CLINICAL PERSPECTIVES
UEDVT is an increasingly important clinical entity with potential for considerable morbidity,
which incidence is increasing paralleling the increased use of central venous catheters. UEDVT has been reported in up to one fourth of patients with these catheters.
Pulmonary embolism is present in up to one third of patients affected.
For these reasons,
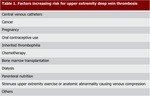
it is imperative that radiologists recognize UEDVT risk factors (Table 1) understanding it clinical presentation.
UEDVT is classified as primary or secondary on the basis of pathogenesis.
Primary UEDVT
Primary UEDVT is a rare disorder that refers either to Paget-Schroetter Syndrome,
thoracic outlet obstruction or idiopathic UEDVT.
- Paget-Schroetter Syndrome:
It refers to development of spontaneous UEDVT,
usually in dominant arm of an otherwise healthy and young patient,
after strenuous activity. The heavy exertion causes microtrauma to the vessel intima and leads to activation of the coagulation cascade.
Significant thrombosis may occur with repeated insults to the vein wall,
especially if mechanical compression of the vessel is also present.
- Thoracic outlet obstruction:
It refers to compression of the neurovascular bundle (brachial plexus,
subclavian artery and subclavian vein) as it exits the thoracic inlet.
Compression of the subclavian vein typically develops in young athletes with hypertrophied muscles who do heavy lifting or completely abduct their arms.
Cervical ribs,
long transverse processes of the cervical spine,
musculofascial bands,
and clavicular or first rib anomalies are sometimes found in these patients.
These patients have no known trigger or obvious underlying disease.
However UEDVT may be sometimes associated with occult cancer or with hypercoagulable states.
Screening for coagulation disorders should be performed in these patients,
since the yield of these tests is highest for patients presenting with idiopathic UEDVT.
For instance,
elevated antiphospholipid antibodies in the presence of UEDVT establish the diagnosis of the antiphospholipid antibody syndrome.
Secondary UEDVT
Secondary UEDVT accounts for most cases of UEDVT and develops in patients with central venous catheters,
pacemakers or cancer.
- Catheter-related thrombosis:
It is caused by several factors.
The vessel wall may be damaged during catheter insertion or during infusion of medication.
Also,
the catheter may impede blood flow through the vein and cause areas of stasis. Patients with incorrectly placed catheters are more likely to develop deep vein thrombosis.
Blood flow is most rapid in the SVC,
which may sufficiently dilute the infusate and reduce the risk of thrombophlebitis.
Therefore,
catheter tips should be positioned in the lower third of this vessel or at the junction of the superior vena cava and right atrium.
Presenting symptoms and signs
Axillary or subclavian vein thrombosis may occasionally be completely asymptomatic.
More often,
patients complain of vague shoulder or neck discomfort and arm edema.
When thrombosis causes obstruction of the superior vena cava,
the patient may complain of arm and facial edema,
head fullness,
blurred vision,
vertigo or dyspnea.
Patients with thoracic outlet obstruction may have pain that radiates into the fourth and fifth digits via the medial arm and forearm,
attributable to injury of the brachial plexus.
Symptoms may be position dependent and worsen with hyperabduction of the shoulder or lifting.
If thoracic outlet syndrome is suspected,
the examiner should palpate the supraclavicular fossa for brachial plexus tenderness,
inspect the hand and arm for atrophy,
and perform provocative tests,
such as Adson’s and Wright’s maneuvers.
Physical examination may reveal multiple signs attributable to thrombosis. However,
the signs and symptoms of UEDVT are non-specific and may occur in patients with lymphedema,
neoplastic compression of the blood vessels,
muscle injury,
or superficial vein thrombosis.
Fewer than half of these symptomatic patients will have imaging evidence of an UEDVT.
Therefore,
it is important to confirm or exclude the diagnosis with an imaging test.
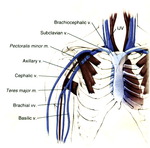
UPPER EXTREMITY VENOUS ANATOMY
The venous system of the UE is divided into deep and superficial vessels.
The distal deep veins are paired and accompany the arteries: radial and ulnar.
More proximally the venous system continues as the brachial,
axillary,
subclavian,
and brachiocephalic veins (Fig. 1).
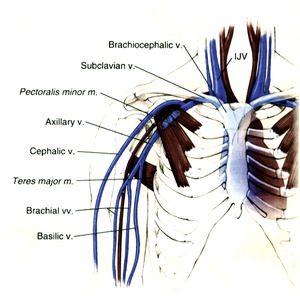
Fig. 1: Schematic drawing demonstrating venous anatomy of the upper extremity.
