed of the ventral rami of L1–L4 and is anatomically located behind or less commonly within the psoas muscle,
making it difficult to identify on CT and MRI.
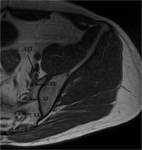
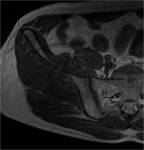
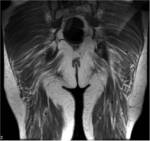
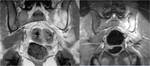
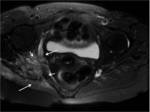
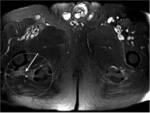
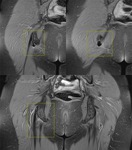
A minor branch of L4 combines with the ventral ramus of L5 to form the lumbosacral trunk (Fig.
2,
3).
It descends over the sacral ala and combines with the ventral rami of S1,
S2 and S3 (and a branch of S4) to form the sacral plexus.
These branches decreases in thickness from top to bottom.
The first branch obliquely crosses the upper edge of the piriformis,
the second running ahead of this muscle and the third branch passes through its lower edge [3] (Fig.
2,
3).
Smaller field-of-view coronal oblique images aligned with the sacrum and sagittal images centered around the sacrum allow the sacral plexus to be seen.
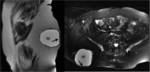
The lumbosacral trunk and the anterior divisions of the sacral plexus combine to form the medial and anterior component of the sciatic nerve (tibial nerve),
while the posterior divisions merge to form the lateral and posterior component (peroneal nerve).
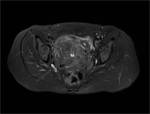
Both pass through the anterior third of the greater sciatic foramen enclosed in a common sheath and supported in the piriformis muscle arrangement perpendicular to its long axis,
just behind the sacrospinous ligament insertion into a ischial spine (Fig 1,
5,
7).
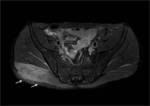
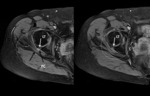
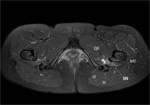
Within the superior subgluteal space sciatic nerve runs inferior and laterally ahead of the pyramidal (Fig.
5).
After curves it takes the longitudinal direction of the thigh,
behind the obturator internus-gemelli complex and cuadratus femoris and lateral,
in intimate contact,
to the conjoined tendon of the long head of biceps and semitendinosus,
lying near the posterior capsule of the hip joint.
(Fig.
9).
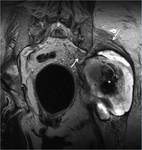
At this level the sciatic nerve is located in the middle third and least frequently in the inner third of the line between the ischium and the greater trochanter,
at a mean of 1.2 -0.2 cm from the most lateral aspect of the ischial tuberosity.
At the level of the sacrospinous ligament insertion the sciatic nerve can be identified as a robust and circular structure with a diameter of between 0.9 and 1.2 cm [1] (Fig.
6).
Throughout its course the nerve is covered by the gluteus maximus.
There is no artery after a similar course because the chief blood supply to the thigh is through the anterior femoral artery.
With hip flexion,
the sciatic nerve experiences a proximal excursion of 28.0 mm.
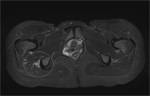
The superior gluteal nerve is formed by the posterior roots of L4,
L5,
and S1.
It exits the pelvis and bends under the greater sciatic notch,
just above the piriformis muscle,
which it crossed as an elongated X from front to back and from inside to outside [4].
More rarely crosses it.
Surrounded by a prominent venous network and superior gluteal artery,
extends along the fat-filled space between the medius and minimus gluteus.
Lavigne y Loriot identified four distribution categories,
the most common being the division into two branches at the entrance of the SS.
The thick craneal branch ascends,
then terminates in the gluteus minimus muscle.
The thin caudal branch gives rise to branches to the gluteus medius and minimus and ends in the tensor fasciae latae [4].
The superior gluteal nerve is seen on coronal and sagittal images as it exits the pelvis,
closely applied to the bony brim,
in the suprapiriformis foramen.
The superior and inferior branches of the nerve are also occasionally detected.
However,
distinguishing the nerve from the adjacent superior gluteal vascular pedicle may be difficult.
The nerve can also be seen on axial images as it travels laterally in the fat plane between the gluteus minimus and medius muscles (Fig.
3,
5,
8).
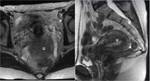
The inferior gluteal nerve originates from the dorsal L5,
S1,
and S2 rami.
It lies medial to the sciatic nerve and exits the pelvis through the greater sciatic foramen,
inferior to the piriformis muscle.
At the lower border of the piriformis muscle,
the nerve turns backward and divides into upward and downward diverging branches,
which enter the gluteus maximus (Fig.
6).
SIGNS AND SYMPTOMS: WHAT THE RADIOLOGIST NEEDS TO KNOW
Like other syndromes,
the deep subgluteal syndrome is a complex clinical entity caused by different etiologies and characterized by a set of symptoms and semiological data occurring isolated or in combination.
The most common symptoms include hip or buttock pain and tenderness in the gluteal region and retro-trochanterically,
sciaticalike pain,
often unilateral but sometimes bilateral,
that are exacerbated with rotation of the hip in flexion and knee extension.
Intolerance of sitting more than 20-30 min,
limping,
disturbed or loss of sensation in the affected extremity,
lumbago and pain at night getting better during the day are other symptoms reported by patients.
Many of them have undergone lumbar spinal surgery without improvement and require high doses of narcotics to control their pain.
Patients presenting with sciatic nerve entrapment often have a history of trauma and may present with neurological symptoms of abnormal reflexes or motor weakness.
Symptoms related to nerves other than the sciatic nerve in the subgluteal space may be observed such as weakness of the gluteus medius and minimus muscles (superior gluteal nerve),
weakness of the gluteus maximus (inferior gluteal nerve),
perineal sensory loss (pudendal nerve),
or loss of posterior cutaneous sensation (posterior femoral cutaneous nerve).
Symptoms of advanced injury of the sciatic nerve such as atrophy of the hamstring muscles below the knee,
weakness in knee flexion (hamstring),
eversion of the foot (peroneal),
inversion of the foot (tibialis anterior),
plantar flexion foot (gastrocnemius and soleus),
dorsiflexion of the foot (tibialis anterior),
weakness of the flexors and extensors planar,
loss of sensation in the lateral aspect of the leg and foot (except the medial malleolus innervated by the saphenous nerve ) or abolition of ankle reflexes are uncommon.
As the causes are multiple other many symptoms characteristic of each disorder can be added.
Physical examination tests that have been used for the clinical diagnosis of sciatic nerve entrapment include passive stretching tests and active contraction tests [1].
Radiologist must to know these tests to interpret clinical information and to differentiate it from test used for other conditions such as femoroacetabular impligement,
snapping hip,
etc.
