We reviewed all the cases of parasitic diseases that had been admitted to our institution since 2002 to 2012 and selected those which had imaging tests.
We examined these tests and selected representative images of the parasitic diseases,
reviewing the main imaging findings described in the current literature.
These are the selected parasites we found as representative cases.
- ECHINOCOCCOSIS or HYDATID DISEASE
Echinococcosis is caused by various forms of Echinococcus tapeworms (E.
granulosus,
E.
multilocularis mainly,
but also E.
vogeli and E.
oligarthrus).
Canides are the definitive host of this nematode,
but livestock and humans can be intermediate hosts.
Human echinococcosis occurs after the ingestion of the tapeworm eggs present in dog faeces.
These eggs eventually develop into larvae that enter portal circulation and end up forming cystic lesions in human tissues.
Any organ of the human body can be infected by these cysts,
but most frequent locations are liver and lung.
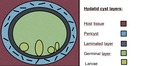
Hydatid cysts structure is very characteristic,
consisting in different layers. Fig. 1.
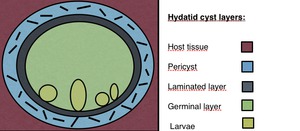
Fig. 1: Hydatid cyst structure. Pericyst: it is the outer layer and is fundamentally composed of a fibrous tissue formed as a protective response of the host to the parasite. Middle laminated layer: which is acellular and permits the passage of nutrients. Germinal layer: where the larvae are produced. This two last layers form the true wall of the cyst, usually referred as the endocyst.
Chronic hydatid cysts can calcify when the larvae die but sometimes calcified cysts can still have alive larvae.
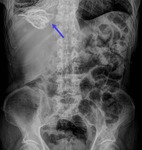
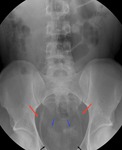
Some hydatid cysts are discovered incidentally as characteristic calcified lesions or a mass on conventional plain abdominal radiographs. Fig. 2.
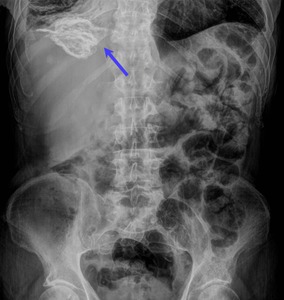
Fig. 2: Anteroposterior abdomen radiograph showing an hepatic calcified hydatid cyst (blue arrow) in a 75 year old patient found incidentally.
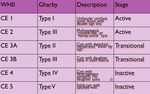
There are two international classificaions of echinococcal cysts based on ultrasonographic patterns that correlate with the evolution and viability of the cyst. Table 2.
- Gharby classification (1981)
- WHO Informal Working Group on echinococcosis (2003)
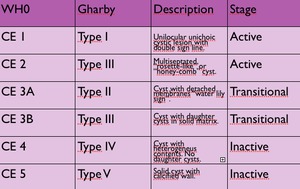
Table 2: WHO and Gharby comparative ultrasound classification on echinococcal cysts.
CT scanning is also a precise test for the diagnosis and classification of the disease.
MR,
if available,
also makes an accurate demonstration of the morphologic patterns of the cyst.
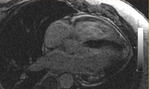
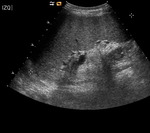
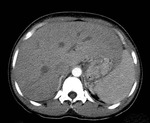
Examples of hydatid cysts found in US,
CT and MR: Fig. 3, Fig. 4, Fig. 5, Fig. 6, Fig. 7, Fig. 8, Fig. 9, Fig. 10.
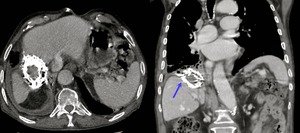
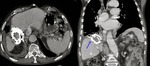
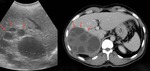
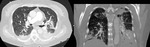
Fig. 3: Axial and coronal CT views showing a calcified hepatic hydatid cyst with transdiaphragmatic migration of the cyst. A rare complication seen in patients with hydatic hepatic disease.
