One of the most important contributions of the use of CEUS in abdominal pathology is its ability to detect enhancement since this property allows the identification of the cystic or solid components and visualize the vascularity of the internal nodules,
septa and cystic wall.
We illustrate examples of abdominal cystic lesions in different areas including liver,
spleen,
pancreas,
renal,
mesentery and abdominal cavity,
gastrointestinal tract or gynecologic organs.
Key findings will be discussed.
LIVER PATHOLOGY
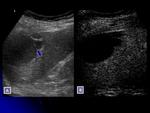
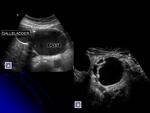
-The most common non neoplastic benign lesions of the liver are simple cysts.
They show anechoic content,
with smooth borders,
thin-walled (1 mm or less) with no septations or mural nodularity.
Fig. 1
Simple cyst is detected in US,
no further imaging studies are required.
Cyst content greater than water may be due to hemorrhage or infection.
In CEUS,
both show internal absence of uptake (intralesional enhancement).
The infected cyst can show a peripheral enhancement corresponding to the thick wall.
Fig. 2
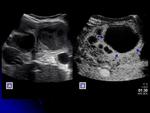
- Biliary cystic neoplasms are suspected with large,
uni or multilocular anechoic mass.
Cystadenoma are more typically multilocular lesion and cystadenocarcinoma may demonstrate nodularity of margins and mural or septal nodule with diameter > 1 cm.
CEUS is useful to demonstrate vascularity of the lesions and mural or nodule enhancement.
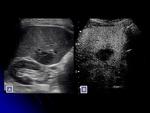
-Hepatic pyogenic abscesses may manifest as a single lesion or cluster of small abscesses coalesce into a single large cavity.
They may be hypoechoic and ill-defined,
sometimes at initial stages look like solid lesions or indeterminate.
The lesion will be more defined and the content will be more liquid when the liquefaction process will be occurring.
Three patterns of enhancement have been described:
- type 1,
peripheral ring enhancement and absence of enhancement intralesional;
- type 2,
enhancement areas (septa) alternating with liquefied areas (honeycomb appearance);
- type 3,
most of the lesion shows enhancement at arterial phase and subsequent washing out. Fig. 3
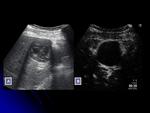
- Hydatid cyst has variable appearances in US: well-defined unilocular or multilocular,
numerous peripheral daughter vesicles,
cyst with undulating floating membranes,
solid pattern or calcification (curved,
peripheral or thick).
The usefulness of CEUS is to confirm the absence of intralesional enhancement in cases with semisolid appearance or demonstrate membranes or daughters vesicles. Fig. 4
-Other lesions as cystic metastases (mucinous carcinoma,
neuroendocrine,
testicular carcinoma or sarcoma) may mimic a liver abscess and are differentiated in CEUS by enhancement of irregular walls or mural nodularity.
-Lesions treated with radiofrequency are seen on US as hypoechoic or anechoic lesions.
CEUS allows assessment of any viable tumor component that is seen as hypervascular nodules with subsequent washing localized in the periphery or intralesional.
Fig. 5 Besides being a fast and efficient technique,
can serve as a guide to direct and complete the treatment.
PANCREATIC PATHOLOGY
One of the major benefits of the use of contrast in the pancreas is its ability to differentiate between pseudocysts and cystic tumors:
- Pseudocysts are nonvascular,
and therefore,
show no internal enhancement at all vascular phases,
regardless of their internal content in B-mode US Fig. 6 .
In some cases,
arterial macrovessels can be seen through the lesion or the capsule can enhance,
findings typically found in the early stages.
The accuracy of CEUS to differentiate between pseudocysts and cystic neoplasia is about 100%.
-By contrast,
the detection of microvessels or enhancement of septa or nodules is typical of cystic tumors Fig. 7 Using surgical pathological findings as the reference standard,
there are not significant differences between CEUS and MRI,
regarding in the detection of intralesional septa or nodules.
