Normal Anatomy
The portal vein drains the entire gastrointestinal tract excluding the anal canal.
The three main portal venous tributaries are the splenic,
superior mesenteric and inferior mesenteric veins.
Smaller tributaries include the left gastric (coronary),
right gastric,
cystic,
and paraumbilical veins.
The portal vein divides into the left and right lobar veins. The right portal vein divides into anterior and posterior sectoral branches,
which subdivide into superior and inferior segmental branches which supply segments 5,
6,
7 & 8 (Fig.
1).
The left portal vein follows a concave anterior path to the ligamentum teres,
supplying the lateral sector of the left lobe (segments 2 and 3) and terminates in the superior and inferior segmental branches of segment 4 (Fig.
1).
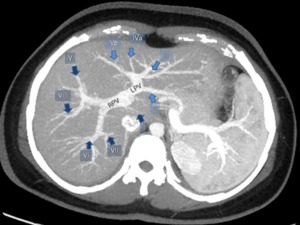
Fig. 1: Maximum intensity projection (MIP) contrast enhanced computed tomography (CECT) depicts the segmental divisions of the right portal vein (RPV) and left portal vein (LPV).
Variant Anatomy
Variation in the branching pattern of the portal vein have been reported in up to 35% of the population (1,2).
The five most common patterns account include:
- Trifurcation of the main portal vein (Fig.
2a)
- Right posterior sectoral branch arising from the main portal vein (Fig.
2b)
- Segment VII branch arising as a separate branch of right portal vein (Fig.
2c)
- Segment VI branch arising as a separate branch of right portal vein (Fig.
2d)
- Right anterior sectoral branch arising off the left portal vein (Fig 3.
a,b)
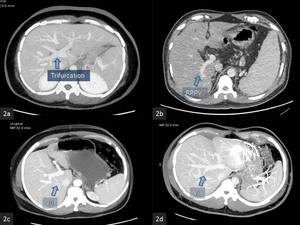
Fig. 2: MIP CECT
(a). Trifurcation of the main portal vein.
(b) Right posterior sectoral portal vein (RPPV) branch arising from the main portal vein
(c) Segment VII branch arising as a separate branch of right portal vein.
(d) Segment VI branch arising as a separate branch of the right portal vein
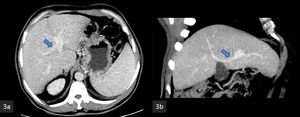
Fig. 3: MIP CECT
(a,b). The anterior sectoral branch (arrow) of the right portal vein is shown arising off the left portal vein.
Portal venous anatomy has been classified into 5 types,
which are described in table 1 (4):
|
Table 1.
Major portal venous variants (Atasoy et al.,
reference no.
4)
|
|
Type
|
Portal Vein Variant
|
Incidence%
|
|
1
|
Standard anatomy
|
65
|
|
2
|
Trifurcation
|
9
|
|
3
|
Right posterior portal vein as first branch of main portal vein
|
13
|
|
4
|
Segment VII branch as separate branch of right portal vein
|
1
|
|
5
|
Segment VI branch as separate branch of right portal vein
|
6
|
|
|
Other
|
6
|
Uncommon variants include Portal vein quadrification (Fig.
4a,b),
absence of portal vein bifurcation (Fig.
5 a,b),
collective branching intrahepatic PV (total ramification) and anomalous origin as well as branching of the segmental veins (3).
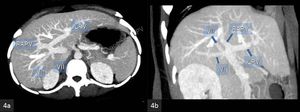
Fig. 4: MIP CECT
(a) Axial and (b) coronal images show hilar quadrification with common origin of the left portal vein, right anterior sectoral portal vein branch (RAPV) and segment 6 and 7 branches.
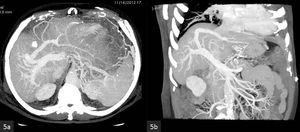
Fig. 5: MIP CECT
(a) Axial and (b) coronal images show absence of the portal vein bifurcation with aberrant origin of the left portal vein from the right anterior sectoral portal vein.
Clinical significance
- A type 2 portal vein increases the complexity of surgical procedures,
because portal vein clamping is technically challenging in such cases (5).
- Failure to recognize the anomalous origin of the right anterior sectoral branch (from the left portal vein) will result in unintended devascularisation of hepatic segments V and VIII when a left hemihepatectomy is perfromed.
- A type 3 portal vein variant is also more difficult for surgery because two portal vein anastomoses have to be done on two separate veins in the recipient.
- Anomalous segmental branches crossing the interlobar boundary render a potential donor unsuitable,
because such an anomalous vessel would result in a devascularized segment following hemihepatectomy.
Congenital Anomalies
Congenital anomalies of the main portal vein include congenital agenesis of the portal vein,
congenital agenesis of the major branches of the portal vein,
prepancreatic portal vein and duplication of the portal vein.
Congenital agenesis of the major branches of the portal vein may represent true aplasia or thrombosis of the affected lobe or segment during embryologic growth (6).