Lasègue sign is pain with straight leg raise testing (to 90° hip flexion).
It will alert the examiner of radicular pain; however,
this test is not always positive in DGS.
Pace’s sign is pain and weakness with resisted abduction and external rotation of the hip.
Freiberg’s sign is pain with internal rotation of the extended hip.
A variant of the Freiberg test involves flexion,
adduction,
and internal rotation of the hip with the knee flexed (relaxing the sciatic nerve),
sometimes referred to as the FAIR test.
The seated piriformis stretch test is a flexion and adduction with internal rotation test performed with the patient in the seated position.
The examiner extends the knee (engaging the sciatic nerve) and passively moves the flexed hip into adduction with internal rotation while palpating 1 cm lateral to the ischium (middle fi nger) and proximally at the sciatic notch (index finger).
A positive test is the recreation of the posterior pain at the level of the piriformis or external rotators.
All these tests are reported to be positive in retro-trochanteric pain syndrome,
but they are poorly validated,
despite being frequently used in the clinical setting.
Other clinical signs include a reported discrepancy in leg length,
tenderness over the sciatic notch and isolated atrophy of the gluteus maximus muscle.
Injection tests have been advocated for supporting the diagnosis of DGS and may be helpful in examining whether the symptoms originate from the spine or from the subgluteal space.
Double injection technique of an anesthetic and corticosteroids toward the piriformis muscle in many cases relieved the pain while enabling diagnosis and excluding articular pathology or disordes within other spaces.
Guided injections of the fatty space around the sciatic nerve utilizing ultrasound CT,
or open MRI have been used to avoid injection to an incorrect site (Fig.
57,
58).
ETIOLOGY
In a prospective MRI study of the lumbar spine in asymptomatic patients 20% of theme between 20 and 59 years of age had a herniated disc and 57% of the 60- to 80-yearold group had evidence of a herniated disc or canal stenosis [6].
In 20% of cases the sciatica is of both discogenic and non-discogenic origin [7].
However,
in practice,
causes of non-discogenic entrapment are often overlooked,
partly due to the high sensitivity of lumbar spine MRI in asymptomatic patients.
It is known that extrasespinal sacral plexus and sciatic nerve entrapments may be result from a high espectrum of extrapelvic (within the subgluteal space) or intrapelvic pathology.
Due to the variation of anatomical entrapment,
the term “deep gluteal syndrome” may be a more accurate description of this nondiscogenic sciatica.
An extensive list of orthopaedic and non-orthopaedic conditions may manifest as a DGS that may be grouped into traumatic,
inflammatory,
infectous,
vascular,
tumorous,
iatrogenic,
muscle-tendinous entrapments (pirimorfis syndrome,
external rotators muscles entrapments,
gluteal pathology,
hamstring syndrome,
ischiofemoral impingement) and fibrous bands containig blood vessels.
TRAUMATIC
Traumatic nerve injury may vary from disruption of axonal conduction with preservation of anatomical continuity to complete loss of continuity of the nerve trunk.
Injury may be caused by laceration,
stretching or compression in the early period,
or later on by scar tissue,
fibrous bands or heterotropic ossification encasing the nerve.
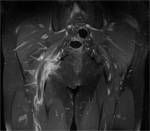
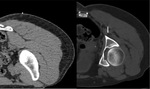
Fractures: Quite often,
DGS develops following fracture of the sacrum (Fig.
10) and the sacroiliac joint.
In addition,
the sciatic nerve may be injured in cases of acetabulum fractures,
femur fractures or femoral head dislocation.
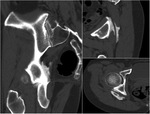
Other sources of sciatic nerve entrapment include malunion or healed of the ischium avulsions and lesser/greater trochanter fractures (Fig.11).
Haematomas: A gluteal haematoma is usually related to trauma,
hip surgery,
haemophilia or anticoagulation therapy.
Haematoma can damage the nerve either directly (increased pressure on the nerve) or indirectly (ischaemia as a result of vasa vasorum compression).
The MRI signal characteristics of the mass vary according to its contents of haemoglobin degradation products.
Acute haematoma (1–4 days) has low signal intensity in all sequences and may be surrounded by edema.
A mass in its early (2–7 days) and late (1–3 weeks) subacute phases is hyperintense on the T1 weighted images owing to its methaemoglobin content.
The signal intensity on the T2 weighted images is low during the early subacute period but high during the late subacute period.
The wall of a chronic haematoma has a hypointense signal on both sequences owing to haemosiderin accumulation.
A mass that has appeared following (usually) a trauma,
stagerelated signal changes and size reduction over time are pathognomonic for haematoma.
(Fig.12)
Direct traumatic neuropathy or contusions: During traumatic compressions nerves injured by contact with bone structures due to its deep location (acetabulum in the case of the sciatic nerve).
MR neurography revealed mild enlargement and focal T2 hyperintensity of the sciatic nerve without macroscopic pathology [8] (Fig.
13).
Direct sciatic neuropathy rarely presents in nonpenetrating trauma because of protection of the nerve by the pelvis,
the gluteal muscles,
and the tissues in the posterior thigh.
Traumatic stump neuroma: In patients undergoing proximal lower limb amputations MR neurography often reveals a low signal intensity mass within the sciatic nerve on T1-weighted spin-echo images that shows mild enhancement following intravenous administration of gadolinium.
Proximal to these neuromas,
the sciatic nerve often is enlarged with marked focal fascicular enlargement and abnormal increased T2 hyperintensity [8].
IATROGENIC
Surgery: The most common cause of sciatic neuropathy at the hip is iatrogenic and is associated with total hip replacement for primary osteoarthritis,
related to stretching or direct trauma of the nerve.
Excessive retraction,
inadvertent resection during total hip replacement or as a result of poor positioning of the patient are the most well-recognized cause of superior gluteal nerve injury.
The inferior branch of the nerve is vulnerable during the direct lateral approach of Hardinge.
The branches to the tensor fascia lata are at risk with an anterolateral or anterior approach.
The high variability in the branching pattern and course of the nerve makes it even more susceptible to injury at surgery.
Inferior gluteal entrapment neuropathy is rarely reported but is recognized as a complication of the posterior approach to hip arthroplasty.
Subclinical electromyographic abnormalities of both the superior and inferior gluteal nerves have been described by Abitbol et all.
in up to 77% of patients after total hip replacement,
regardless of the surgical approach.
Injury to the superior gluteal nerve is also a well-recognized complication of percutaneous placement of iliosacral screws. The pattern of muscular atrophy,
a characteristic sign of entrapment,
reveal which nerve is affected and the level of injury (Fig.14).
Inyecciones intramusculares: nerve injury may be caused by accumulation of injected drugs within the perineural space,
direct needle trauma,
secondary to a compression by scar tissue or direct nerve fibre damage caused by neurotoxic chemicals in the injected drug.
(Fig.15)
Radiotherapy: Post-radiation neuropathy is a rare condition most frequently observed following higher doses of radiation (5000 rads).