However,
the use of contrast does not differentiate between benign and malignant tumors.
- Serous cystoadenoma typically have a microcystic appearance,
which multiple septa.
CEUS can improve tumor characterization showing enhancement of septa and typical pattern in honeycomb Fig. 8 .
When cysts are small,
CEUS may suggest a solid lesion because of the enhancement is homogeneous.
- Mucinous cystadenomas and cystadenocarcinomas show thick walls and internal nodules that enhance on CEUS,
similar to intraductal papillary mucinous tumor of secondary ducts.
However,
CEUS cannot detect the communication with the pancreatic duct,
which is the key for the diagnostic process.
- Neuroendocrine tumors are often associated with multiple endocrine neoplasia (MEN).
They typically present as hyperenhaced masses on CEUS,
but in larger masses they may have cystic degeneration.
In these cases,
CEUS demonstrates intense and early enhancement of the solid component Fig. 9
SPLENIC PATHOLOGY
Splenic cystic lesions are usually solitary and include primary or true cysts,
acquired or pseudocysts and cystic neoplasms.
- Congenital cysts as the epidermoid cyst present incidentally; most of them are unilocular and solitary.
They usually have a thin wall and may contain debris,
hemorrhage or cholesterol crystals inside.
No rim or intracystic enhancement following contrast agent injection.
- Inflammatory cysts include hydatid cysts and abscesses.
A typical abscess shows absence of enhancement during all vascular phases.
However,
it can be impossible to differentiate an abscess with an enhancing capsule from a tumour with central necrosis and peripheral enhanced ring.
- In the context of acute trauma,
a pseudoaneurysm is easily demonstrated after contrast administration,
because of the intense and persistent enhancement seen from the arterial phase into a cystic lesion Fig. 10
- Cystic lymphangioma appear as a multilocular lesion with thin septa.
CEUS shows a characteristic moderate enhancement of the septa without enhancement of cystic spaces interposed Fig. 11 .
- “Cystic” splenic metastases can be seen in melanoma,
ADK of breast,
ovary and endometrium.
CEUS can show an irregular enhancement both inside as peripheral,
difficult to distinguish from bacterial abscess.
Consider the clinical setting.
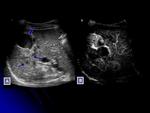
RENAL PATHOLOGY
Kidney cysts are very common (> 50% of patients over age 50 develop cysts).
Benign renal cysts can be simple or complex secondary to infection,
inflammation or bleeding.
They are defined as complex cysts if show internal echogenicity,
calcifications,
septa,
thick-walled or solid nodules.
B-mode US is not reliable to differentiate between benign complex cysts and cystic renal carcinomas (10%).
- The main clinically recognized indication of CEUS in the urinary system is the characterization of renal complex cysts.
CEUS has proven to be a useful technique to characterize complex renal cysts according to the Bosniak classification,
with similar results to CT or MRI.
CEUS detects the presence of enhancement in the thickened irregular wall,
septa or mural nodules,
which are the most specific signs suggesting malignancy.
Furthermore,
due to the absence of radiation,
CEUS is a useful technique for monitoring lesions managed conservatively.
The classification of cysts by Bosniak is defined with CEUS in this manner:
-Bosniak I: thin-walled cyst,
anechoic content,
without septa,
calcifications,
or solid components and there are not enhancement after contrast administration.
-Bosniak II: cyst with thin septa,
thin calcifications in a segment of the wall and may have a minimum enhancement of the septa without nodular enhancement. Fig. 12
-Bosniak IIf: containing multiple thin septa,
minimum wall thickening,
thin or nodular calcifications and can present minimal enhancement of the septa without nodular enhancement.
These are lesions that require monitoring.
-Bosniak III: irregular thickening of the walls or septa of the cyst with enhancement after contrast administration.