References: Rumack CM, Wilson SR, Charboneau JW, Levine D. Diagnostic Ultrasound. Philadelphia, PA: Elsevier Mosby, 2011: 1035
The proximal extent of vein pairing is variable.
Althoug the majority of patients have single brachial veins,
pairing can be seen involving the axillary veins.
Communication among the veins of the deep system is also variable.
Two main veins constitute the superficial venous system of upper extremity: the cephalic vein,
along the lateral aspect of the arm,
and the basilic vein along the medial side.
These veins communicate at the antecubital fossa via the median antecubital vein.
The basilic vein pierces the deep fascia on the medial aspect of the mid-upper arm to merge with the brachial vein,
and this combined channel becomes the axillary vein as it enters the axilla.
The cephalic vein passes more cephalad along the lateral aspect of the biceps.
At the level of pectoralis major it courses medially and deeply to pierce the clavipectoral fascia below the clavicle,
and joins the axillary vein.
Other tributaries from the region of the shoulder joint and the lateral chest wall drain into the axillary vain.
As it crosses the first rib,
the axillary vein becomes the subclavian vein.
The main tributary of the subclavian vein is the external jugular vein.
The subclavian vein unites with the internal jugular vein behind the medial aspect of the clavicle to form the brachiocephalic vein,
also known as the innominate vein.
The right and left brachiocephalic veins merge to form the superior vena cava,
which subsequently enters the right atrium.
Perforating veins form important pathways of collateralization in the presence of partial thrombosis.
In the absence of thrombus they are typically too small to see,
but become more pronounced when they are recruited to divert flow around a clot.
Valves are present within the veins of the UE.
As one moves peripherally the location of the first valve is quite variable,
but typically is encountered in the proximal brachial vein.
TECHNICAL PROCEDURES
The ultrasound examination of the UE veins for diagnose DVT relies on similar principles to examination of lower extremity venous examination: gray scale compression,
color Doppler and spectral Doppler.
The study is typically performed with the patient in the supine position and the arm in a neutral anatomic position.
The arm should be partially abducted to examine the axillary vein.
A linear transducer is used to perform the study.
Seven to 12 MHz is a good frequency range to begin the study,
particularly in large or edematous arms.
A higher-frequency transducer may be used for superficial veins or in thinner arms.
On the other hand,
a lower-frequency tranducer should be used in obese and extreme edematous arms.
1.
Ultrasound and Compression
The lumen of a normal vein is anechoic.
Therefore,
thrombus can be directly visualized as an echogenic material affixed to the vessel wall.
Fresh thrombus may be extremely hypoecoic difficulting it visualization and,
therefore,
is essential to perform venous compression in a transverse plane to rulle out the presence of UEDVT.
Normal UE veins are easily compressible and transducer pressure will obliterate their lumens. Compression should be light because fresh clots are soft and firm pressure may give a false impression of patency.
Since the compression techniques cannot be used in portions of subclavian vein and in the inominate vein because of the overlying structures,
there is a greater reliance on color-Doppler and spectral-Doppler assessment to rulle out an obstruction.
2.
Ultrasound Color-Doppler
Color Doppler is a useful adjunct to confirm venous patency.
The entire lumen should fill with color. As a result of right atrial contraction (a-wave),
it is a normal finding to observe pushes back on venous return in the larger central veins,
resulting in a temporary reversal of flow. The color Doppler signal will fluctuates in direction.
It is also important also to ensure that the Doppler settings are tuned for the slower velocities found in veins. With the pulse repetition frequency adjusted to a higher level,
the wall filter may suppress perception of slower laminar flow along the wall,
appearance that can be confusing,
mimiking a clot adherent to the wall.
On the other hand,
in the larger veins with the color-Doppler pulse repetition frequency set relatively low,
with a brisk augment,
aliasing may occur.
Both findings should be recognized as Doppler artifacts.
3.
Ultrasound Spectral-Doppler
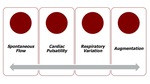
The spectral-Doppler flow profile can be used to great diagnostic advantage.
A group of parameters may be analysed:
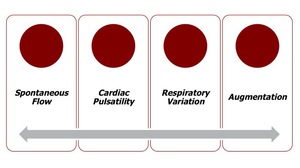
Fig. 2: Schematic image showing the spectral-Doppler findings.
- Spontaneous flow,
respiratory variation and cardiac periodicity:
If there is no thrombus occluding the vein,
the examiner will detect color spontaneously saturating all lumen of the vessel.
Because the veins of the UE are in close proximity to the heart,
it is normal to see a cardiac pulsatility in the spectral analysis.
Spectral analysis of the caudal internal jugular vein and the medial subclavian vein demonstrate central venous transmitted cardiac pulsatility with a,
c,
v peaks,
and x and y descents. The presence of this pulsatility proves the patency of the conduit between the point of Doppler interrogation and the right atrium.