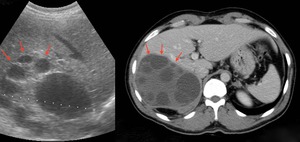
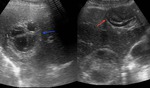
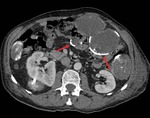
Fig. 4: Sagital sonogram and axial enhanced scan showing a multivesicular hydatid cyst. Note the typical peripheral location of the daughter vesicles within the mother cyst (red arrows).
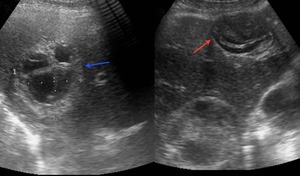
Fig. 5: Sagital sonogram showing two hydatid cysts. Note that one of them contains detached membranes with a serpentine appearance (red arrows). The other one shows a different appearance, an hydatid cyst with daughter vesicles. Note that the daughter cysts occupy almost the entire volume of the mother cyst (rosette sign).
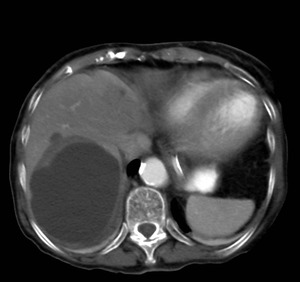
Fig. 6: Axial enhanced CT scan showing an unilocular hydatid hepatic cyst.
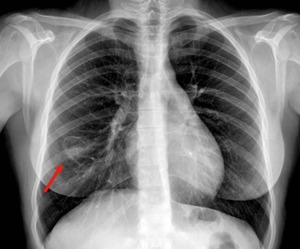
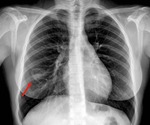
Fig. 7: Posteroanterior chest radiograph showing a cavitary lesion in the right inferior lobe with solid contents settled on the most dependent part of the cavity corresponding to a hydatid cyst.
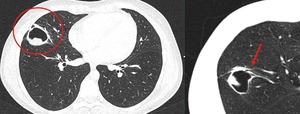
Fig. 8: Axial high resolution CT showing the hydatid cyst with solid content. Axial oblique MPR was performed confirming free communication of the cavitary lesion with the airway (red arrow).
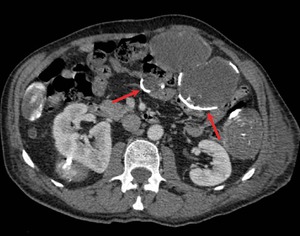
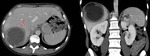
Fig. 9: Multiple peritoneal hydatid cysts secondary to seeding of a ruptured hepatic hydatid cyst. Note the peripheral calcifications of the cysts (red arrows).
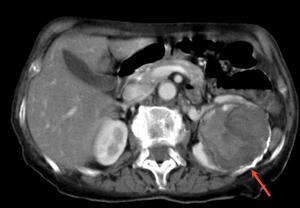
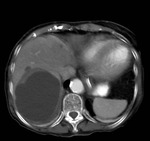
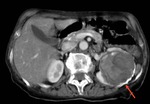
Fig. 10: Renal hydatid cyst with heterogeneus content on an axial enhanced CT scan. Note the peripheral calcification of the cyst (red arrow).
Strongyloidiasis is a prevalent disease worldwide caused by S.
stercolaris. The filariform larvae penetrate the human skin to start its parasitic cycle.
This nematode usually affects inmunompromised patients.
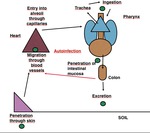
It is important to stand out that the most dangerous aspect of this parasite is that it can undergo a complete life cycle within the human body (autoinfection or hyperinfection) and cause a fatal hyperinfection syndrome. Fig. 11.