Solids poles are not observed.
Fig. 13 Hemorrhagic or inflammatory complex cysts are included in this category. Fig. 14
-Bosniak IV: nodule or soft tissue mass with contrast enhancement,
regardless of the enhancement of the wall or septa. Fig. 15
- In renal inflammatory pathology,
the use of sonographic contrast allows identify foci of pyelonephritis and confirm the presence of collections or abscesses,
showing the necrotic avascular component and the enhancement of the wall Fig. 16
GYNECOLOGICAL PATHOLOGY
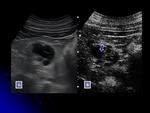
- Cystic adnexal lesions may be classified according to their sonographic appearance as uni or multilocular cystic masses.
US features suspicious for malignancy include a large adnexal mass,
the presence of solid component,
a wall thickness or papillary projection greater than 3 mm and the presence of ascitis and/or peritoneal disease.
CEUS is useful in demonstrate presence of vascularity of nodules or internal septations,
although there are overlap especially between benign and borderline tumors. Fig. 17 Fig. 18
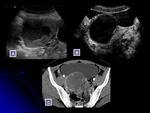
-The uterine cystic lesions are grouped into endometrial lesions (hematometra,
pyometra or submucosal lesions) or myometrial conditions (bicornuate uterus with rudimentary horn,
myoma with cystic degeneration or cystic adenomyosis).
CEUS may help to define its location and dependence as well as confirm cystic nature. Fig. 19 Fig. 20
PATHOLOGY OF THE PERITONEAL CAVITY OR RETROPERITONEAL
Cystic lesions of the intra or retroperitoneum can be classified as either neoplastic or nonneoplastic.
Neoplastic lesions include mesenteric cyst (cystic lymphangioma,
cystic teratomas,
enteric duplication cyst,
cystic mesothelioma),
epidermoid cyst,
tailgut cyst,
pseudomyxoma peritoneal and cystic change in solid neoplasms.
Nonneoplastic lesions include post-surgical collections (lymphocele,
urinoma,
and hematoma) and abscesses.
- Post-surgical collections,
both intra or retroperitoneal,
include lymphocele,
seroma,
hematoma,
peritoneal inclusion cyst,
biloma or urinoma.
The antecedent of surgery or trauma is the key for the diagnosis,
but some of them may persist over time or can assess with difficulty due to the conditions under which the study is conducted.
In the case of trauma is useful for detecting active bleeding.
Fig. 21
- Abdominal abscesses are most commonly secondary to post-operative complication although can be in relation to acute appendicitis,
cholecystitis,
diverticulitis or Crohn´s disease.
CEUS may add information about etiology Fig. 22 ,
confirm the nature of collections,
define more accurately the size and complexity of echostructure Fig. 23 ,
so that asses the need for drainage.
- CEUS is very helpful in differentiating between phlegmon and abscess,
especially in cases where Doppler color signal is not very accurate within the inflammatory mass.
Phlegmons have internal enhancement while abscesses have peripheral enhanced wall,
Fig. 24 This is important in patient management,
because abscesses may require percutaneous or surgical drainage.
-Pseudomyxoma peritonei is seen as ecographic multiseptated peritoneal fluid.
Its appearance resembles semisolid component,
however on CEUS it is well differentiate because the masses or collections not show contrast enhancement.
The origin is the rupture of mucinous tumor of appendix.
Appendiceal mucocele is located in the right lower quadrant and manifests in US as well-demarcated cystic lesion,
round or ovoid,
sometimes with curvilinear calcification in the wall.
The CEUS can determine the malignant nature if is identified nodular,
thick or irregular wall or the presence of thick septa. Fig. 25
-In the evaluation of cystic tumors there are many overlapping characteristics among the various types.
Overall,
the intra-abdominal cystic tumors will require other techniques for characterization.
CEUS is useful to determine the solid component of the lesion.
Fig. 26