Sciatic nerve may be damaged following radiotherapy either directly (by the harmful effects of radiation itself) or indirectly (by diffuse fibrosis of tissues surrounding the nerve).
Imaging reveals diffuse nerve thickening with no observable mass.
Rarely,
however,
focal mass-like lesions similar to those of tumour recurrence may be observed,
being hypointense on the T1 weighted and hyperintense on the T2 weighted MR images.
Contrast enhancement following intravenous administration of gadolinium may also be observed (Fig.16).
Foreign Bodies: Foreign bodies of any nature can injure or compress the sciatic nerve,
usually after high-energy trauma,
surgery or due to iatrogenic causes,
such as silicone injections not contained in a prosthesis.
(Fig.17).
INFLAMMATORY/INFECTOUS
Sacroiliitis: Sacroiliitis is a frequently encountered finding among seronegative spondyloarthropathies.
Sciatica may be the result of referred pain or the inflammatory changes in the immediate neighborhood of the sacroiliac joint directly affecting the nerve (Fig.18).
Osteoartritis: DGS can be seen as a result of mechanical compression of the sciatic nerve related to degenerative changes (osteophyte formation) in the hip joint especially in elderly patients (Fig.19).
Abscesos: Gluteal and pelvic region abscesses are seldom encountered.
They are usually related to gastrointestinal and urinary tract infections and may affect the sacral plexus by spreading via normal anatomical routes.
A gluteal and presacral abscess also related to sacral osteomyelitis may directly affect the lumbosacral plexus and sciatic nerve.
Myositis: Myositis is a rare muscle infection,
with the most commonly implicated bacteria being Staphylococcus and Streptococcus.
When it affects gluteal or pelvic-trochanteric muscles may cause DGS.
Piriformis myositis has been rarely reported in the literature.
Recognized predisposing factors for the condition are mainly previous viral or parasitic infections,
rheumatic disease and HIV infection.
Brucella spp.
is also a known cause of myositis [9].
It is also important to address the fact that muscle infections will finally extend to the retrofascial compartment and can become a source of confusion for the clinician,
as the problem would seem worse at a more distant site than at the site of origin.
Myositis is known to have three distinct phases,
as described by Chiedozi.
During the first phase inflammation is minimal.
The muscle becomes hardened,
with mild leukocytosis with no evidence of pus.
After two to three weeks of the initial symptoms the inflammation increases with evidence of purulence.
The third phase is characterized by signs of systemic toxicity (Fig.20).
Bursitis: Distention of periarticular bursae of the hip is a recognized and frequent cause of DGS.
Most commonly,
bursitis have an inflammatory origin (secondary to degenerative tendinopathy of gluteal tendons inserting on trochanter) but may be pyogenic,
traumatic ...
(Fig.21).
Sciatic nerve sarcoidosis: Sciatic nerve MR neurography may reveals a mildly enhancing mass centered within the sciatic nerve.
The mass is hypointense on T1-weighted images and T2-weighted imaging reveals fascicular enlargement proximal and distal to the mass.
On the basis of homogeneous loss of normal fascicular architecture at the mass level and enhancement characteristics a preoperative diagnosis of neurofibroma often is made.
Surgical internal neurolysis and biopsy is neccesary to demostrates chronic inflammatory changes and multinucleated giant cells with noncaseating granulomas,
characteristic of sarcoidosis [8].
VASCULAR
Persistent sciatic artery: The persistent sciatic artery (PSA) is a rare congenital anomaly with an incidence ranging from 0.025% to 0.04% of the population.
This artery is prone to atherosclerotic change and is associated with aneurysmal change in 46.1% of the cases.
The most frequent symptoms associated with PSA aneurysm are a painful pulsatile buttock mass,
sciatic neuropathy caused by sciatic nerve compression (According to Pris et al.
sciatica is found in 36%),
and lower limb ischemia caused by thrombosis or distal embolization.
The aneurysms complications are embolism,
thrombosis,
rupture,
fissuring,
and sciatic nerve compression,
because the course of the persistent sciatic artery through the thigh is varied but always close to the sciatic nerve.
PSA is classified as complete or incomplete.
In the incomplete type,
PSA is hypoplastic,
and the superficial femoral artery is the major blood supply to the lower extremity.
In the complete type,
the PSA continues to the popliteal artery with a minimal change in size,
while the superficial femoral artery is small and provides only collaterals to the lower limb.
Persistent sciatic artery might be rarely bilateral with an incidence between 12% and 32%,
according to the literature.
Radiological investigations are important to identify,
classify and aid in performing endovascular intervention.
Treatment of persistent sciatic artery aneurysm is surgical (graft interposition and femoropopliteal bypass,
iliopopliteal transobturator bypass…) or by endovascular intervention [9].
Aneurism or pseudoaneurism of iliac artery: The lumbosacral trunk is anterior to the sacrum and posterior to the iliac vessels,
and any aneurysmal or pseudoaneurysmal expansion of the iliac artery (especially the internal iliac artery) and its branches may affect the sciatic nerve.
The basic mechanism of aneurysm-related sciatica is compression to the nerve.
Although nerves are fairly resistant to ischaemia and the lumbosacral plexus is rich in vascular supply,
ischaemia may play an additional role (secondary to vasa vasorum compression).
Arteriovenous malformation or arteriovenous fistula: Although quite rarey,
direct pressure on the sciatic nerve by an arteriovenous malformation or arteriovenous fistula may also cause sciatica.
Thrombosis of the superior gluteal artery: These is a cause of buttock claudication,
occurring consistently after 150 meters walking and disappearing after a short rest.
As magnetic resonance angiography (MRA) often not shows any explanation,
selective angiography of left internal iliac artery is requests,
which clearly identifies a tight stenosis of the onset of the artery.
Per-cutaneous angioplasty together with stenting of this artery,
induced a prompt and complete relief of pain [10].
Varicosity-caused deep gluteal syndrome : gluteal vascular compressive neuropathy coursing with intractable DGS which was not elicited by posture change or cough should add value to the presence of varicosities.
Sitting on the affected side causes more pain than standing or walking [11].
Magnetic resonance imaging reveals compression of the gluteal portion of the sciatic nerve by varicotic gluteal veins.
Ligation and resection of varicotic vein results in relief of the patient's pain.
Cases with varicosity-caused sciatica are limited in the literature review (Fig.22).
Traumatic venous varix causing sciatic neuropathy: Development of traumatic venous varix of the inferior gluteal vein may causes compression sciatic neuropathy,
usually after a fall onto the buttock [12].
Gradient echo and two-dimensional time-of-flight magnetic resonance imaging sequences confirm vascular lesion originating from the gluteals vein and compressing the sciatic nerve.
Surgical resection is successful in obliterating the lesion and relieving the symptoms (Fig.23).
GYNECOLOGICAL
Few cases of sciatica related to gynecological and obstetrical diseases and complications have been reported.
However,
pathological conditions in the pelvis and gynaecological diseases must be borne in mind,
especially when the right side is affected.
