INTRODUCTION
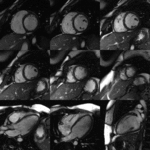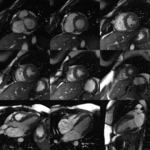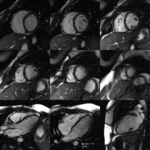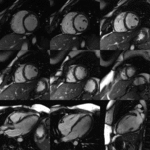
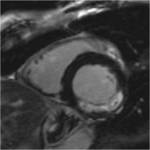
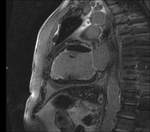
The CMR is an imaging method that permits a non invasive assessment of the heart and it has been established as the gold standard for the evaluation of cardiac anatomy and function Fig. 2,
as well as for the myocardial viability1.
Late gadolinium enhancement (LGE) technique has been the standard of reference for detecting focal myocardial fibrosis in clinical practice.
LGE can visualize the myocardial infarction (MI) or the focal fibrosis in non ischaemic cardiomyopathies by using contrast agents that distribute through the extracellular space2 (either because the membranes are not intact such as in acute MI Fig. 3 or myocarditis,
or because there is replacement fibrosis such as in chronic infarction Fig. 4 or cardiomyopathies Fig. 5).
MI and focal fibrotic processes of non-ischaemic origin appear bright on LGE images because gadolinium shortens T1.
While LGE technique is clinically useful3 (it provides diagnostic and prognostic information),
reliance on relative signal intensity changes and nulling of “normal appearing” myocardium make it difficult to identify subtle abnormalities such as diffuse interstitial fibrosis4,
because there is no normal tissue from which abnormal tissue could be delineated.
A unique feature of CMR is its ability to use protons relaxation times,
such as T1,
T2 and T2* to characterize myocardial tissue4.
These techniques will solve the problem of detection of processes that affect diffusely the myocardium.
These relaxation times can be quantified using recently created mapping sequences6-7.
Basically it consists in showing the tissues relaxation times as color map where the relaxation time T1 Fig. 6 ,
T2 Fig. 7 or T2* Fig. 8 is encoded in the intensity of each pixel.
The native,
non-contrast T1 lengthens with interstitial expansion caused by fibrosis,
oedema,
infarction and amyloid infiltration.
This is believed to be due to the increase in free fluid associated with these pathophysiological states.
As fat and iron are known to possess a very low T1,
it is shortened in these diseases.
T2 lengthens with oedema (acute MI,
myocarditis,
heart transplant rejection).
T2* lengthens with iron overload (haemochromatosis,
thalassaemia).
MYOCARDIAL FIBROSIS AND INTERSTITIAL SPACE
Diffuse myocardial fibrosis is a covert process that occurs as a part of normal aging8 but it is accelerated in diseases such as hypertension,
aortic stenosis,
diabetes mellitus,
cardiomyopathies,
and in the myocardium remote from infarction9-12.
Diffuse myocardial fibrosis implies an increment in the content of collagen and means an increase in the myocardial extracellular volume (ECV)13.
Diffuse myocardial fibrosis is a final end point in most cardiac diseases.
The pathophysiological mechanisms that lead to this fibrosis are various,
with some being acute,
as in MI,
and others being progressive and potentially reversible,
as in hypertensive cardiomyopathy.
Myocardial fibrosis is associated with worsening ventricular systolic function,
abnormal cardiac remodeling,
and increased ventricular stiffness14.
Moreover,
myocardial fibrosis plays an important role in the development of the second major factor contributing to morbidity and mortality of patients with chronic cardiovascular diseases and arrhythmia because it can cause significant alteration of the electric properties of the myocardium15.
Furthermore,
it has been shown that myocardial fibrosis is an independent predictor of major adverse cardiac events (heart failure,
arrhythmia and death)16.
In the therapeutic guidelines for heart failure due to various cardiomyopathies,
there are no specific therapeutic strategies based on the tissue composition of the myocardial wall,
either in the early or more advanced stages of disease.
This lack of specific treatment might result in inappropriate therapies,
which can lead to increased morbidity and additional financial burden to health care services17.