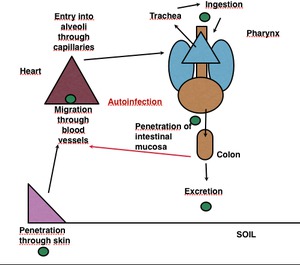
Fig. 11: Life cycle of S. stercolaris in the human body.
Hyperinfection syndrome consists of a massive parasitic infestation leading to a systemic inflammatory response syndrome in the setting of an inmunocompromised patient.
Radiologic findings of this syndrome consist of extensive pulmonary opacities due to the parasite itself,
or by the host response to the parasite (noncardiogenic oedema,
haemorraghe or both).
In the gastrointestinal tract we can find non specific changes such as bowell wall thickening. Fig. 12 and Fig. 13.
Cysticercosis is caused by the larval form of the pork tapeworm Taenia solium and it is an infection with worldwide prevalence.This infection is caused by ingesting raw or undercooked meat from an infected animal host.
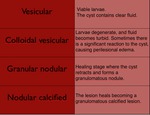
Neurocysticercosis has been classified into three main stages depending on the developmental stage and host response to the parasite. Table 3.
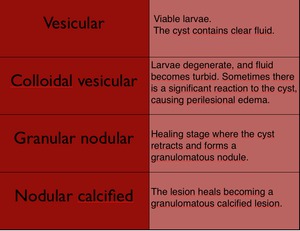
Table 3: Developmental stages of Neurocysticercosis
Neurocysticercosis is the most common parasitic disease of the central nervous system and the most common cause of acquired epilepsy,
being a major public health problem worldwide.
The diagnosis of neurocysticercosis is based on a combination ofclinical findings and noninvasive imaging findings (CT and MR imaging ),
so radiologysts play an important role in detecting and monitoring this disease.
The best diagnostic technique to study neurocysticercosis is MRI,
being the most sensitive imaging modality to detect all of the stages of the disease with the exception of the forth one,
the nodular calcified,
which is better detected by non-enhanced CT. Fig. 14, Fig. 15, Fig. 16 and Fig. 17.
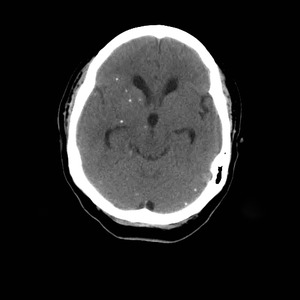
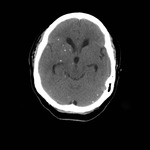
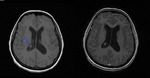
Fig. 14: Axial non-enhanced CT of the brain show multiple, punctate, calcified nodules corresponding to neurocysticercosis at the nodular calcified stage.
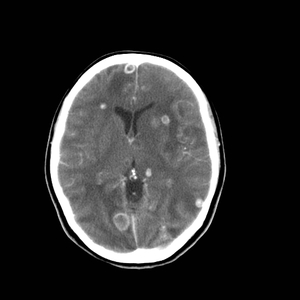
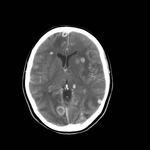
Fig. 15: Axial enhanced CT of the brain showing multiple enhancing lesions surrounded by brain edema in a patient presenting neurocysticercosis at a colloidal vesicular stage.
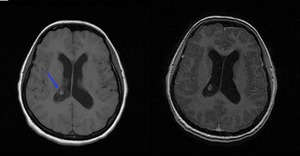
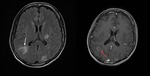
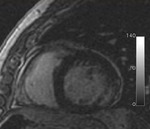
Fig. 16: Subarachnoid vesicular neurocysticercosis. Axial T1-WI and axial T1-WI gadolinium enhanced MR images showing a cystic lesion in the right lateral ventricular trigone containing an eccentric scolex (blue arrow) hyperintense to CSF with no edema and no rim enhancement.