Prepancreatic Portal Vein
In the embryo venous blood from the foregut is drained by the parallel vitelline veins (Fig.
6a),
which are connected by three anastomoses:
- The superior (cranial) anastomosis (intrahepatic)
- The middle anastomosis behind the duodenum
- The inferior (caudal) anastomosis in front of the duodenum.
In the ensuing weeks the caudal anastomosis,
the lower portion of the right vitelline vein,
and the upper part of the left vitelline vein disappear,
leaving the middle anastomosis behind the duodenum.
The portal vein is thus derived from lower portion of the left vitelline vein,
the middle anstomosis and the upper portion of the right vitelline vein (7).
Normally,
the ventral and dorsal pancreatic buds arise in front of the right and left vitelline veins,
respectively (Fig.
6b).
A Prepancreatic retroduodenal (PPRD) portal vein develops when the dorsal pancreatic bud develops posterior to the left vitelline vein,
instead of the normal anterior position (Fig.
6c) (8).
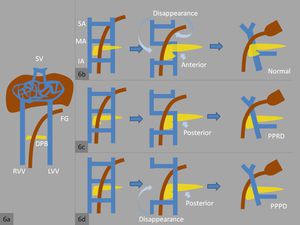
Fig. 6: (a) 5 mm embryo - normally, the ventral and dorsal pancreatic buds arise in front of the right and left vitelline veins, respectively.
(b) 9 mm embryo - normally, the lower portion of the right vitelline vein and and upper portion of the left vitelline vein disappear.
(c) A Prepancreatic retroduodenal (PPRD) portal vein develops when the dorsal pancreatic bud develops posterior to the left vitelline vein.
(d) A Prepancreatic preduodenal (PPPD) portal vein develops when the dorsal pancreatic bud develops posterior to the left vitelline vein, and the upper and lower portions of the right vitelline vein disappear in addition to the middle anastomosis.
References: Ankur Dev
PPRD veins have the following characteristics:
- PPRDs are L-shaped or inverted L-shaped (Fig 7a,b)
- PPRDs runs in front of or parallel to the CBD
- Most PPRDs are not associated with intestinal malrotation or duodenal,
biliary or pancreatic anomalies
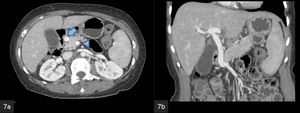
Fig. 7: MIP CECT
(a) Axial and (b) coronal images show prepancreatic retroduodenal course of the main portal vein.
A Prepancreatic preduodenal (PPPD) portal vein develops when the dorsal pancreatic bud develops posterior to the left vitelline vein,
and the upper and lower portions of the right vitelline vein disappear in addition to the middle anastomosis (Fig.
6d).
PPPDs are frequently associated with intestinal malrotation or duodenal,
biliary or pancreatic anomalies.
A prepancreatic portal vein that is unrecognised clinically carries the risk of intraoperative complications,
such as hemorrhage and accidental ligation of the anomalous portal vein.
Duplication of the portal vein
Duplication of the portal vein is an uncommon developmental anomaly in which the splenic vein gives rise to the retropancreatic retroduodenal portal vein.
In addition,
a second preduodenal portal vein develops from the confluence of the superior and inferior mesenteric veins,
which runs anterior to the head of the pancreas and duodenum (Fig.
8); upon reaching the gastrohepatic ligament,
it penetrates the quadrate lobe of the liver to the left of the gallbladder (9).
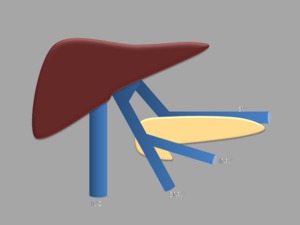
Fig. 8: The splenic vein (SV) gives rise to one portal vein, in addition a second prepancreatic portal vein arises from the confluence of the superior and inferior mesenteric veins (SMV, IMV). (IVC - inferior vena cava).
References: Ankur Dev
Congenital extrahepatic portocaval shunt (congenital absence of the portal vein, Abernethy malformation)
The Abernethy malformation,
first reported in 1793 by the British surgeon John Abernethy,
is a congenital anomaly in which splanchnic blood drains directly into the inferior vena (IVC).
The diversion of splanchnic blood into the IVC may be complete,
in the form an end-to-side portocaval anastomosis with deficient intrahepatic portal veins (Fig.
10) or partial,
via a side-to-side portocaval union (Fig.
11) portal vein (Table 2) (10).
|
Table 2.
Classification of congenital extrahepatic portocaval shunt (Morgan and Superina,
reference no.
10)
|
|
Type 1a
|
End-to-side portocaval anatomosis with independent drainage of the superior mesenteric vein and splenic vein
|
|
Type 1b
|
End-to-side portocaval anastomoses with convergence of the superior mesenteric vein and splenic vein
|
|
Type 2
|
Side-to-side portocaval anastomoses
|
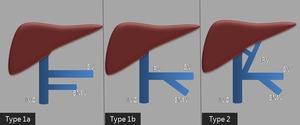
Fig. 9: Classification of congenital extrahepatic portocaval shunts
Type 1a: End-to-side portocaval anatomosis with independent drainage of the superior mesenteric vein (SMV) and splenic vein (SV).