Lack of personalized treatment is also secondary to the absence of accurate clinical tools to precisely phenotype patients with heart disease13.
In this respect the quantitative T1 maps are a promising method to detect diffuse myocardial fibrosis.
1. ETIOPHYSIOPATOLOGY OF MYOCARDIAL FIBROSIS
The myocardial interstitial or extracellular space exists as a complex and dynamic environment,
vital for normal cardiac structure and function.
The interstitium is a complex three-dimensional space into which cardiac cells,
blood vessels and nerves are embedded.
In the normal human heart,
the extracellular matrix is predominantly made up of collagen scaffolding of types I (80%) and III (11%)18-19 Fig. 9 .
One of the distinctive factors of pathology is the interstitial space expansion normally through the development of fibrosis.
Myocardial fibrosis,
defined by a significant increase in the collagen volume fraction of myocardial tissue (normal value: 25%),
is always present in end-stage heart failure.
The distribution of myocardial fibrosis,
however,
varies according to the underlying pathology.
Subtypes of myocardial fibrosis
Different types of myocardial fibrosis have been reported according to the cardiomyopathic process Fig. 10.
1.
Reactive interstitial fibrosis
The first type of fibrosis is interstitial reactive fibrosis with a diffuse distribution within the interstitium.
This type of fibrosis has a progressive onset and follows the increase in collagen synthesis by myofibroblasts under the influence of different stimuli.
It has mostly been described in hypertension9 and diabetes mellitus10,
but it is also present in the aging heart8,
in idiopathic dilated cardiomyopathy20,
and in left ventricular pressure-overload and volume-overload states induced by chronic aortic valve regurgitation and stenosis21.
It has also been reported in the remote non-infarcted myocardium after infarction12.
Reactive interstitial fibrosis precedes irreversible replacement fibrosis22.
Reactive interstitial fibrosis is reversible under specific therapy23.
Therefore,
there is some clinical interest in its assessment for the management of patients with hypertension,
diabetes,
primary dilated cardiomyopathy,
and valvular disease.
2.
Infiltrative interstitial fibrosis
This subtype of fibrosis is induced by progressive deposit of insoluble proteins (amyloidosis)24 or glycosphingolipids (Anderson-Fabry disease)25 in the cardiac interstitium Fig. 11.
Their pathophysiology follows similar patterns,
and the early detection of cardiac involvement is of critical importance to therapeutic management.
3.
Replacement fibrosis
This replacement or scarring fibrosis correspond to the replacement of myocytes after cell damage or necrosis by plexiform fibrosis,
mainly type I collagen26.
Replacement fibrosis appears as soon as the myocyte integrity is affected.
It can have a localized distribution (ischaemic cardiomyopathy,
myocarditis,
hypertrophic cardiomyopathy,
sarcoidosis) or a diffuse distribution (chronic renal insufficiency,
toxic cardiomyopathies,
inflammatory disease) according to the underlying etiology.
2. DETECTION OF MYOCARDIAL FIBROSIS
Currently,
the only method to quantify diffuse fibrosis is invasive biopsy,
which carries significant morbidity,
is prone to sampling error and fibrotic involvement of the whole left ventricle cannot be determined27.
Blood biomarkers for fibrosis assessment are also known to have complex confounding factors.
Therefore,
non-invasive imaging assessment of the interstitial space has de potential to provide a CMR biomarker for myocardial fibrosis.
Preliminary studies suggest that these techniques are reproducible and we can rely more on them than on the biopsy,
because the biopsy represents less than a thousandth of the whole myocardium volume.
Furthermore,
these techniques can potentially quantify de fibrosis of the whole heart which really reflects the global myocardial fibrosis burden.
These new biological parameters have the ability to detect early disease,
guide therapy and predict outcomes.28
T1 mapping measures myocardial longitudinal magnetic relaxation time,
with high spatial resolution within a single breath hold.6 Compared with LGE images Fig. 12,
T1-mapping CMR techniques allow us to eliminate the influences of windowing and variations in signal enhancement by directly measuring the underlying T1 relaxation times Fig. 13.
Therefore,
it allows signal quantification (in milliseconds) on a standardized scale of each myocardial vóxel to characterize myocardial tissue.