Furthermore,
it has been suggested that the sigmoid colon plays a role in preventing pressure on or stimulation of the sacral plexus on the left side.
It is recommendes that women in their late 30s or premenopause period presenting with DGS should have a thorough gynaecological examination before back surgery or subgluteal space arthroscopy is attempted,
even where radiological imaging suggests a herniated intervertebral disc.
Paying attention to gynecological pathology is essential to avoid false negatives cases which lead the patient to unnecessary and ineffective treatments.
Adenomyosis: DGS with a predominant L5 root irritation component caused by adenomyosis of the uterus has been reported [13].
This condition is a benign invasion of the myometrium by the endometrium usually affecting women over the age of 30,
and found in approximately 20% of removed uteri.
It is otherwise known to manifest itself classically by progressive menstrual bleeding,
increasingly painful dysmenorrhea,
and a gradually enlarging,
tender uterus
Retroversion of the uterus is one of the most frequent and undervalued gynecological causes of DGS (Fig.24).
Hysteromiomas,
tuboovarian abscess,
ovarian cysts and teratomas (Fig.26).
has also been reported to be a cause of lumbosacral plexopathy.
Ectopic endometriosis: Endometriosis was reported several times as a source of cyclic sciatica.
Endometriosis may compress the sciatic nerve within the pelvis,
sciatic notch or subgluteal space being most frequent at the level of the sciatic notch and on the right.
Sciatic pain typically starts a few days before menstruation,
intensifies progressively and shows some relief a week after menstruation is over.
A focal mass is classically seen as high signal intensity on both T2 and T1 weighted images,
suggesting acute haemorrhage; however,
signal intensities can vary depending on the nature of the haemoglobin breakdown products.
Endometriosis is the most frequently encountered benign mass causing sciatic nerve entrapment (Fig.25).
Pregnancy: Pregnancy-related DGS may be caused by the direct compression of the nerve roots or secondary to the possible ischaemia of the neural elements caused by compression of the aorta or vena cava by the gravid uterus [14].
L5 root irritation,
with unilateral foot drop in some cases,
may occur through injury of the lumbosacral trunk by the fetal head or forceps during delivery.
Up to 50% of women report back pain at some stage during pregnancy and in one-third the severity of the pain is such that it interferes with daily life.
Although in many cases backache resolves shortly after delivery,
in some it continues for months; in others it begins postpartum.
Furthermore,
during pregnancy,
“piriformis syndrome” is more likely to occur.
The pregnant woman begins to secrete the hormone “relaxin” which increases pelvic stretch and can potentially open up the space in the sacro-iliac joints. Her spine also increases in lordosis (the curvature going anterior),
which tilts the pelvis forward moving it into increased flexion. These things combined cause the gluteal muscle group of the hip/pelvis to become elongated and strained,
especially the deep,
small muscles of the hip.
Massage therapy can be used as a preventative measure and to relieve symptoms when aggravated.
PSEUDOTUMORALES Y TUMORS
Lesiones primarias del nervio ciático:
-
Hypertrophic neuropathy: In this pathologyc condition MR neurography revealed an infiltrating and enhancing sciatic nerve mass centered within the nerve with enlarged peripherally displaced fascicles displayed abnormally elevated T2 hyperintensity.
A central mass (white arrow) that is isointense to muscle and displaces several mildly swollen fascicles to the nerve periphery is also seen.
More distally abnormal T2 hyperintensity (but preserved fascicular morphology) reflecting a compressive neuropraxic injury is frequently found.
Biopsy revealed fibrosis and ‘‘onion bulb’’ lesions,
characteristic of hypertrophic neuropathy,
and a surgical internal neurolysis of the mid-thigh sciatic nerve must be performed.
By their characteristic,
preoperative diagnosis of schwannoma is common [8].
-
Schwannomas and neurofibromas: Schwannoma is the most common primary tumour of the sciatic nerve.
It originates from the Schwann cells forming the sheath of the nerve.
Pelvic region schwannomas are quite rare.
The imaging features of a schwannoma overlap those of a solitary neurofibroma (originating from nerve fibres),
and often they are indistinguishable.
-
Lipomatosis and intraneural lipomas: Lipomatosis,
also known as fibrolipomatous hamartoma or fibrofatty proliferation,
is a rare benign tumorlike lesion that most often affects the median nerve or its branches [16].
Rarely,
involvement of the nerves of the lower extremities have been reported.
Over 50% of patients are symptomatic because of mass sensation or focal compressive neuropathy.
Several serpentine intralesional softtissue attenuation structures that has a spaghettilike appearance in longitudinal section and a coaxial-cable-like appearance in cross-section are considered pathognomonic of lipomatosis of nerve.
Differential diagnostic considerations of pelvic lipomatosis of nerve include retroperitoneal intraneural lipoma and retroperitoneal well-differentiated liposarcoma.
Intraneural lipomas are focal encapsulated tumors located within the nerve sheath that do not involve individual nerve bundles and appear as focal round or oval lesions of homogeneous or nearly homogeneous fat attenuation with crowding of adjacent normal nerve bundles,
whereas lipomatosis of nerve tends to involve nerves diffusely.
Bone and soft tissue benign and malignant tumors.
Pseudotumoral lesions:
-
Many benign tumours and pseudotumoral lesions occur along the course of the sacral plexus and sciatic nerve and involve or impinge on its various components.
Lipoma,
osteochondroma and ganglion cyst are the most common benign tumours causing DGS.
However,
a wide range of other benign tumours and pseudotumoral lesions has been reported in the literature.
Although the appearance of many benign processes is non-specific,
some offer imaging characteristics that suggest the exact diagnosis (Fig.27).
-
Calcinosis tumoral (massive periarticular calcinosis): Tumoral calcinosis is a rare benign disease characterized by the presence of calcified,
soft and periarticular masses,
variable in size and slowly progressive growth.
It is secondary to diffuse calcium phosphate deposition into soft tissue and may be seen in the setting of uremia,
hyperparathyroidism,
or vitamin D intoxication.
Unfortunately,
the term of tumoral calcinosis has been liberally and imprecisely used to describe any massive collection of periarticular calcification,
although it actually refers to a hereditary condition associated with massive periarticular calcification and it is more appropriate to use the term "massive periarticular calcinosis" [18].
If the radiologist is unfamiliar with tumoral calcinosis or disease processes that mimic this condition diagnosis could be impeded,
treatment could be delayed,
and undue alarm could be raised,
possibly leading to unwarranted surgical procedures.
The soft-tissue lesions of tumoral calcinosis are typically lobulated,
well-demarcated calcifications that are most often distributed along the extensor surfaces of large joints.
There are many conditions with similar appearances,
including the calcinosis of chronic renal failure,
calcinosis universalis,
calcinosis circumscripta,
calcific tendonitis,
synovial osteochondromatosis,
synovial sarcoma,
osteosarcoma,
myositis ossificans,
tophaceous gout,
and calcific myonecrosis.