Type 1b: End-to-side portocaval anastomoses with convergence of the superior mesenteric vein and splenic vein.
Type 2: Side-to-side portocaval anastomoses.
IVC - inferior vena cava).
References: Ankur Dev
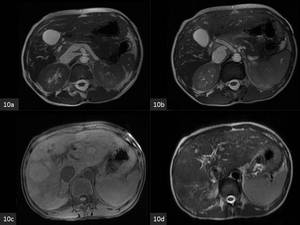
Fig. 10: Axial MRI
(a). T2-weighted steady-state free precession image shows an end to side portocaval shunt.
(b). Fig 10 a. T2-weighted steady-state free precession image shows hypertrophy of the hepatic artery and absent intrahepatic portal vein.
(c). T1-weighted image shows multiple hyperintense large regenerative nodules.
(d). The large regenerative nodules are hypointense on the T2-weighted image.
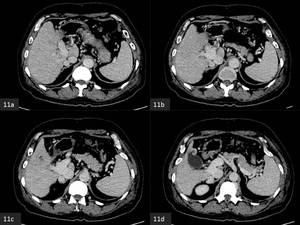
Fig. 11: Axial CECT
(a-d) A side to side portocaval shunt is observed on serial images.
The age at diagnosis ranges from intrauterine life to 76 years.
Patients may be asymptomatic and may be diagnosed incidentally when imaging is performed for some other reason.
Others may develop mild liver dysfunction with transaminitis.
Growth retardation has been reported in children.
Uncommon clinical presentations include hyperammonemia,
hepatic encephalopathy,
pulmonary hypertension and hepatopulmonary syndrome.
Regenerative nodules and benign as well as malignant hepatic neoplasms have been reported in 40% of published cases,
therefore some patients present with a hepatic mass (11).
Anomalies associated with the type 1 Abernethy malformation include (in decreasing frequency):
- Congenital heart disease
- Polysplenia
- Biliary atresia
- Skeletal anomalies
- Renal tract anomalies
Management strategies for patients with an asymptomatic shunt include clinical observation,
monitoring of hepatic function and imaging follow-up.
Liver transplantation is the only definitive treatment option in patients with type I malformation,
indicated in cases with refractory hepatic encephalopathy,
hepatopulmonary syndrome,
associated biliary atresia and clinically significant or malignant liver neoplasms.
In patients with symptomatic type 2 malformation,
the shunt can be occluded by means of surgery or transcatheter coil embolisation.
Acquired Abnormalities
Portal hypertension
Definition: Portal venous pressure greater than 12 mm Hg or hepatic venous pressure gradient (the difference between the wedged and the free hepatic venous pressures) greater than 5 mm Hg
Aetiology:
- Presinsoidal - Extrahepatic portal venous obstruction, hepatic fibrosis
- Sinusoidal - cirrhosis,
sclerosing cholangitis
- Postsinusoidal - Budd-Chiari syndrome,
heart failure,
constrictive pericarditis
Cross-sectional imaging findings (12,13):
- Portal vein calibre > 13 mm (Fig.
12a)
- Coronary vein calibre > 4mm
- Recanalized umbilical vein calibre > 3mm
- Portosytemic Collaterals
- Main portal vien diameter of less than 1 cm is highly specific but not sensitive for hepatofugal flow (Fig.
12c,d)
- Splenomegaly
- Ascites
Doppler ultrasound findings (14):
- Slow flow with peak velocity less than 16 cm/sec
- Hepatofugal flow
- Loss of respiratory increase of splanchnic vein calibre
Portosystemic collaterals: More than twenty different splanchnic to systemic venous collateral pathways have been described; most of these derive from the mesenteric and splenic veins.
The portal vein gives rise to the following collaterals (15):
- Coronary - gastro-oesophageal veins - azygos vein/gastrorenal shunt (Fig.
12b)
- Paraumbilical vein - superior epigastric - internal thoracic - subclavian vein (Fig.
13 a,b)
- Paraumbilical vein - superior epigastric - inferior epigastric - external iliac/femoral vein (Fig.
14 a,b)
- Portal vein peripheral branch - left inferior phrenic vein - left renal vein (left infradiaphragmatic shunt) (Fig.
15 a-d)
- Left portal vein - aberrent left gastric vein - azygos vein/gastrorenal shunt (Fig.
16 a)
- Portal vein peripheral branch - intercostal - internal thoracic vein (right infradiaphragmatic shunt) (Fig.
16 b)
- Right posterior portal vein branch - IVC (Fig.
16 c,d)
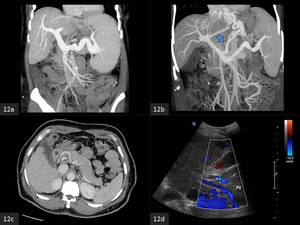
Fig. 12: (a) Coronal MIP CECT. Dilatation of the portal and splenic veins is observed.