In addition,
the examiner should identify the respiratory phasicity superimposed on cardiac pulsatility. This can be differentiated from cardiac pulsatility by observing directly a patient’s respiratory cycles. In contrast to the lower extremity veins,
the velocity of blood flow in the veins of the upper extremity increases during inspiration due to negative intrathoracic pressure and increased venous return toward the heart.
Changes in these parameters may indicate central vein stenosis or occlusion.
Spectral findings of the medial and lateral aspect of the subclavian vein should be also compared because a change in the tracings may be due to stenosis in its midportion.
Normal venous flow is slow.
Its perception on Doppler can be enhanced applying manual compression,
distally to the point of assessment.
In a normal venous system there will be a rapid rise and fall in the frequency shift,
whereas if there is a thrombosed venous segment it will resist flow with damping or absence of the augmentation response.
The squeeze should be rapid and not excessive,
as there is a small potential risk of dislodging a fresh friable thrombus.
With slow flow or poorly distended veins,
one can also enhance the perception of the color signal by asking the patient to perform a Valsalva maneuver.
The resultant increased intrathoracic pressure resists venous return and allows more blood to pool peripherally.
Finally,
respiratory phasicity and cardiac pulsatility can also be modified by asking the patients to breathe deeply and then hold their breath.
CRITERIA OF UEDVT
Criteria for diagnose UEDVT are similar to those used in the lower extremity.
One should diferentiate between a normal examination,
a non-obstructive or partial UEDVT and obstructive UEDVT,
as listened above:

Table 4: Diagnostic criteria of upper extremity deep vein thrombosis. Normal findings.
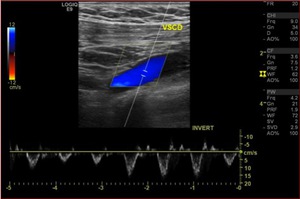
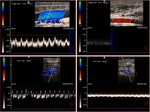
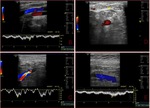
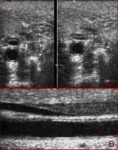
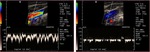
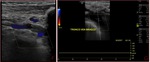
Fig. 3: Triplex sonogram of the right subclavian vein. In addition to the rapid phasic changes in cardiac pulsatility from atrial contractions, there is a further variation in amplitude due to normal respiratory variation.

Table 5: Diagnostic criteria of upper extremity deep vein thrombosis. Non obstructive thrombosis.
The thrombus may be directly visualized in the vessel lumen as an echogenic material affixed to the vessel wall.
Fresh clots,
however,
will be extremely hypoechogenic difficulting their visualization.
Whenever possible,
compression will help to rulle out a thrombus.
If there is a non obstructive clot in the lumen,
the examiner will only observe a partial compression.
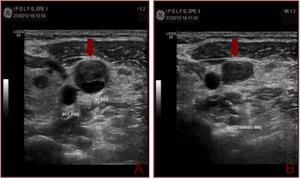
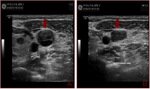
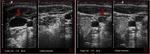
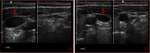
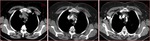
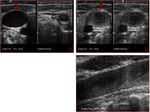
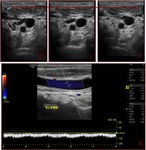
Fig. 4: Gray-scale sonogram of the left internal jugular vein (arrows) in the transverse view shows some echogenic material within it (a). Probe pressure is being exerted over the vein (b) and the thrombus is preventing the compression of the vein. This is the key to positively identifying the presence of this non-occlusive thrombus within the vein.
The color Doppler will confirm the partial patency of the lumen. Cardiac periodicy and fasic respiratory variation can be present or damped according to the volume of the thrombus.
Small eccentric clots may not influence these parameters. The augmentation response will be also damped.
After the release of Valsalva,
absence of a surge in antegrade flow indicates a central clot.
Comparison with flow dynamics on the contralateral side may be helpful in localizing the level of the clot.

Table 6: Diagnostic criteria of upper extremity deep vein thrombosis. Obstructive thrombosis.
An occlusive thrombus can be visible filling the vein lumen with nonmobile echogenic material. Although fresh thrombus is relatively hypoechoic,
it becomes increasingly echogenic as it matures. Fresh thrombus has a tendency to expand the vein and make it look rounder and fuller than a normal vessel,
and is not particularly adherent to the vein wall.
In addition,
lack of compressibility of the deep veins of the arm and neck and absence of flow on color or power Doppler are diagnostic of obstructive thrombosis.
