Radiographic features
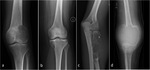
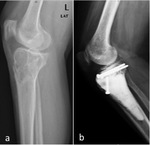
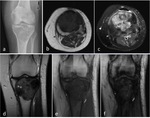
GCT almost exclusively occurs in patients with closed physes and typically presents as an eccentric meta-epiphyseal lytic lesion abutting the articular surface,
with a narrow non-sclerotic zone of transition (Fig.1a).
Rarely it may present sclerotic margins (Fig.1b) or a wide zone of transition (10%–20% of cases).
[1]
Thought it is usually eccentrically located,
large lesions and lesions located in small caliber bones can appear central in location.
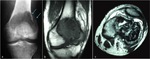
GCT may show expansile bone remodeling (47%–60%) and have a multiloculated appearance due to pseudotrabeculation (33%–57%),
derived from uneven bone destruction in three dimensions.
Pronounced trabeculation may create a “soap-bubble” appearance (Fig 1c).
[1,6]
GCT may demonstrate aggressive features,
with cortical bone thinning,
cortical penetration and soft tissue extension (Fig.
1d),
features reported to be more common in small-caliber bones such as the fibula or ulna.
Although invasion of the ligaments is possible,
articular cartilage is usually preserved and intra-articular extension of the tumor is extremely rare.
Characteristic radiographic findings of GCT:
1) Patients with closed physes
2) Extension to the subchondral bone
3) Well defined lytic lesion with non-sclerotic margin
4) Eccentric location
Uncommon radiographic findings
GCT is less likely to demonstrate the above-described classic appearance when it is located in flat bones (ilium,
sacrum,
spine,
ribs,
calvaria and calcaneus) or apophyses (namely the greater trochanter of the femur).
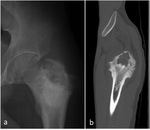
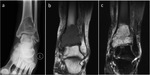
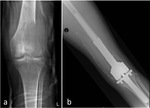
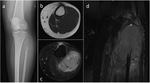
Other uncommon features may include periosteal reaction,
reported in 10%–30% of cases (Fig.2),
fluid-fluid levels due to secondary ABC,
described in 14% of cases,
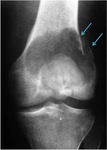
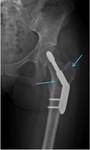
and pathologic fracture,
occurring in 11-37% of patients (Fig.
3).
[1]
Imaging GCT with CT and MR
Although radiographic features usually suggest the diagnosis,
CT and MR imaging are required for accurate tumor staging.
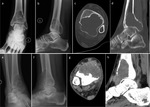
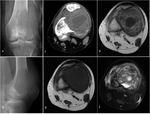
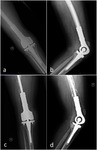
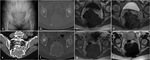
CT allows better detection of cortical thinning,
periosteal reaction,
pathologic fractures,
and expansile remodeling of the bone compared with radiography (Fig.
4).
It also confirms the absence of calcified matrix in GCT,
identifies callus formation after pathologic fracture and demonstrates a calcified peripheral rim after medical therapy with Denosumab.
Solid portions of the tumor show attenuation similar to that of the muscle.
Identifying fluid attenuation areas alerts to the presence of secondary ABC.
Although less accurate in detection of calcified matrix,
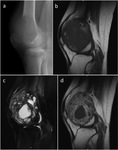
MR imaging is paramount to define soft tissue extension of the tumor due to it´s superb contrast resolution (Fig.
5).
GCT usually appears as a well-defined lesion,
with intermediate or decreased signal intensity on T1-weighted images (T1WI) and increased signal intensity on T2-weighted images (T2WI) (Fig.6).
GCT may show a low signal intensity margin due to osseous sclerosis or pseudocapsule.
Although hyperintensity is the most frequent finding on T2 WI,
GCT may also show low signal intensity due to the presence of hemosiderin or high collagen content (Fig.7) [7].
After intravenous administration of gadolinium the tumor shows avid enhancement (Fig.
5e,
8d).
MR is also superior to radiography and CT in demonstrating the presence of areas of ABC,
which translate into cystic areas with fluid-fluid levels,
markedly hyperintense on T2WI and hypo or hyperintense on T1WI (Fig.
8).
This appearance may mimic a primary ABC,
however,
unlike the primary counterpart,
secondary ABC will show solid components enhancing diffusely after contrast administration (Fig.8).
By identifying solid portions in the tumor,
MR imaging is not only essential to differentiate these two entities,
but is also vital to direct the biopsy to the solid parts.
Treatment
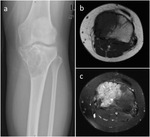
Surgical treatment with curettage is the typical treatment for GCT,
with recurrence rates of 15%–45%.
With the use of a high-speed burr and allograft or bone cement (polymethylmethacrylate) placement (Fig.9),
recurrence rates after intralesional surgery dropped to 12%–14%.
Aggressive GCT usually require wide excision and reconstruction with endoprosthesis (Fig.10),
with reported recurrence rate of 6%.
[2,8,9]
Patients submitted to curettage and cement placement should be monitored carefully with serial radiographs,
always comparing the follow-up images with the baseline postoperative images.
Local recurrences usually occur between 12 and 18 months after surgery (being rare after 3 years) and should be suspected when new lucencies appear at the cement-bone interfaces (Fig.