Tumoral calcinosis involving nerve structures is challenging to treat,
primarily because of its extensive size and propensity to infiltrate.
Although surgical intervention can often provide symptomatic improvement,
this lesion tends to recur in the presence of elevated calcium phosphate levels,
and its management therefore requires a combined multidisciplinary surgical and medical approach [19].
-
Soft tissue sarcomas: Similar to intramuscular metastases,
soft-tissue sarcomas are seen as isohypointense T1 weighted and hyperintense T2 weighted MRI lesions.
However,
necrosis,
peritumoral,
oedema and lobulation are less frequently encountered in soft-tissue sarcomas than in metastatic lesions [20].
Histopathological examination is mandatory for a definitive diagnosis (Fig.28).
-
Intramuscular metastasis: metastatic tumour involvement of skeletal muscle is rare.
The frequency of this metastasis is reported to be 0.8–16% in autopsy studies.
Lung carcinomas are usually the primary source of metastasis,
and involvement of muscles neighbouring the sciatic nerve may also be observed.
Intramuscular metastasis is seen as a low-attenuation mass in contrast CT images,
often demonstrating peripheral contrast attenuation.
En RM son isohypointense on the T1 weighted and hyperintense on the T2 weighted MRI images when compared with surrounding muscle tissues.
The mass causes expansion of the involved muscle,
and accompanying peritumoral oedema may be noticeable.
In addition,
haemorrhage,
necrosis and calcification within the mass may be observable.
The variety of metastases that can be found is as wide as the variety of malignant tumors.(Fig.35).
Intra-abdominal and intrapelvic benign and malignant tumours:
The lumbosacral plexus may be affected as a result of compression or invasion by intra-abdominal or intrapelvic masses.
Colorectal carcinoma is the most frequently encountered malignant causes followed by de uterine,
prostatic and ovarian tumours.
In addition masses which extend through the sciatic foramenes may cause sciatic nerve entrapment in the SS (Fig.
29,
30,
31,
33,
34,
38,
39).
Lymphomas deserve special attention.
There are three ways in which it may affect the sciatic nerve.
The most frequent cause of lymphoma-related DGS is compression of the nerve by the enlarged lymph nodes.
Secondly,
extranodal involvement of soft tissues such as muscle (e.g.
piriformis and gluteus muscles) may affect the sciatic nerve.
In such cases,
asymmetrical muscle expansion,
heterogeneous or low focal density on the CT images,
or focal or diffuse low T1 weighted signal intensity or high T2 weighted signal intensity on the MRI images are radiologically observed.
A uniform or ring-form contrast attenuation may be seen or the lesion may not attenuate contrast at all.
And lastly,
although very rare,
direct lymphoma invasión of the sciatic nerve has also been reported.
(Fig.36,
37).
MUSCULAR-TENDINOUS ENTRAPMENTS
A common cause recent knowledge and of sciatic nerve entrapment is the formation of fibrovascular bands secondary to acute or chronic inflammatory pathology in the subgluteo space.
Multiple orthopedic non-orthopaedic entities can cause DGS by this mechanism.
Among them we highlight:
Obturator internus and gemellus syndrome: The obturator internus muscle is located inferior to the piriformis and originates at the medial surface of the pubis,
covers the obturator foramen and passes through the lesser sciatic notch to insert onto the greater trochanter laterally.
Pathology of the internal obturator internus muscle may be obscured by the complex anatomy in the region.
Because of its proximity and similarity in both structure and function,
most treatments for the piriformis syndrome also affect the internal obturator.
Although it may not be common,
compression of the sacral plexus caused by stretched or alterated dynamic motion of the obturator internus muscle should be included as a possible diagnosis for sciatic pain.
The tendon of the piriformis muscle was found to have fused with the internal obturator tendon in 48 of 112 cases in an anatomical study,
which can indicate a strong interaction between the piriformis and internal obturator muscles and the sciatic nerve [21].
In fact,
it also runs parallel to the piriformis muscle in its attachment to the trochanter major.
A new anatomical variation,
where a high division of the SN occurs in relation to the superior gemellus muscle is also may be seen [22].
In this case,
the common fibular nerve passed beneath piriformis,
and the tibial nerve was observed under the inferior margin of the superior gemellus muscle and over obturator internus,
instead of its normal position on the superior margin of the gemellus.
It has been described cases of sciatic nerve entrapment with markedly hyperintense and flattened nerve between the hypertrophied obturator internus and gluteus maximus in a bodybuilder.
The obturator internus/gemelli complex is commonly overlooked in association with sciatica-like pain.
As the sciatic nerve passes under the belly of the piriformis and over the superior gemelli/obturator internus,
a scissor effect between the two muscles can be the source of entrapment.
Obturator internus penetrating the sciatic nerve.
In a study of 6 patients who underwent surgery for suspected piriformis syndrome,
all were observed intraoperatively to have increased obturator internus muscle tension,
hyperemia,
and hypertrophy.
Furthermore,
the obturator internus muscle was observed impinging on the sciatic nerve during an intraoperative Lasègue maneuver.
Because of this proximity,
similar pathway,
and similar function,
most treatments for patients with piriformis syndrome would affect the internal obturator muscle as well.
Cox et al.
argued that the gemelli-obturator internus muscles and its associated bursa should be regarded as possible sources of retro-trochanterically located DGS.
(Fig.40,
54).
Gluteal compartiment syndrome: Gluteal compartment syndrome (GCS) is rare and usually follows trauma,
substance abuse or general anaesthesia with a prolonged period of immobility and/or reduced consciousness [23].
Other recognized associations include severe exertion and anticoagulation treatment.
Sickle cell trait,
intramuscular injection,
spontaneous gluteal artery rupture,
and prolonged surgical procedures associated with the lateral decubitus are other rarer causes.
The diagnosis can be made on the basis of the clinical signs/symptoms and can be confirmed by measurement of the elevated compartment tissue pressure by the use of a wick catheter.
Pressures between 30 and 50 mmHg may produce muscle ischaemia and necrosis will occur if pressure is maintained at these levels for 4-8 hours.
The time interval between the precipitating event and the onset of symptoms is usually 10-15 hours but it may be as long as 6 days.
Both the duration and magnitude of the pressure rise are important factors in the production of soft tissue injury.
Hargens et al.
showed that the time required to produce peripheral nerve conduction block is inversely proportional to intracompartmental pressure.
As first suggested by Rommel et al.
and Small et al.
the differential diagnosis of painful swelling in the lower extremities presenting as a DVT should include compartment syndrome.
The sciatic nerve does not lie within the fascial envelope except in cases where it perforates the piriformis but the nerve is vulnerable to compression by swelling in the adjacent muscles.
Contracture of the gluteus maximus muscle: Gluteal contracture is well documented in the orthopedics literatura.
Most of the patients reported are school-age children (6–18 years old) and the lesions are secondary to multiple intramuscular injections in the buttocks.
Intramuscular fibrosis with subsequent retraction of the scar tissue accounts for the contracture of the hip.