Patent segments below the thrombus will show some slow antegrade flow,
particularly if collateral channels are adequate,
without any respiratory variation or cardiac pulsatility and collateral vessels may show reverse flow.
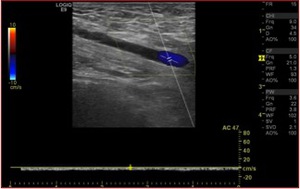
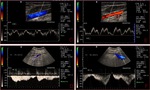
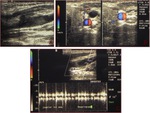
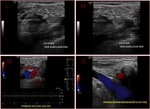
Fig. 5: Triplex sonogram in the longitudinal view shows an occlusive thrombus in the left axillary vein. Patent segment below the clot demonstrates some slow anterograde flow without respiratory variation or cardiac pulsatility.
The larger,
more proximal veins,
such as the axillary and subclavian,
cannot be compressed,
due to their location,
diagnosis of thrombosis in these vessels will therefore depend on careful assessment using Doppler. Loss of respiratory phasicity or cardiac periodicity indicates proximal occlusion.
If the subclavian and jugular veins are markedly distended but Doppler reveals a flattened flow profile,
an obstruction to the antegrade flow of blood and the retrograde propagation of the waveform infers the presence of a central clot:
- If these findings are bilateral,
the level of obstruction is at the superior vena cava;
- If only one side manifests these changes,
the obstructing clot is inferred to be at the level of that brachiocephalic vein.
Particularly,
patients with catheter-related thrombosis,
who demonstrate clots that propagate along the length of the catheter to the catheter tip,
positionated within the superior vena cava,
brachiocephalic or more distal veins, have a potential risk to manifest a venous return compromised.
Doppler findings will reveal not only direct signs of thrombus near de portion of the catheter visualized in the subclavian vein,
but also will predict the presence of a clot in non accessible vessels.
POTENCIAL PITFALLS
Rouleaux
Blood flow is anechoic because individual red blood cells are too small to reflect the incoming sound wave.
However,
in certain conditions as infection,
diabetes mellitus or cancer,
red blood cells may stick to each other,
a finding that is named rouleaux.
These aggregates are large enough to interact with the insonating beam,
manifesting as echoes in the bloodstream,
and are more likely to occur in areas of slow flow,
especially in the sinus behind the cusps of valves.
If compression easily dislodges these Rouleaux aggregates,
presence of a clot is excluded.
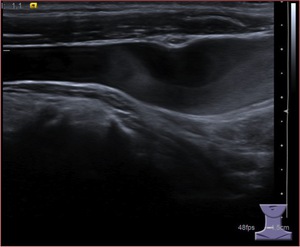
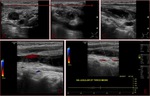
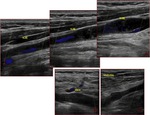
Fig. 6: Gray-scale sonogram in the longitudinal view shows an area of slow flow in the right internal jugular vein mimicking deep venous thrombosis. The internal echoes were noted to swirl on real time, suggesting slow flow rather than DVT. These echoes merely represent swirling of aggregates of red blood cells large enough to interact with the insonating beam (rouleaux formation), in the setting of slow flow. Compression will confirm the complete coaptation of the vessel wall.
Arm abduction
Caution should be exercised in interpreting the distal subclavian vein,
which may appear falsely narrowed as it crosses between the clavicle and the first rib at the thoracic inlet,
due to complete abduction of the upper extremity during examination.
Limited acoustic window
Limited acoustic window due to bandages used to secure the catheters,
radiation-induced changes on the chest wall or the presence of indwelling catheters,
can make assessment of vessel compressibility challenging.
Large venous collaterals
In chronic obstruction,
large venous collaterals often coexist and can be misinterpreted as representing patent normal vessels.
CHRONIC CHANGES AFTER UEDVT
Valves
Valve leaflets should be delicate,
moving briskly with fluctuations in flow direction throughout the venous waveform,
and the sinus of the valve leaflets should be relatively free of echoes. If valve cusps appear rigid or fixed,
this usually represents the sequela of prior UEDVT.
Walls
The walls of a normal vein are smooth and non obstructive.
Following recanalization after DVT they become irregular,
thickened,
echogenic and rarely calcified.
Venous collaterals
Their visualization suggests chronic thrombosis. When the normal venous channels are occluded,
blood may be seen in collateral veins.
In the acute stage intramuscular channels will not have developed significantly.
Increased velocity and flow may be seen.
Over a period of time the intramuscular venous channels expand and become apparent on color Doppler.
Collateral perforating veins themselves may serve as a pathway for the propagation of a clot from the superficial to the deep system.
This aspect is important in the diagnosis of thrombophlebitis.