11).
[4] CT,
and particularly MR,
may be diagnostic in equivocal cases,
presenting signal alterations similar to that of the primary tumor.
Postoperative complications of endoprosthesis may include fracture or dislocation of the implant,
instability,
infection and aseptic loosening (Fig.12).
Radiotherapy was previously used to treat GCT in difficult locations,
such as the spine and sacrum,
however long-term risk of malignant transformation of GCT and post-radiation sarcoma has limited its routine use.
Presently,
patients with aggressive or recurrent tumors,
with tumors in difficult locations and with multiple lesions,
as well as patients who are poor surgical candidates,
may be treated with a monoclonal antibody – Denosumab.
Denosumab inhibits the receptor activator of nuclear factor κB ligand (RANKL),
which is an essential mediator of osteoclast formation,
function,
and survival,
present in osteoclast-like giant cells and their precursors,
as well as some of the mononuclear stromal cells.
By inhibiting osteoclast-mediated bone destruction,
Denosumab induces tumor necrosis,
sclerosis and reconstitution of the cortical bone (Fig.
13).
[9,10]
Review of 20 cases of GCT
We retrospectively analyzed 20 cases of patients with histologically confirmed GCT that were assessed,
staged with MR,
and treated at Hospitais da Universidade de Coimbra (Centro Hospitalar e Universitário de Coimbra,
Coimbra,
Portugal) from 2003 to 2013.
Our series demonstrated a slight female preponderance (55%),
in agreement with results from other authors [11].
65% of GCT occurred between 20 and 50 years of age,
with 45% occurring in the third and forth decades (Fig.
14).
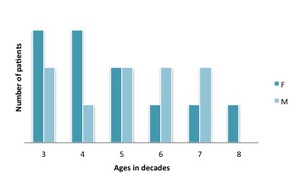
Fig. 14: Age (per decades of life) and gender of patients included in our series.
References: Radiology, Centro Hospitalar e Universitário de Coimbra, Hospitais da Universidade de Coimbra - Coimbra, Portugal
Tumors primarily originated near the knee (55%) (Fig.
15),
with 35% located on the proximal tibia (Fig.
16),
15% on the distal femur and 5% on the proximal fibula.
Other locations included the distal tibia (15%),
ilium and sacrum (15%) and radius (5%).
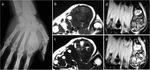
One case occurred in the second metacarpal bone of the hand (Fig.
17).
One case occurred within an apophysis,
in the greater trochanter of the femur (Fig.2).
We report no cases of multifocal GCT.

Fig. 15: Different locations of GCT in our series.
References: Radiology, Centro Hospitalar e Universitário de Coimbra, Hospitais da Universidade de Coimbra - Coimbra, Portugal
Most GCT located on tubular bones had an eccentric position (76%),
with exception to GCT of small caliber bones and extremely large lesions,
which had a central location (23%).
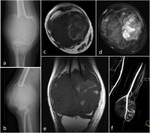
Of the three pelvic GCT of our series,
one was confined to the ilium,
another to the sacrum (Fig.22),
and one extended through the sacroiliac joint affecting both the ilium and sacrum (Fig.13).
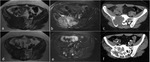
To assess the MR signal of GCT we classified tumor intensity as follows (Fig.18):
Hypointense
Isointense
Hyperintense
Hypointense
Hyperintense
Mixed signal intensity
- With cystic areas
- Without cystic areas
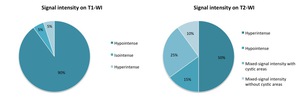
Fig. 18: Signal intensity characteristics of GCT in our series on T1-WI and T2-WI.
References: Radiology, Centro Hospitalar e Universitário de Coimbra, Hospitais da Universidade de Coimbra - Coimbra, Portugal
Most GCT were hypointense on T1WI (90%),
one being isointense,
and one being slightly hyperintense comparing to skeletal muscle.
Three tumors (15%) showed focal areas of hyperintensity due to hemorrhage (Fig.
19).
Signal intensity on T2WI was more variable,
with 50% of cases showing hyperintensity (usually heterogeneous) and 15% showing hypointensity.
Tumor hypointensity on T2 WI is reported to be secondary to hemosiderin deposition or dense collagen matrix.
These two components can be distinguished by gradient-recalled-echo sequences,
with the former showing signal loss with increasing TE (time to echo) due to the magnetic susceptibility effect of hemosiderin (Fig.20).
[7] 35% of the tumors in our series had hemosiderin at histological examination (of which 42% showed characteristic MR features).
35% of GCT showed mixed-signal intensity on T2WI,
with 25% showing internal cystic areas.
Two GCT (10%) contained fluid-fluid levels due to secondary ABC,
confirmed with histological analysis.
Solid parts of GCT showed avid enhancement after contrast administration.
Most of the tumors exhibited aggressive imaging features (Fig.21),
with cortical bone destruction (85%) and soft-tissue extension (60%).
Periosteal reaction,
described as unusual in the literature,
was present in only one case of GCT located on the greater trochanter (Fig.2).
Three of the tumors were treated with Denosumab (due to their location - iliac bone and sacrum),
with dramatic response.
Tumors became hypointense on T2WI and developed a sclerotic border with reconstitution of the cortical bone,
the latter better depicted with CT (Fig.
22).