A constitutional fibrogenic diathesis has also been reported as an important factor.
Although the diagnosis is clinical,
MRI may be requested for evaluation of the the extent of fibrosis and degree of muscle atrophy.
Intramuscular fibrotic cord extending to the thickened distal tendon with atrophy of the gluteus maximus muscle and posteromedial displacement of the iliotibial tract are the major MRI features of fibrous muscular contracture.
These features were present in all of our patients.
The differential diagnosis includes disuse,
denervation,
chronic inflammatory myopathy,
chronic compartment syndrome,
fibromatosis or posttraumatic hemosiderin deposition.
Conservative measures such as passive stretching are usually not helpful.
In cases of severe contracture or for cosmetic reasons,
however,
surgery is the only way to correct the deformity [24].
Quadratus femoris and isquiofemoral space pathology:
-
Strains and tears of quadratus femoris muscle (QFM): Tear of the QFM is infrequently reported in the medical literature.
Patients may present with acute or chronic hip,
groin and/or posterior gluteal pain.
QFM tears appear as an area of intramuscular fluid signal intensity with surrounding muscle edema.
Published cases of QFM tears usually involve the distal myotendinous junction,
at the posteromedial aspect of the proximal femur,
although tear at the femoral tendon insertion has also been reported.
STIR,
fat-suppressed PD or fat-suppressed T2-weighted images are required to detect muscle edema.
A grade I strain appears as muscle edema and may occur at the ischial origin of the QFM or as central or diffuse muscle edema,
indistinguishable from that seen with IFI.
Isolated QFM strains or tears are uncommon.
With severe edema,
irritation of the adjacent sciatic nerve may cause DSG (Fig.
41,
53).
-
Ischiofemoral impingement (IFI): Of 239 patients with nondiscogenic sciatica,
4.7% were diagnosed with ischial tunnel syndrome [1,
25].
Ischiofemoral impingement as an etiology of posterior hip pain has recently been reported.
It is described as a narrowing of the ischiofemoral space and typically occurs in middle-aged to elderly women.
Bilateral hip involvement has been observed in 25-40% of patients.
In patients with IFI,
ischio-femoral space (IFS) narrowing has been reported as bilateral in 25-40%,
consistent with a congenital etiology.
Several acquired conditions may narrow the IFS and QFS,
including superior and medial migration of the proximal femur due to osteoarthrosis,
osteochondroma,
prominent lesser trochanter,
enlarged ischium from prior fracture,
QFM atrophy,
displaced intertrochanteric fracture and valgus intertrochanteric osteotomy.
Radiographs are often normal but may revealed narrowing between the ischium and lesser trochanter,
with enlargement of the lesser trochanter and subcortical cysts and/or sclerosis of the opposing surfaces.
Using MR,
it has been recognized that a narrowed ischiofemoral space may cause impingement or compression of the QFM,
resulting in muscle edema and,
over time,
muscle atrophy.
The axial plane is best for this and also demonstrates the QFM origin and insertion,
adjacent muscles and tendons and the sciatic nerve.
Edema is centered on the muscle belly,
at the site of maximal impingement.
With increased severity of impingement,
it becomes more severe and diffuse and may involve the surrounding soft tissue.
Secondary irritation of the adjacent sciatic nerve with scar tissue of the perineural fatty may cause DGS.
Bursa-like formation may occur about the lesser trochanter and sciatic nerve.
Mild muscle enhancement is observed following intravenous administration of gadolinium.
No definitive treatment strategy for IFI has been established.
For patients who do not respond to conservative management,
surgical excision of the lesser trochanter and removal of reactive or bursal-type tissue has been performed without reports of complication [25](Fig.44).
Hamstring origin enthesopathies:
The hamstring tendon insertion can be thickened over the ischium and onto the sciatic nerve due to trauma,
avulsion,
tendinosis or strain.
Calcifying tendinosis is in our experience one of the most common causes of DGS.
In the acute phase edema predominates as characteristic imaging finding.
At the lateral insertion of the hamstring tendons to the ischial tuberosity,
these tight tendinous structures,
chronic inflammatory changes and adhesions are thought to cause scarring or fibrotic bands between tendons and sciatic nerve that result in decreased mobility and entrapment during hip motion.
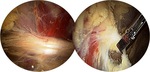
Puranen and Orava first reported on the surgical release of the sciatic nerve from adhesions in the proximal hamstring area (Fig.
42,
43,
45).
Piriformis syndrome:
Piriformis syndrome (PS) is a often undiagnosed neuromuscular condition characterized by a sciatic nerve is compromised by an abnormal piriformis muscle.
PS can be classified as a subgroup of DGS,
meaning that all PS are DGS,
but not all DGS are PS.
The true prevalence is difficult to determine accurately because it is often underdiagnosed or confused with other conditions.
Reported incidence rates for piriformis syndrome among patients with low back pain vary widely,
from 5% to 36%.
It is more common in women than men.
Filler et al did publish an investigation of 239 patients with sciatica who had either failed spine surgery (46%) or a failure to determine the exact cause of their sciatica.
After performing MR neurography and interventional MR imaging,
the final rediagnoses was piriformis syndrome in 67.8% of patients.
There are two types of piriformis syndrome,
primary and secondary.
Primary piriformis syndrome has an anatomic cause (anatomical variants).
Secondary piriformis syndrome occurs as a result of a precipitating cause including macrotrauma,
microtrauma,
ischemic,
mass,
inflammation,
infection...
Fewer than 15% of cases have primary causes.
In approximately 50% of cases of piriformis syndrome,
there is a history of trauma that is usually not dramatic and may occur several months before the initial presentation of symptoms.
Radiographic studies have limited application to the diagnosis.
Until now,
MRI and CT were to be reserved for ruling out disc and vertebral pathologic conditions and were limited to assess the presence of enlargement of the piriformis muscle.
Advances in the knowledge of the SS pathology,
the dynamics of the sciatic nerve and technical improvement of MRI make it a required test to assess pyramidal syndrome.
Potential etiologies include:
- Hypertrophy of the piriformis muscle:
Asymmetrical enlarged of piriformis muscle with anterior displacement of the sciatic nerve or high T2 weighted signal intensity has been attributed to sciatic nerve entrapment.
However,
of 14 patients with posttraumatic piriformis syndrome,
Benson and Schutzer found that only 2 had larger piriformis muscles on the symptomatic side and 7 appeared smaller than the unaffected side.
In a review of 100 patients with no history or clinical suspicion of piriformis syndrome,
Russell et al.
found that muscle size asymmetry of 2 mm was present in 81% of patients.
None of the patients with asymmetry of 4 mm or more had symptoms suggestive of piriformis syndrome.
Muscle asymmetry singly had only a specificity of 66% and sensitivity of 46% in identification of patients with muscle-based piriformis syndrome.
Asymmetry associated with sciatic nerve hyperintensity at the sciatic notch showed a specificity of 93% and a sensitivity of 64% in patients with piriformis syndrome distinct from that which no had similar symptoms.