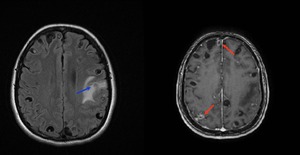
Fig. 17: Axial fluid-attenuated inversion recovery and axial T1-WI gadolinium enhanced MR images showing multiple cystic cortical and subcortical lesions some of them showing ring enhancement (red arrows) and one of them surrounded by edema (blue arrow) in a neurocysticercosis at a colloidal vesicular stage.
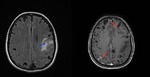
Toxoplasmosis is an infection caused byT.
gondii which is a protozoan with worldwide prevalence.
It is more frequent in the tropics and in areas where cats are numerous.
T.
gondii life cycle has two differentiated phases.
The so-called sexual phase takes places in feline,
which are the parasite’s primary host.
The following asexual phase can occur in other warm-blooded animals,
including humans.
Infection in humans can happen by two means,
either the ingestion of infected cat’s faeces or blood transmission from the mother to the foetus.
Primary infection is asymptomatic in 90% asymptomatic or can cause influenza-like symptoms.
After the infection T.
gondii can remain latent in the human organism.
Fetal infection can cause congenital toxoplasmosis.
Immunocompromised individuals can develop more aggressive forms of the disease,
specially in the central nervous system.
Radiological studies are useful in the diagnosis of both adult and congenital disease.
This parasite infects the Central Nervous System of approximately 10% of AIDS or inmunocompromised patients,
being the lesions generally located in the basal ganglia,
corticomedullary junction,
white matter and periventricular region.
In congenital infection,
calcifications,
hydrocephalus and microcephaly are common.
However,
calcifications are very rare in the adult form,
where we usually find lesions with surrounding oedema and mass effet that enhance on post-contrast images.
Fig. 18 and Fig. 19.
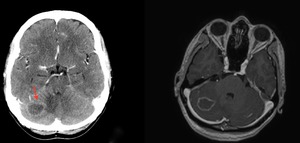
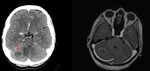
Fig. 18: Intracranial toxoplasmosis in a 45 year-old patient with AIDS. Axial enhanced CT and T1-WI gadolinium enhanced MR image showing a lesion with ring enhancement (red arrow), mild mass effect and surrounding edema in the right hemisphere of cerebellum.
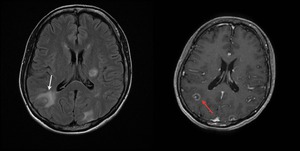
Fig. 19: Axial fluid-attenuated inversion recovery and T1-WI gadolinium enhanced MR images showing multiple lesions with perilesional edema (white arrow), and ring enhancement (red arrow).
Schistosomiasis is a group of parasitic infections that are endemic in Africa,
the Middle East,
and some parts of India and South America.
It is caused by various trematodes of the genus Schistosoma,
being the only trematode that invades human body through the skin (the other trematodes infect only via ingestion).
This parasite's intermediate host are fresh-water snails,
and humans are its definitive host. Fig. 20.
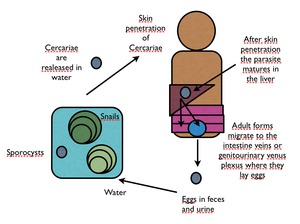
Fig. 20: Schistosoma species life cycle.
There are two major forms of schistosomiasis: intestinal and genitourinary.
Intestinal forms are less frequent.
Genitorurinary schistosomiasis is caused by S.
haematobium,
one of the five fluke species that account for most human cases of schistosomiasis.
S.
haematobium tends to affect the inferior tract of the genitourinary system,
being the ureter and the bladder chiefly affected. Fig. 21 and Fig. 22.
Kidneys are not affected until a late stage of the disease.