(b). Coronal MIP CECT. A dilated coronary vein draining into gasto-oesophageal varices is observed.
(c). Axial CECT. A portal vein (measuring less than 10 mm in calibre) indicates hepatofugal flow in cirrhotics, confirmed on the Doppler ultrasound (d).
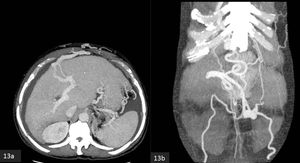
Fig. 13: Axial (a) and coronal (b) MIP CECT. A dilated paraumbilical vein shunts blood to the internal thoracic veins via the the superior epigastric veins. Shunting is also evident into the inferior epigastric veins.
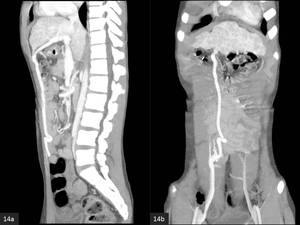
Fig. 14: Axial (a) and coronal (b) MIP CECT. A dilated paraumbilical vein shunts blood to the right external iliac vein via the the right superior and inferior epigastric veins.
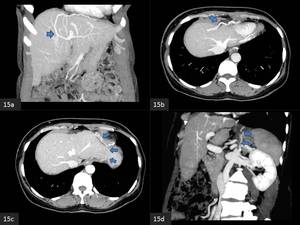
Fig. 15: Coronal (a,d) and axial (b,c) MIP CECT. An extrahepatic shunt drains splanchnic blood from a peripheral left portal vein branch into the left renal vein via the left inferior phrenic vein.
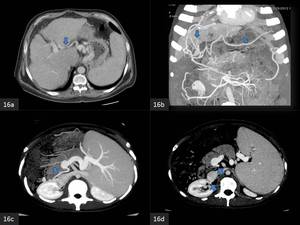
Fig. 16: (a). Axial MIP CECT. An aberrant left gastric vein runs along the hepatogastric ligament, shunting blood between the left portal vein and gastric veins.
(b). Coronal MIP CECT. An extrahepatic shunt drains splanchnic blood from a peripheral left portal vein branch into the left intercostal vein.
(c,d). Axial MIP CECT. A collateral is observed arising from the portal vein and draining into the inferior vena cava.
Portal vein aneurysm
The maximum portal vein calibre never exceeds 1.5 cm in in healthy individuals and 1.9 cm in cirrhotic patients.
A calibre exceeding 2 cm is thus regarded as aneurysmal (16).
Aetiolgy:
- Portal hypertension underlying hepatocellular disease (most common)
- Portal vein thrombosis
- Severe pancreatitis
- Trauma
- Malignant invasion of the portal vein
Intrahepatic aneurysms have a tendency to occur at bifurcations (Fig 17 a,b).
The commonest site for extrahepatic portal vein aneurysms is at the confluence of the superior mesenteric and splenic veins.
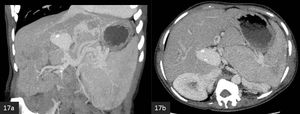
Fig. 17: (a) Coronal and (b) axial MIP CECT. A portal vein aneurysm is visualised at the hepatic hilus.
Asymptomatic,
small aneurysms in patients without portal hypertension or cirrhosis are often managed conservatively,
with regular ultrasound follow-up.
Bypass surgery or aneurysmorrhaphy is reserved for symptomatic cases,
or if the aneurysm expands and is deemed to be at high risk of thrombosis or rupture (17).
Arterioportal fistula
Communication between a hepatic arterial branch and the portal vein at the level of the trunk,
peribiliary venules or sinusoids results in redistribution of the arterial inflow into a localised region of portal venous flow.
Arterioportal shunting is characterised by early and sustained enhancement of the portal vein during arterial phase.
Distal to the shunt,
a wedge shaped subsegmental area of transient arterial enhancement which becomes isoattenuating to liver parenchyma in the portal venous phase,
is typically present (18).
Reduction or loss of geographic enhancement of the spleen may be seen in large arterioportal shunts,
attributed to reduced splenic artery inflow (19).
Aetiology:
- Posttraumatic - occurring after blunt or penetrating injury
- Iatrogenic - occurring after biopsy or an interventional procedure
- Spontaneous - in the cirrhotic liver
- Hypervascular tumors - hepatocellular carcinoma and hepatic hemangiomas (Fig.
15 a,b)
- Spontaneous rupture of a hepatic artery aneurysm into a portal vein
- Arteriovenous malformations (Fig.
15 c,d)
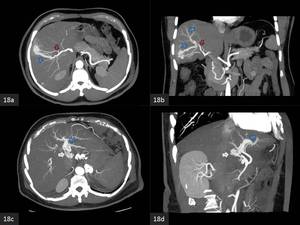
Fig. 18: MIP Arterial phase CECT
(a) Axial and (b) coronal images show an arterioportal fistula due to a haemangioma in the right hepatic lobe. Feeding arteries (red arrows) and the draining right anterior portal vein (blue arrows) are observed.