In our opinion many asymmetries given as hypertrophy are really contralateral pirammidal atrophies.
- Anomalous course of the sciatic nerve through the muscle:
Variations exist concerning the relationship between the piriformis muscle and sciatic nerve.
The fact that these piriformis-sciatic nerve anomalies exist is important for the surgeon to recognize; however,
the anomaly itself may not be the etiology of DGS symptoms as some asymptomatic patients present with these variations and some symptomatic patients do not.
The existence of a variable relationship between those two structures appears to be 15–30% in the literatura.
The prevalence of anomalies was 16.9% in a meta-analysis of cadaveric studies and 16.2% in published surgical case series.
Variants are usually bilateral.
What is rare is to find two different types of high divisions on two sides in same patient.
Normally,
the sciatic nerve passes through the greater sciatic foramen below the piriformis muscle.
However,
it may divide into its common fibular and tibial nerve components within the pelvis.
Six categories of variations were originally described by Beaton and Anson:
- A,
undivided nerve comes out below the piriformis muscle
-
B,
a divided sciatic nerve passing through and below the piriformis muscle.
A subset additional variation has been described; in this variation,
the sciatic nerve appeared to pass below the piriformis muscle,
but a smaller accessory piriformis,
with its own separate tendon,
passed between the common fibular and tibial portions of the sciatic nerve,
creating the impression that the sciatic nerve was passing both through and below the piriformis muscle.
- C,
a divided nerve passing above and below undivided muscle
-
D,
an undivided sciatic nerve passing through the piriformis muscle
-
E,
a divided nerve passing through and above the muscle heads
- F,
undivided sciatic nerve passing above udivided muscle.
In his review of multiple cadaveric studies from the late 1800s until 2009,
Smoll presented the overall reported incidence of these six variations in over 6,000 dissected limbs.
The first relationship A was found in 83.1% of limbs,
whereas relationships B,
C,
D,
E,
and F occurred in 13.7,
1.3,
0.5,
0.08,
and 0.08%,
respectively.
Some variants predispose the nerve to stretching more than others.
Other different types have been described subsequently:
1.
Babinskiet al.
described an anatomical variation in which the tibial nerve passed inferior,
and the common fibular nerve superior to the superior gemellus muscle,
and concluded that such variations may contribute to the prevalence of piriformis syndrome.
2.
Sciatic nerve already divided in pelvis and its two divisions comes out below piriformis.
3.
In many cases,
a thick tendon can hide under the belly of the piriformis overlying the nerve (Fig.46).
-
It can also result from direct and prolonged pressure over the muscle in sitting position (wallet sciatica).
Patients who experience pain related to piriformis syndrome will complain of aggravated pain after prolonged periods of sitting or upon rising from a seated position.
Pain may also occur with bowel movements due to the proximity of the piriformis muscle to the lateral pelvic wall.
Clinical and radiological studies are needed to correlate this sings and symptoms with the presence of fibrous bands with secondary entrpment of the sciatic nerve,
as these may be the underlying cause of negative pyramidal-MRI.
-
Piriformis syndrome is most often caused by macrotrauma to the buttocks,
leading to inflammation of soft tissue,
muscle spasm,
or both,
with resulting nerve compression.
Microtrauma may result from overuse of the piriformis muscle,
such as in long-distance walking or running or by direct compression.
Despite the quantity of literature,
few studies have demonstrated statistically significant results that support one form of treatment over another.
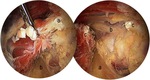
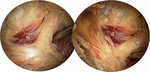
The subgluteal space is a recently defined anatomic region for endoscopic access that can treat these disorders and allows release of the piriformis tendon with exploration of relationship between piriformis and sciatic nerve,
assessing and accounting for anatomic variations that may exist.
In more than 80% of the patients a good or excellent functional outcome was achieved through treatment with imaging-guided injections or deep gluteal space surgery [1,
26-30].
Preliminary results in press awarded for the endoscopic treatment of piriformis syndrome success rates close to 100% (Fig.50).
FIBROUS ENTRAPMENTS
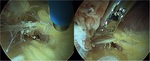
Fibrovascular bands containing vessels usually extend from the posterior border of the greater trochanter to the gluteus maximus onto the sciatic nerve and extend up to the greater sciatic notch.
A typical constricting fibrous bands and greater trochanteric bursae hypertrophy is present in many cases of sciatic nerve entrapment during endoscopy.
In our experience special attention must be given to branches of the inferior gluteal artery in the proximity to the piriformis because of its higher frequency.
Diminished or absent sciatic mobility during hip and knee movements due to these adhesions may be secondary to any of described above pathologies and cause the described symptoms of DSG (Fig.
46,
47,
51).
OTHER CAUSES
Makhsous has confirmed using dynamic MRI that the soft tissue overlying the ischial tuberosity (IT) was found to be significantly thinner in a seated posture than it is in a supine posture even when there is no sitting load.
Their findings indicates that hip flexion alone can bring forth a significant decrease in soft tissue thickness underneath IT.
Furthermore,
the decrease is not uniform among the tissue layers in area around IT.
Fat tissue experienced a significant decrease in thickness from non-weight-bearing sitting to weight-bearing sitting.
Skin and subcutaneous tissue thickness was significantly smaller than the fat and muscle thicknesses in all postures.
Duante la flexion,
rotación interna y la adduccion el nervio cambia de posición experimentando una ligera lateralización y excursión anterior.
Esto explicaría alteraciones de señal discretas a nivel del nervio ciático en pacientes con patología cercana pero no en contacto directo con el nervio.
During flexion,
internal rotation and adduction nerve changes its position and experiencing a slight lateralization and marked anterior excursion.
This would explain sciatic nerve signal changes in patients with pathology close to but not in direct contact with the nerve.
Changes in rotational osseous alignment may affect normal sciatic nerve excursión as shown in 2 of the 5 low-improvement cases with diminished femoral version.
The excursion of the sciatic nerve with straight-leg hip flexion performed by Coppieters et al.
did not indicate the osseous parameters of the hip,
which raises the question of sciatic nerve excursion with normal versus abnormal rotational alignment.
IMAGING FINDINGS
Stewart argued that the piriformis syndrome has been overdiagnosed and proposed that a definitive diagnosis would require surgical exploration to identify compression of the sciatic nerve by the piriformis muscle or by an associated fibrous band.
However,
MR neurography in a recent study demonstrated focal signal abnormalities within the sciatic nerve in the buttock in all patients with unexplained sciatica.
Similar findings were recently reported in a larger series by Filler et al.
These outcomes associated with advances in MRI hardware and development of new imaging techniques allowing detection of anatomical variations and fibrovascular bands that restrinc mobility to nerve,
suggest that until now the depp gluteal syndrome has probably been underdiagnosed,
rather than overdiagnosed,
and MR neurography is the diagnostic procedure of choice.