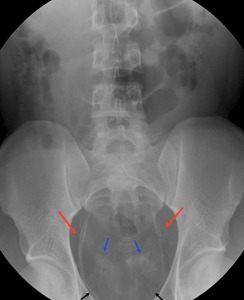
Fig. 21: Anteroposterior abdomen radiograph showing calcification of the pelvic segment of both ureters (red arrows), calcified vas deferens (blue arrows) and bladder (black arrows). Case courtesy J.Barrera/ES
In the early stages,
the mucosa is oedematous and haemorrhagic.
Later,
the tissue becomes fibrotic and calcificied granulomas appear (dead calcified eggs).
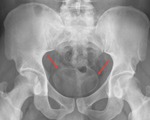
There is a pathognomonic classic sign for chronic urinary schistosomiasis: a totally calcified bladder resembling a foetus head in the pelvis. Fig. 22.
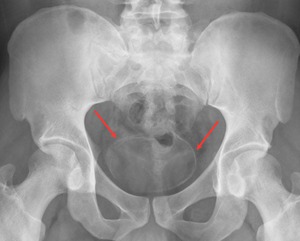
Fig. 22: Anteroposterior pelvis radiograph showing a totally calcified bladder in a chronic form of schistosomiasis.
Amoebiasis is widely spread all over the world with an estimated 10% of the world´s population being infected.
It is more prevalent in India,
the Far East,
Africa,
and Central and South America.
E.
histolytica infects the gastrointestinal tract sometimes remaining in a latent stage for many years,
but some other times it causes more agressive forms of the disease ranging from mild colitis to dysentery or development of liver abscesses.
Amoebic liver abscess is the most frequent extraintestinal complication of amoebiasis.
Amoebic liver abscess has imaging features sometimes indistinguishable from a pyogenic abscess,
however,
epidemilogic and clinical features,
as well as positive amebic titers,
help to make the diagnosis.
US,
CT and MRI are useful to detect this liver lesions. Fig. 23.
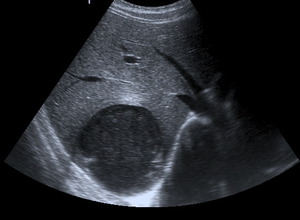
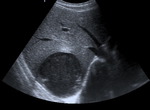
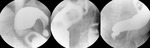
Fig. 23: Axial sonogram showing rounded, well-defined cystic hypoechoic lesion with posterior acoustic enhancement in relationship with an amoebic liver abscess.
These abscesses tend to be located near the capsule and usually have an enhancing thick wall with little perilesional oedema,
characteristic findings of amebic abscess. Fig. 24.
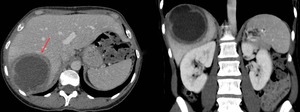
Fig. 24: Axial and coronal enhanced CT scan images showing a large cystic mass in the right hepatic lobe corresponding to an amoebic abscess. Note the enhanced thickened wall of the lesion (red arrow).
Dracunculiasis is caused by the Guinea worm,
Dracunculus medinensis. The disease is limited to remote rural villlages in Sub-Saharan Africa.
Erradication programs have succesfully reduced this disease in last 20 years.
Human infection occurs with the ingestion of water contaminated with larvae,
which eventually migrate to the subcutaneous tissues.
This parasite infection commonly affects the lower limb joints,
specially the knee.
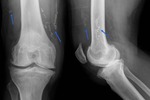
We usually find this parasite incidentally as soft tissue “worm-like” calcifications. Fig. 25
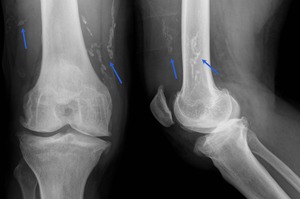
Fig. 25: Posteroanterior and lateral knee radiograph showing multiple calcified Guinea Worms in a 45 year-old Sub-Saharan patient.
Tripanosoma cruzi is the aetiologic agent for trypanosomiasis or Chagas’ disease.
This parasite is adquired by humans when the vector of the parasite,
an insect from the Reduviidae family,
defecate infected faeces on the skin and bites the individual.
Scratching of the bite favours the inoculation of the parasites which eventually enter the blood stream.
