INTRODUCTION:
The earliest change seen in Crohn disease occurs in the submucosa and consists of lymphoid hyperplasia and lymphedema.
The main anatomopathological findings at this stage are aphthoid ulcers.
As the disease progresses,
it extends transmurally to the serosa and beyond to the mesentery and adjacent organs.
The bowel wall is thickened by a combination of oedema,
inflammatory infiltrates and fibrosis.
Aphthoid ulcers develop into linear ulcers (deeper) and fissures,
causing an ulceronodular or “cobblestone” appearance.
There is the possibility of deep ulcers to develop into fistulation between adjacent bowels as well.
Stricture formation,
bowel obstruction,
abscesses or phlegmon,
fistulas,
and sinus tracts are common complications of advanced disease.
Although not common,
toxic megacolon and neoplasms such as carcinoma and lymphoma may also occur.
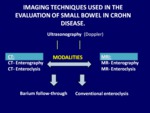
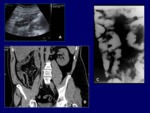
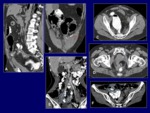
Imaging of pathologic processes occurring in the small bowel (Fig.
1,
2) has traditionally been performed with fluoroscopy (barium follow-through) because its simplicity and radiologist wide experience.
Conventional enteroclysis evaluates accurately the localization and extension of the pathology,
because its excellent capacity of loop distention,
with the inconvenient of having more technical difficulties.
They are both used for the evaluation of mucosal lesions (they are good at detecting early lesions),
having limited capacity for demonstrating the transmural and extramural extension of the disease.
Technical advances have improved imaging evaluation of the small bowel.
With the introduction of cross-sectional examinations (ultrasound,
and CT and MRI in two different modalities: enterography and enteroclysis),
it is possible to get a better assessment of bowel wall,
extramural changes and signs that indicate inflammatory activity.
Crohn disease often affects young patients who will need repeated examinations over the years.
The non-use of ionizing radiation gives a great advantage to ultrasound and MRI,
interesting to take into consideration.
The role of ultrasounds,
in most of the hospitals,
is secondary in this field,
having the advantage of being harmless and useful as the initial examination in patients in emergency service.
The imaging modality chosen (for diagnosis or follow-up),
at the end,
will depend on the patient characteristics,
the radiologist experience and the equipment availability.
SUBTYPES:
The classification of patients by subtypes depending on the course of Crohn disease in the small bowel has an important clinical utility,
helping to decide therapeutic decisions.
Crohn disease may be classified in four subtypes:
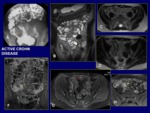
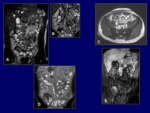
- Active inflammatory disease (Fig.
14,
15): Presence of superficial or deep ulcers (outbreak activity).
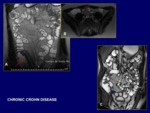
- Fibrostenosing disease (Fig.
12,
13): Chronic inflammation within the bowel wall progresses to mural fibrosis.
When fibrosis is associated with stricture formation,
bowel obstruction may occur.
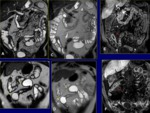
- Fistulizing/Perforating disease (Fig.
8,
10,
18): The most aggressive.
It manifests with septic complications (fistula,
phlegmon and/or abscess).
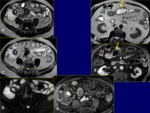
- Reparative/Regenerative disease (Fig.
13): Mucosal atrophy and regenerative pseudopolyps (different from cobblestoning).
“Halo sign” secondary to fatty infiltration of the mucosa.
Patients may exhibit characteristics of more than one disease subtype (Fig.
16,
17).
TECHNIQUES AND FINDINGS:
Barium follow-through
Barium follow-through is designed to demonstrate the small bowel from the duodenum to the ileum region including the duodenum,
jejunum,
ileum and the junctions; superiorly the stomach and inferiorly the ascending colon (Fig.
3).
The technique should ensure a non fragmented dense column of contrast agent passes smoothly through the small bowel.
Films are taken at 20-minute intervals until the contrast medium passes into the colon,
ensuring that the early films include the stomach.
A pad under the abdomen in prone position or a compressor in supine position is useful to separate out the loops of bowel (Fig.
4).
The patient may need fluoroscopic spot films of areas of interest,
especially the terminal ileum.
This technique is cheap,
easy to perform and non-invasive with some inconvenient as using ionizing radiation,
taking a long time to be performed or the necessity of additional examinations if the bowel distention is inadequate or in case of bowel overlapping.