Fig 18 (c) Axial and (d) coronal images show an arterioportal fistula due to a hepatic arteriovenous malformation in the left lobe (white arrow) with early enhancement of the left portal vein (blue arrow).
Minute arterioportal shunts commonly produce perfusion anomalies in cirrhosis, appearing as small,
wedge-shaped,
subcapsular areas of increased attenuation on the hepatic arterial phase which become isoattenuating during the portal venous phase.
It may be difficult to distinguish an arterioportal shunt from an early small hepatocellular carcinoma.
In such situations,
repeat imaging in 6 months usually demonstrates the resolution or stability of an arterioportal shunt,
as opposed to growth for a hepatocellular carcinoma.
Portal vein thrombosis
Thrombosis of the portal vein and/or its branches and tributaries,
which include the splenic vein and the superior and inferior mesenteric veins.
Aetiology (Virchow's triad):
- Altered hemodynamics - liver cirrhosis,
Budd-Chiari syndrome
- Hypercoagulable state
- Endothelial damage- sepsis,
cholangitis and pancreatitis
Clinical symptoms – Thrombosis of the portal vein and/or its branches and tributaries,
which include the splenic vein and the superior and inferior mesenteric veins (20).
Acute thrombosis may be difficult to detect with grey-scale ultrasound alone,
as the thrombus is hypoechoic,
colour Doppler,
however,
demonstrates absent flow in the portal vein.
Chronic thrombus is more echogenic and easier to detect.
If portal vein Thrombosis (PVT) is diagnosed in B-mode ultrasound,
the flow profile should be assessed.
Pulsatile color signals within the thrombus on Doppler ultrasound, has been found to be 95– 100% specific for a malignant thrombus,
however the sensitivity is low (62%) (21).
Pulsatile flow is produced by tumour vessels within the thrombus and corresponds to the “thread and streak sign” on CT (Fig.
20 a,b) as well as conventional angiography (22,23).
On CT,
fresh bland PVT,
less than one month old,
is iso- to hyper-dense on the unenhanced study.
Older thrombi appear slightly hypodense.
Bland thrombus do not show any evidence of enhancement on the post-contrast scan (Fig.
19).
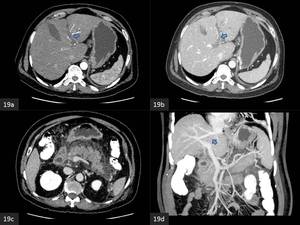
Fig. 19: Bland portal vein thrombus in acute pancreatitis (a) arterial phase and (b) portal venous phase CECT images show a non-enhancing thrombus in the left portal vein. (c) Peripancreatic stranding is observed, the splenic vein is patent. (d) Coronal MIP CECT image shows non-visualisation of the left portal vein.
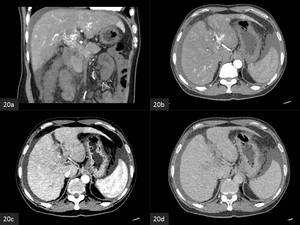
Fig. 20: Malignant portal vein thrombus in hepatocellular carcinoma (not shown) (a) Coronal and (b) axial arterial phase scan shows linear foci of enhancement in the portal vein "thread and streak sign". (c) Portal venous and (d) delayed phase scan shows subtle washout.
MRI shows varying signal in PVT depending on the age of the thrombus with overlap between bland and malignant PVT.
Diffusion weighted MRI shows restriction in both bland and malignant thrombi.
In a recent study,
the ratio of the apparent diffusion coefficient (ADC) of the thrombus to the ADC of the primary tumour in the bland thrombus group was significantly higher in comparison to the thrombus- tumour ADC ratio in the tumour thrombus group (24,25).
Extrahepatic portal venous obstruction (Cavernomatous transformation of the portal vein)
Occusion or severe stenosis of the portal vein results in development of extensive collateral circulation involving paracholecystic,
paracholedochal and pancreaticoduodenal veins (Fig 21) (26).
Portal venous occlusion involving the left gastric (coronary) vein results in preferential collateral venous flow routing around the pancreatic head rather than via the more commonly encountered gastroesophageal variceal circuit.
Cavernous transformation may develop as early as 6 to 20 days following acute thrombosis (27).
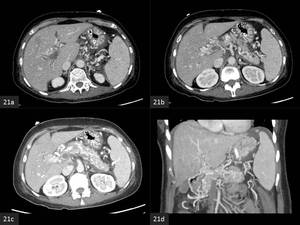
Fig. 21: Extrahepatic portal venous obstruction. (a,b,c) axial CECT images show acute bland portal vein thrombus with multiple paracholedochal collaterals in the hepatoduodenal ligament. (d) Coronal portal venous phase CECT images show thrombosis of the superior mesenteric vein. Extensive pancreatic collaterals are seen because the gastrocolic trunk has been cut-off.