Chagas’ disease is widely spread in Central and South America.
Around 15-30% of the infected individuals develop chronic forms of the disease,
mainly affecting the heart,
digestive tract and nervous system.
Chagas’ disease has different stages.
In early phases of the disease,
the patient may present cardiologic and neurologic symptoms (myocarditis and meningoencephalitis).
Later,
in the sub-acute phase,
hepatoesplenomegaly and lymphadenopathy are the most common findings.
Megaoesophagus (Chagasic achalasia) and megacolon are found in the chronic phase of the disease. Fig. 26.
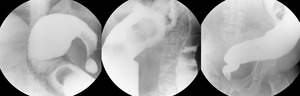
Fig. 26: A barium enema of a 42 year-old Bolivian patient showing megacolon, fundamentally at the expense of sigmoid colon. Chagas disease was confirmed by serologic testing.
Chronic Chagas disease can also afect the heart,
causing myocardial necrosis and fibrosis either by local parasite's persistance and/or autoinmmune affection.
Myocardial fibrosis or necrosis is seen in MR as a characteristic delayed contrast enhancement after Gadolinium contrast injection. Fig. 27 and Fig. 28.
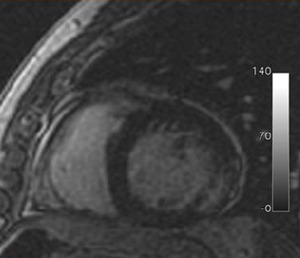
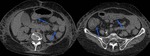
Fig. 27: Cardiac chronic Chagas disease. South American male diagnosed of T. cruzi infection. Cardiac MR delayed contrast enhanced sequence with two chambers-short axis view. We can appreciate delayed transmural enhancement in non-coronary territory (basal infero-lateral segment), indicating myocardial fibrosis or necrosis.
Case courtesy B.Cabeza and A.Bustos/ES
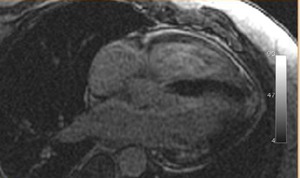
Fig. 28: Cardiac chronic Chagas disease. South American male diagnosed of T. cruzi infection. Cardiac MR delayed contrast enhanced sequence with four chambers view. We can appreciate delayed transmural enhancement in the cardiac apex, indicating myocardial fibrosis or necrosis.
Case courtesy B.Cabeza and A.Bustos/ES
Leishmaniasis is a protozoal disease that cause a wide spectrum of clinical findings and symptoms from cutaneous ulcerations to systemic disease and it is transmitted by the bite of certain species of sandflies from the genus Phlebotomus.
This parasites have been identified all around the world,
however,
several Leishmania species exist in endemic regions.
Such is the case of as L.
infantum,
a special specie that affects the reticulo-endothelium system of several organs including liver and spleen,
and which may be found in the Mediterranean are and the Middle East.
The imaging findings of leishmaniasis are non-specific,
usually consisting of hepatoesplenomegaly and enlargement of lymph nodes. Fig. 29 and Fig. 30.
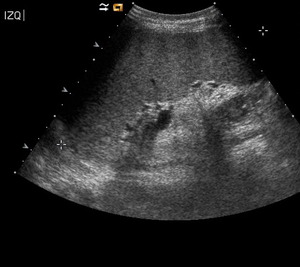
Fig. 29: Axial sonogram showing esplenomegaly in a 52 year-old patient presenting with fever, weight loss and pancytopenia. Serological exams confirmed visceral leishmaniasis.
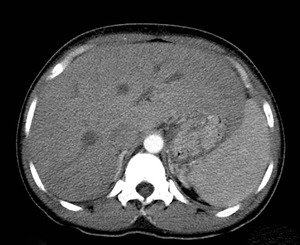
Fig. 30: Axial enhanced CT scan showing hepatoesplenomegaly in a patient with visceral leishmaniasis.