Features that suggest Crohn disease:
Aphthous ulcers. Early Crohn disease is characterized by discrete aphthoid ulcers,
which are seen as punctate or slitlike collections of barium surrounded by radiolucent mounds of oedema.
The aphthoid ulcers are often separated by normal bowel and presented as skip lesions.
Linear ulcers (Fig.
4). As more severe Crohn disease develops,
the small ulcers become enlarged and deeper,
and they connect to one another,
forming stellate,
serpiginous,
and linear ulcers.
These ulcers are found most frequently in terminal ileum along the mesenteric border.
These are pathognomonic of Crohn disease.
On small-bowel series,
a mesenteric border ulcer appears as a long 1- to-2 mm barium collection that parallels a short,
straight mesenteric border.
A radiolucent collar usually parallels the linear barium collection at the margin of the ulcer.
The antimesenteric border of the bowel is usually uninvolved and pulled into the ulcer collar,
creating radiating folds.
Cobblestoning. As inflammation penetrates the submucosa and muscularis layers,
deep knife-like linear clefts form the basis of "cobblestoning" and fissure or fistula formation.
They appear as a barium-filled reticular network of grooves that surround round or ovoid radiolucent islands of mucosa.
Eventually,
transmural inflammation leads to decreased luminal diameter and limited distensibility (Fig.
14).
This leads to a radiographic string sign that represents long areas of circumferential inflammation and fibrosis resulting in long segments of luminal narrowing (Fig.
4,
5).
Mesenteric inflammation and fibrofatty proliferation produce a displacement of the involved loop away from the normal small bowel (Fig.
4).
Small bowel barium enteroclysis
It consists on using nasojejunal intubation until Treitz angle by fluoroscopic guidance.
Afterwards,
once we make sure the guide is fixed by a balloon,
barium should be introduced first and metilcellulose second,
with the idea that barium gets attached to the bowel wall (getting double view with two moments of film; after barium introduction (as in BFT) and after metilcellulose with the advantage of a better mucosa evaluation) (Fig.
3).
This technique allows obtaining a better loop distention and mucosal details and evaluating accurately the localization and extension of the pathology and complications (fistulas).
It has the inconvenient of using more ionizing radiation and needs nasojejunal intubation (most patients dislike this).
Features that suggest Crohn disease:
Similar to those described on barium follow-through examination (Fig.
4,
5).
Ultrasonography
Ultrasonography can be an alternative in the evaluation of the intraluminal and extraluminal manifestations of Crohn disease with the inconvenient that intraluminal gas and bowel peristalsis impede the evaluation of normal bowel in many cases.
Nevertheless,
it isn´t a problem in pathological bowel where thickened wall and reduction of gas and peristalsis facilitate its visualization.
It has the advantage of being harmless,
available and cheap with the inconvenient of being operator-dependent.
Prospective studies have shown utility for the initial diagnosis,
normally in emergency service,
assessment of disease activity and detection of complications as fistulas,
strictures and abscesses.
If the patient is candidate to surgery,
CT needs to be done to confirm features seen in ultrasound.
Features that suggest Crohn disease:
Wall thickening (Fig.
6). A normal intestinal wall thickness should measure less than 3 mm.
A bowel segment with a wall thickness > 4 mm is pathological (in Crohn disease,
it ranges from 5 to 15 mm).
Normally,
proportional thickening between layers is maintained,
although occasionally,
thickening of submucosal layer is more evident (as hypoechoic ring).
The results in ultrasonography are better when the affected segment corresponds to ileum or ascendant colon.
Besides this,
peristalsis is reduced or absent,
and the diseased segment is non-compressible and rigid.
Loss of wall layering. It is possible to find either partial or total loss of layering in advanced stage or in acute outbreak,
which reflects transmural oedema,
inflammation,
or fibrosis.
A correlation between wall thickening and loss of layering with inflammatory activity has not been found.
Changes in adjacent mesentery. Transmural inflammation and fibrosis induce changes in the echogenicity of the adjacent mesentery,
leading to fingerlike projections of mesenteric fat that creeps over the serosal surface of the bowel.
This creeping fat eventually envelops the diseased bowel segment.
On ultrasouds,
this appears as a uniform hyperechoic mass,
which is classically seen at the cephalic margin of the terminal ileum.
With long-standing disease,
this becomes more heterogeneous or even hypoechoic.
Enlarged nodes. On sonograms,
enlarged nodes can be seen as oval hypoechoic masses in the mesentery.
With confluence,
they become lobulated masses of different sizes.
Complications.
Phlegmon appears as a hypoechoic mass with irregular borders and no identifiable wall or fluid.
Abscess is observed as a fluid collection with a thickened wall containing air or echogenic debris.