Explicit indications for ordering an MR neurographic study and selection criteria for patients who might benefit from MR neurography after negative results of routine MR imaging of the lumbar segments of the spinal cord is beyond the scope of discussion.
Good management of specific clinical tests by specialists is essential.
Although the primary goals of MR neurography are to localize the site of a nerve injury/fixation and to to depict the lesions causing nerve entrapment or impingement,
MRN has also been successfully used to assess the extent of the abnormality,
to exclude the diagnosis of neuropathy by showing normal nerves and regional muscles; to detect incidental patologies that mimic neuropathy symptoms; and to provide imaging guidance for perineural medication injections.
Thus the performance of MRI has important implications in treatment decisions.
Thoracic wrap-around phase-array coil placed over the pelvis or thighs provides optimal coverage of the lumbosacral plexus and sciatic nerve.
Pelvic phase-array coils are inadequate for examining the sciatic nerve in most patients.
Imaging coverage from L3 through the ischial tuberosity centered in the midline is most suitable.
The normal sciatic nerve is a welldefined and easily identified oval structure with discrete fascicles (40–60 fascicles) isointense to adjacent muscle tissue on T1-weighted images.
Normal nerve fascicles should be of uniform size and shape (Fig.
1-3,
5,
8,
9).
Perifascicular and perineural high signal intensity from fat makes nerves conspicuous on T1-weighted images.
None structure must be present normally in the perineural fat.
On T2-weighted or fast spin-echo inversion recovery images,
the normal sciatic nerve is isointense or mildly hyperintense to muscle and hypointense to regional vessels,
with clearly defined fascicles separated by interposed lower signal connective tissue.
Normal endoneurial fluid,
when protected by an intact perineurial blood-nerve barrier,
is thought to be primarily responsible for the normal signal intensity pattern of nerves at T2-weighted.
A little to moderate amount of fat is contained within the nerve.
The fluid-attenuated inversión recovery sequence is sensitive to increased water content,
as seen with edema or other pathological processes.
It also has inherent fat suppression.
This makes STIR sequences ideal for imaging of sacral plexus and sciatic nerve entrapments [31].
Neural enlargement,
loss of the normal fascicular appearance,
or blurring of the perifascicular fat are morphologic changes that suggest neural injury (Fig.
10,
12,
16-18,
20,
23,
29,
36).
Size differentials and fascicular appearance are more difficult to discern in smaller nerves such as the inferior and superior gluteal nerves.
Increased perifascicular and endoneurial signal intensity on T2-weighted images (Fig.
54,
55) reflects a nonspecific response of the nerve to injury.
This phenomenon has been explained by various hypothesized mechanisms,
such as vascular congestion,
blockade of axoplasmic flow,
leading to abnormal proximal accumulation of endoneurial fluid and distal wallerian degeneration changes [32].
However,
increased signal intensity in the nerve does not always indicate underlying disease.
The magic angle effect is a well-recognized artifact in lumbosacral plexus and sciatic nerve imaging.
Unlike in tendons,
however,
where the magic angle artifact can be overcome with longer echo times (>40 msec),
spurious high nerve signal intensity related to the magic angle can persist at higher echo times (66 msec) as well as on short inversion time inversion-recovery images.
These angle-specific signal changes must be kept in mind,
particularly when increased nerve signal intensity is the sole abnormality [33].
Mild T2 hyperintensity within the nerve can often be observed in asymptomatic subjects,
probably related to this effect or subclinical neuropathies.
Because normal nerve and plexus are slightly hyperintense on fluid-sensitive images,
high signal intensity that is focal or similar to that of adjacent vessels is more likely to be significant (Fig.
13).
In addition muscle SI changes can indicate or can further confirm the presence of neuropathy.
These denervation-related MR signal abnormalities have been shown to be a relative shift between intra- and extracompartmental fluid components and do not reflect real edema.
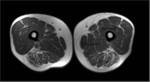
Muscles may show acute denervation changes (edemalike SI) as early as 24 hours from the onset of neuropathy; subacute changes (edemalike SI and minimal fatty replacement),
weeks to months after injury; or chronic changes (fatty replacement and atrophy),
months to years after nerve injury (Fig.
14,
56).
Sciatic nerve denervation involves hamstring muscles and the hamstring component of the adductor magnus.
Signs of denervation of the gluteal muscles are common.
In the early stage,
the T1 signal of muscle remains normal.
There are no abnormalities of the subjacent fascia or subcutaneous tissues; the normal architecture and size of the muscle are preserved.
These features help distinguish denervation from other causes of increased muscle signal intensity,
such as muscle strain,
tear,
infection,
or infarct.
The increased T1 signal indicates denervation-related muscle atrophy.
With prolonged denervation,
the bulk of the muscle will decrease and be completely replaced by fat.
This process indicates irreversible end-stage disease and muscle atrophy.
Although denervation often results in stereotypic muscle involvement,
aberrant or cross-innervation can produce atypical denervation patterns.
Through the increasing use of 3-T MR scanners,
new phasedarray surface coils,
and parallel imaging aids in the acquisition of high-resolution and high-contrast images in short imaging times it is now possible to discriminate which particular small elements of the lumbosacral-sacral plexus are involved in tumors or neuropathies.
Focal alterations in nerve contour,
course,
fascicular pattern,
size,
caliber or presence of perineural fibrovascular bands are best depicted on longitudinal high contrast isotropic 3D images reconstructed along the course of the nerve using multiplanar reconstruction (MPR) or curved-planar reconstruction.
T2-weighted SPAIR images provide more homogeneous fat suppression than frequency-selective T2 fat-suppressed images in off-center areas.
Contrast-enhanced fat-saturated T1-weighted images is only required when a infections (Fig.
20),
inflammations,
diffuse peripheral nerve lesions,
tumors and postoperative scar is suspected to be the cause of the symptom.
Enhancement in denervated muscles can be seen in this images.
Between 8 and 10 weeks after open neurolysis signal is normalized (Fig.
49-53).
However ,
there is no experience when signal increased normalizes after arthroscopic release,
since it is a recent developed technique.
Persistent nerve enlargement,
abnormal T2 hyperintensity approaching the SI of adjacent vessels,
encasing perineural fibrosis,
and fascicular abnormality suggests reentrapment although preliminary data suggest this is not frequent.
Finally,
diffusion tensor imaging and especially diffusion tensor tractography of the sciatic nerve in the near future will bring more physiologic information in pacientes with sciatic nerve entrapment.
Sacral plexus and ciatic nerve normative quantitative diffusion data such us apparent diffusion coefficient (ADC),
fractinonal anysotropy (FA) should be collected as a reference in this patients [34].
Other imaging techniques show less importance in the diagnosis of DSG.
Ultrasound enables dynamic imaging,
easy differentiation of sciatic nerve from vessels using Doppler imaging and to perform infiltration test.
On the other hand,
ultrasound remains operator-dependent and nerve imaging may be degraded by dense scarring and shadowing from surrounding calcifications.
The sacral plexus and pre-piriformis region are deep situated and are not assessable.