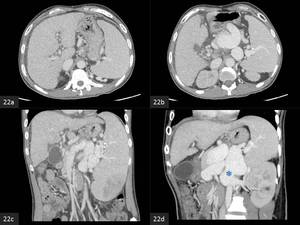
Fig. 22: Fig 22. Extrahepatic portal venous obstruction with portal hypertension. (a,b) axial CECT images show multiple paracholedochal collaterals in the hepatoduodenal ligament. (c,d) Coronal MIP CECT images show ectasia of the splenic vein and a large splenorenal shunt (*).
Extrahepatic portal vein obstruction (EHPVO) is an important cause of noncirrhotic portal hypertension (Fig 22),
especially in the developing world.
Aetiolgy:
- Rare developmental anomalies such as portal vein stenosis and portal vein atresia
- Direct portal vein injury resulting from omphalitis,
umbilical vein catheterization and neonatal peritonitis
- Conditions causing portal vein thrombosis such as severe dehydration and hypercoagulable states.
Besides variceal bleeding,
which is the commonest presentation,
patients may have symptomatic portal biliopathy,
hypersplenism.
The majority of patients are managed by surgical shunting or variceal ablative procedure to prevent rebleeding.
The overall prognosis of patients with EHPVO is good with 75% 10 year survival (26).
Portal biliopathy
Portal biliopathy refers to biliary obstruction associated in the setting of chronic portal vein occlusion with cavernous transformation of the portal vein (28).
Patients with portal biliopathy typically have asymptomatic elevation of liver enzyme levels.
Icterus,
cholangitis,
and choledocholithiasis are rare clinical presentations
Early changes of PB occur with engorgement of the paracholedochal veins of Petren and the epicholedochal venous plexus of Saint (28).
Recent studies have suggested that the mechanism of biliary abnormalities in extrahepatic portal vein obstruction is either extrinsic compression by collaterals or ischemic injury due to venous thrombosis (29)
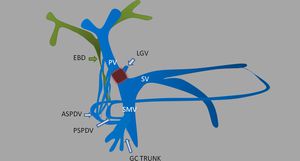
Fig. 23: Pathogenesis of portal biliopathy. The gastrocolic trunk (GCT) gives off the anterosuperior pancreaticoduodenal vein (ASPDV), which communicates with the posterosuperior pancreaticoduodenal vein (PSPDV). When PVT extends into the proximal superior mesenteric (SMV) the left gastric pathway is cut-off and the SMV venous return collateralizes via the anterior and posterior pancreaticoduodenal veins that join the portal vein (PV) close to the hilum. The resultant dilatation of the pancreaticoduodenal veins causes compression and kinking of the extrahepatic bile duct (EBD) superior to the pancreatic head. (SV - splenic vein).
References: Ankur Dev
Venous engorgement of the paracholedochal veins results in scalloping of the ductal lumen,
while engorgement of the smaller epicholedochal veins results in thickenening and enhancement of the bile duct walls on contrast enhanced cross-sectional imaging.
Extrahepatic bile duct kinking and narrowing eventually develops due to extrinsic compression by paracholedochal and pancreaticoduodenal veins (Fig.
23,
24) (30).
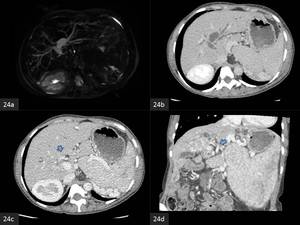
Fig. 24: Portal biliopathy.
(a) Axial MRCP and (b) axial CECT images shows intrahepatic biliary dilatation with obliteration of the hilar confluence.
(c) axial and (d) coronal CECT images show paracholedochal collaterals.
|
Table 3.
Classification of portal biliopathy (Chandra et al.,
reference no 31)
|
|
Type I: Extrahepatic bile duct involvement
|
|
Type II: Intrahepatic bile duct involvement
|
|
Type IIIa: Involvement of extrahepatic and unilateral (right or left) intrahepatic bile duct
|
|
Type III b: Involvement of extrahepatic and bilateral intrahepatic ducts
|
Obliterative portal venopathy
Obliterative portal venopathy (OPV),
also known as idiopathic portal hypertension,
non-cirrhotic portal fibrosis (NCPF),
and hepatoportal sclerosis,
is characterised by intimal fibroelastosis of the medium-sized portal veins.
OPV is a poorly understood condition typically affecting young adults (mean age = 28 years) and is diagnosed on histopathologic findings. Intimal fibroelastosis of the medium-sized portal veins,
invariably associated with cellular adventitial reaction and/or scarring (Fig.
25) (32).
Vasculitis or active thrombosis are not demonstated.
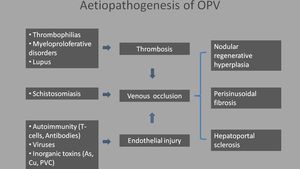
Fig. 25: Aetiopathogenesis of obliterative portal venopathy
References: Ankur Dev
OPV is a major cause of noncirrhotic portal hypertension (NCPH).
NCPH is diagnosed clinically in cases with portal hypertension in the absence of liver cirrhosis and extrahepatic portal vein obstruction (EHPVO) (33).