It can be spontaneous,
secondary to fistulas or postsurgical.
Color-Doppler is useful to distinguish between these two entities; increased vascularization is found inside the phlegmon,
nevertheless abscess only has increased signal in the periphery.
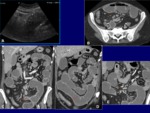
Obstruction appears as dilated hyperperistaltic fluid filled segments,
proximal to a strictured segment (seen as a thickened wall,
non-compressible and rigid bowel,
with echogenic intraluminal contents) (Fig.
9).
Perforation appears as bright echoes with distal acoustic shadows outside the boundaries of bowel loops.
A fistula,
on the other hand,
appears as a hypoechoic tract.
If gas is presented in the fistulous tract,
it contains hyperechoic foci with acoustic shadowing.
Palpation of diseased loops during sonography enables tract identification.
In addition,
sonography should be able to identify gas bubbles in abnormal locations,
such as air in the bladder or vaginal vault,
the retroperitoneum,
the subcutaneous tissue,
or the urachal remnant.
CT
The introduction of multidetector-row CT scanners with thinner collimation and faster intravenous injections of contrast material have allowed more detailed evaluation of the bowel.
We distinguish two different modalities,
enterography and enteroclysis CT.
In the first one,
the patient must ingest a big amount of oral contrast (1000-1500 ml),
in order to get well-distended bowel loops,
useful to identify the pathological segment.
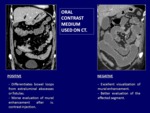
There are two kinds of oral contrast mediums (Fig.
7),
positive (high attenuation in CT.
Eg.
Diluted barium or Gastrografin®) and negative (low attenuation.
Eg.
Water) ones.
With positive contrast medium,
it is possible to differentiate bowel loops from extraluminal abscesses or fistulas,
with the inconvenient of a worse evaluation of mural enhancement after intravenous contrast injection.
Negative oral contrast medium allows an excellent visualization of mural enhancement and a better evaluation of the affected segment.
When the contrast (eg.
Methylcellulose,
as a neutral contrast medium) is administrated through a nasojejunal catheter,
the technique is called CT-enteroclysis,
obtaining the advantages and inconvenients of conventional enteroclysis (better loop distention vs.
higher ionizing radiation exposure and discomfort of intubation) and CT.
Intravenous contrast is an indispensable tool for bowel wall enhancement assessment,
improves the mesenteric vessels identification and adjacent structures differentiation.
The role of CT in the evaluation of Crohn disease is well accepted.
The ability of CT to depict bowel involvement and extraluminal pathology (eg,
abscess,
obstruction or fistula) makes it an essential imaging tool for patient care.
In most hospitals,
CT is indicated when complications in a patient with Crohn disease are suspected.
The big problem of CT is the use of ionizing radiation,
applied to young patients with inflammatory bowel disease who will undergo several imaging examinations during the disease process.
Features that suggest Crohn disease:
Wall thickening (Fig.
6,
8). A bowel segment with a wall thickness > 4 mm is considered pathological,
having in consideration that the loop must be well-distended.
A heterogeneous pattern means stratification of the thickened bowel wall,
either in two or three alternating layers.
The parietal enhancement with three layers is called target sign and corresponds to enhanced mucosa,
surrounded of a edematous submucosa (hypodense ring) and high-density external ring formed by muscularis externa and serosa.
If mural stratification has only two layers,
called double rim sign,
an intense internal enhancement and hypodense periphery,
or the opposite,
is found.
Pre-stenotic dilatation (Fig.
9). A normal bowel lumen measures less than 2.5 cm.
It is easy to recognize by CT the stenotic segment and the pre-stenotic dilatation associated.
Active inflammation. The enhancement intensity of mucosa and serosa reflects clinical activity of Crohn disease.
Submucosal oedema from target sign, seen in active inflammation needs to be differentiated from the ring produced by an increased submucosa fatty deposition seen in long-standing disease.
In active disease,
bowel loops are more thickened as well.
Perienteric hipervascularization (Fig.
9). It is called comb sign,
and suggests acute exacerbation in patients with Crohn disease.
Separated loops. The displacement of the involved loop away from the normal small bowel seen in barium conventional techniques can be secondary to mesenteric lymph nodes,
mesenteric fibrofatty proliferation (creeping fat),
phlegmon or abscess.
CT allows differentiating between these possible causes in order to achieve an appropriate treatment.
Lymph nodes (Fig.
9). Mesenteric lymph nodes found in Crohn disease normally have a small size (3-8 mm).
If bigger adenopathies are found,
lymphoma or carcinoma should be excluded.