The majority of patients have preserved liver function.
The imaging characteristics are sparsely described and include thickening of the portal vein wall,
abrupt tapering of second degree intrahepatic portal vein branches (Fig.
26),
thrombus in the intrahepatic portal vein,
and marked dilatation of portal vein and/or splenic vein (34).
Nodular liver contour,
caudate lobe enlargement and segment 4 atrophy are typically absent (34).
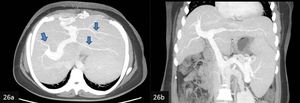
Fig. 26: (a) Axial and (b) coronal MIP CECT show abrupt pruning of the second degree intrahepatic portal vein branches. Dilatation of portal and splenic veins is also seen on the coronal image.
Portal hyperperfusion in small-for-size syndrome
Segmental liver transplantation is a life-saving treatment for patients with end-stage liver disease.
A liver allograft which is significantly below the standard liver weight for a reciepient,
suffers from excessive portal venous inflow with resultant early portal hypertension and increased morbidity and mortality (35).
A small graft is defined as graft-to-recipient weight ratio (GRWR) of less than 0.8% or graft liver volume under 30% of the standard estimated volume (36).
Persisting hyperdynamic portal circulation induces a compensatory decrease in arterial blood flow,
this is termed “buffering response” with impairment of hepatocyte regeneration in the small graft (Fig.
26) (37).

Fig. 27: Pathophysiology of small-for-size syndrome (SFSS)
References: Ankur Dev
SFSS typically occurs within the first week after transplantation (Fig.
28).
SFSS is characterized by cholestasis with elevated serum bilirubin,
coagulopathy,
ascites and gastrointestinal bleeding.
In severe cases SFSS can progress to acidosis,
hypoglycemia,
renal failure and shock (38)
Splenic artery embolization (SAE) is a minimally-invasive technique to treat or prevent SFSS (Fig.
29).
SAE decreases portal graft perfusion and corrects splenic artery stea. Hepatic artery thrombosis is a possible complication of SAE (39).
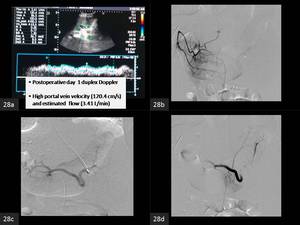
Fig. 28: A 21 year old female underwent adult-to-adult living-related donor liver transplantation for Budd-Chiari syndrome. The Graft-to-recipient weight ratio = 0.7%. The left lobe allograft was slightly small-for-size but was the only life saving option. The surgery was technically successful and without complications.
(a)Post-operative Doppler showed high portal flow (b)The hepatic artery was patent. (C) The splenic artery was relatively dilated. (d)The mid-segment of the splenic artery was embolized with Nester (Cook) Platinum coils with good result.
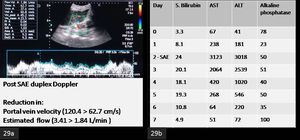
Fig. 29: A 21 year old female underwent adult-to-adult living-related donor liver transplantation for Budd-Chiari syndrome. The Graft-to-recipient weight ratio = 0.7%. The left lobe allograft was slightly small-for-size but was the only life saving option. The surgery was technically successful and without complications.
(a). Post-procedure Doppler showed significant reduction in portal venous flow.
(b). The post- splenic artery embolisation (SAE)liver function tests showed sequential improvement in the ensuing week.
Portal Vein Interventions
Portal vein thrombolysis
The outcome of portal vein thrombosis (PVT) in general is good,
and mortality is primarily associated to the underlying cause and to a much lesser extent to the consequences of PVT and portal hypertension.
The consensus for management of PVT is as follows (40,
41):
- In acute asymptomatic PVT,
anticoagulation and imaging follow-up are the standard of care.
- Anticoagulation is proven to reduce the risk of further thrombotic events after acute PVT by recanalization of the vessel and thus prevents the development of portal hypertension.
- Transcathter thrombolytic therapy via the superior mesenteric artery,
transhepatic and transjugular route has good technical outcome but is reported to carry a high rate of major complications.
Thus,
most centres tend to choose a more conservative therapeutic strategy.
- Thrombolytic therapy is generally reserved for patients with severe disease such as bowel ischaemia - a dreaded complication of PVT.
Both surgical thrombectomy and transcatheter throbolysis have been described (Fig.
30) (42).
- Evidence on the efficacy of anticoagulation and thrombolytic therapy in patients with chronic PVT with or without cirrhosis is lacking and usually not recommended.
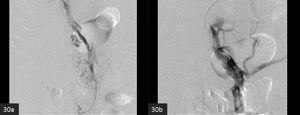
Fig. 30: A 57 year-old-male with portal and mesenteric venous thrombosis developed bowel ischaemia. The patient was high risk for surgery due to Cardiovascular co-morbidity.
(a) Initial Digital Signature Angiogram (DSA)showed thrombosis of the portal vein. (b) Following transcatheter thrombolysis and balloon angioplasty, portal vein patency was reestablished.
Portal vein embolisation
Transhepatic percutaneous portal vein embolization (PVE) is performed prior to major hepatic resection to induce selective hypertrophy of the nondiseased portion of the liver in patients with either primary or secondary hepatobiliary malignancy with small estimated future liver remnants (FLR) (43).
The minimum liver volume required for postresection hepatic function is higher in diseased livers.
A FLR/ total estimated liver volume (TELV) ratio of at least 25% is recommended in patients with normal livers.
A ratio of at least 40% is requisite in patients with chronic liver disease (44).
When FLR/TELV ratios are below these levels,
PVE may be performed in order to enhance the FLR volume (Fig.
31).
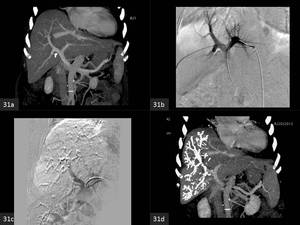
Fig. 31: A 64 year-old-female with cholangiocarcinoma was advised right tri-segmentectomy, however the FLR volume was unsatisfactory. Right portal vein embolisation was performed in order to enhance the FLR volume.
(a) Coronal MIP CECT and (b) DSA show patent portal venous system. (c)DSA and (d) MIP CECT show residual lipiodol-glue cast in the right portal vein.
Liver regeneration typically peaks within the first 2 weeks after PVE. Noncirrhotic livers regenerate at the rate of: 12–21 cm3/day at 2 weeks after PVE,
11 cm3/day at 4 weeks,
and 6 cm3/day at 32 days.
Livers in patients with cirrhosis regenerate more slowly (45).
Transjugular Intra-hepatic Portosystemic Shunt
The transjugular intrahepatic portosystemic shunt (TIPS) is an endovascular procedure indicated for treating sequelae of portal hypertension including variceal bleeding and refractory ascites (Fig.
32,33).
TIPS reduces the portosystemic pressure gradient,
decompressing varices and eliminating ascites (46,
47).
The major disadvantages of the TIPS procedure are deterioration of hepatic function caused by diversion of portal venous blood flow,
hepatic encephalopathy due to increased bioavailability of gut-derived toxins and dysfunction of the TIPS shunt (48).
A post-TIPS portosystemic pressure gradient below12 mm Hg in patients is adequate in preventing a rebleeding episode.
For refractory ascites the recommended portosystemic pressure gradient value is 8 mm Hg.
Portosystemic pressure gradients below 5 mm Hg are associated with liver failure and severe hepatic encephalopathy often necessitating a shunt occlusion or reduction (49).
The most crucial and challenging part of the TIPS procedure is obtaining suitable access to the portal vein.
A right portal vein trunk is usually present and in up to 90% of patients is located between the 10th and 12th ribs,
within 0.5-1.5 vertebral widths to the right lateral margin of the vertebrae (50).
Ultrasound provides information about variant anatomy as well as positional and directional information, significantly improving needle pass efficiency (51).
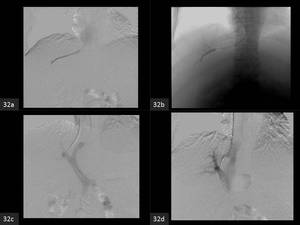
Fig. 32: The TIPS procedure
(a). The right hepatic vein is cannulated with a multipurpose cathter over a hydrophilic guidewire via the right internal jugular vein.
(b). The hepatic venous pressure gradient is estimated with a Swan-Ganz catheter.
(c).
Following portal vein access using a Rösch Uchida portal access needle, the diamond-tip needle is removed and the outer catheter is withdrawn in 1- to 2-mm increments. During needle withdrawal, brisk blood return into the aspirating syringe signals continuity of the needle tip with a vascular structure (the portal vein). A conventional polytetrafluoroethylene (PTFE)-coated guidewire, such as a Bentson wire is manipulated into the portal vein, directing it toward the portal bifurcation, the guidewire is followed with the catheter of the Rösch Uchida system
(d). Following measurement of portosystemic pressures and digital subtraction portography, The length of the parenchymal tract is measured from the junction with the portal vein to the hepatic vein–inferior vena cava (IVC) junction.
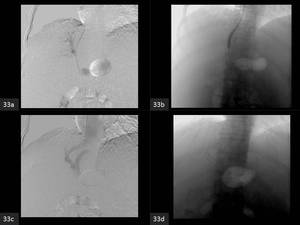
Fig. 33: The TIPS procedure.
(a) Following tract dilatation. An endograft of appropriate length is placed in the tract.
(b) The endograft is molded using the same balloon that was used for initial tract dilation.
(c) Completion portogram shows a patent endograft.
(d) The endograft in-situ on a post-procedure radiograph.
Transjugular intrahepatic portosystemic shunt (TIPS) endografts are reported to have 1-year primary and secondary patency rates of 80% and 100% (52)


