Mesenteric fibrofatty proliferation. It is the most common cause of separated bowel loops.
It consists on an increased volume and attenuation of mesenteric fat secondary to oedema and inflammatory cells accumulation.
Abscess. Abscess is presented as a round- oval mass with attenuation values closed to water.
If there is a well-formed capsule,
a peripheral enhancement can be shown,
while the necrotic material in the central zone doesn´t enhance.
It is possible to find gas inside (as bubbles or air-fluid level).
Phlegmon. It is an inflammatory mass in mesentery,
seen as a non-well defined high density area in the mesenteric fat.
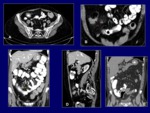
Fistulas and sinus tracts (Fig.
8,
10). These are features found in long-time disease.
On CT,
they are seen as hyperdense,
linear tracts with extension through the fat.
Air bubbles or positive oral contrast is possible to be found inside.
CT can demonstrate enterocutaneous or enterovesical fistulas better than barium conventional techniques,
being barium follow-through and conventional enteroclysis the election techniques in the diagnosis of entero-enteric fistulas.
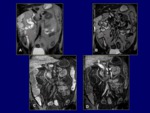
MR
Traditionally,
MRI has had a well-defined role in evaluation of anorectal complications of Crohn disease.
In recent years,
with the introduction of fast MRI sequences,
MR technique has emerged as an imaging modality that can be used in the assessment and diagnosis of Crohn disease,
as well as in the evaluation of inflammatory activity (Fig.
11).
The advantages of MRI include its lack of ionizing radiation and tissue contrast resolution.
It provides,
as well,
accurate anatomic detail,
depicts extraintestinal abnormalities and facilitates distinction between phlegmon,
abscesses,
and mesenteric adenopathy.
MR enterography examinations combine the spatial and temporal resolution of MRI with large volumes of ingested oral contrast material (eg.
Mannitol) to obtain luminal distention.
Although this modality may be less discomforting for the patient,
it may not produce similar distention of the bowel as can be obtained by an MR enteroclysis examination.
The enteroclysis technique enables optimal distention of the small bowel,
which results is accurate visualization of stenoses and obstructions.
RM enteroclysis has shown excellent correlation with conventional enteroclysis in grading small-bowel obstruction and functional information while also providing transmural and extramural visualization.
In this case,
nasojejunal intubation under fluoroscopy is required.
Features that suggest Crohn disease:
Wall thickening (Fig.
12,
15-17). It has been variable in active disease,
as described in many reports.
The general consensus is that concentric bowel wall thickening greater than 4 mm (4-10 mm),
added to hyperintense signal on T2-weighted images and gadolinium enhancement on T1 of the wall,
is suggestive of acute disease.
Enhancement patterns (Fig.
14-17). Contrast enhancement detects hypervascularization while hyperintensity on T2 identifies oedema secondary to inflammation.
Target sign enhancement pattern or mucosal enhancement is suggestive of inflammatory activity.
Mucosal anomalies. Aphthous ulcers and cobblestoning are difficult to be seen on RMI.
Distention and peristalsis. Cineloop sequences allow differentiate strictures secondary to inflammation from fibrotic ones.
Changes in mesenteric fat. Hyperintensity on T2-weighted images in mesentery is found in active inflammatory disease (local oedema and hipervascularization).
It is important to distinguish from fibrofatty proliferation,
seen in active and inactive disease.
Increased mesenteric vascularization (Fig.
15). Comb sign in mesenteric side is typical of an inflamed loop.
Enhanced local lymph nodes. It is also suggestive of activity in Crohn disease.
Abscesses and phlegmon. Well-defined in T1-weighted images with gadolinium,
with peripheral enhancement in abscesses (Fig.
14) and homogeneous in phlegmon.
Fistulas (Fig.
18). It is possible to identify liquid in the tract.
COMPLICATIONS
Common complications in Crohn disease have already been reviewed in the different sections,
corresponding to each imaging modality.
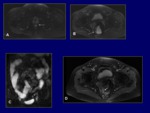
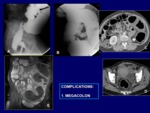
Although not common,
other complications like toxic megacolon (Fig.
19) (mainly in fibrostenosing form) or neoplasms (such as adenocarcinoma (Fig.
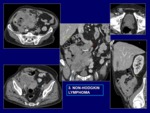
20) and lymphoma (Fig.
21); after 8-10 years of diagnosis of Crohn disease) may also occur.
Megacolon and most neoplasms will be identified by every modality used in the study of small bowels,
having CT and MRI a higher diagnostic sensitivity,
especially in neoplasms.