Compartments of intracranial bleeding:
A.
Intraaxial (=Intraparenchymal)
B.
Extraaxial:
- Subdural or epidural
- Subarachnoid
- Intraventricular
Chronology of bleeding:
–Hyperacute: < 12 hrs of evolution
–Acute: 12 hrs to 72 hrs of evolution
–Early subacute: 3 days to 7 days of evolution
–Late subacute: 7 days to 1 month
–Chronic: > 1 month
* some authors consider a hyperacute hemorrhage up to 24 hrs,
acute between 1 day and 3 days,
early subacute up to 7 days,
late subacute between 7 and 14 days [1].
Pathology of bleeding:
–Hyperacute: ruptured vessel leads to accumulation of red blood cells in the interstitial space
–Acute: red blood cells give up all available oxygen to the adjacent cells
–Early subacute: intracellular proteins such as hemoglobin start to oxidize
–Late subacute: glucose reserves become depleted leading to red blood cell distruction
–Chronic: dead red blood cells constituents are cleared by macrophages
Biochemistry of bleeding (Fig. 1):
–Hyperacute: intact red blood cells containing oxygenated hemoglobin (iron in the ferrous state = Fe2+)
–Acute: red blood cells become desaturated,
and oxyhemoglobin is converted to deoxyhemoglobin (iron in the ferrous state = Fe2+)
–Early subacute: intracellular deoxyhemoglobin within a hemorrhage is oxidized to methemoglobin (iron is oxidized to ferric state = Fe3+)
–Late subacute: red blood cell lysis and extravasation of methemoglobin (iron in the ferric state = Fe3+)
–Chronic: extracellular hemoglobin is oxidized by hemichromes to hemosiderin then phagocytized and accumulated in the lysosomes of macrophages (iron in the ferric state = Fe3+)
Biophysics of bleeding:
–Hyperacute: oxyhemoglobin has no unpaired electrons (diamagnetic): no magnetic moment and no proton relaxation enhancement
–Acute: deoxyhemoglobin contains the same Fe2+ iron but with four unpaired electrons (paramagnetic); heterogeneous distribution of deoxyhemoglobin due to confinement by the cellular membrane of the red blood cell; configuration change in the 3D-structure of the protein does not allow water mollecules closer than 0.3 nm from the paramagentic center
–Early subacute: methemoglobin has five unpaired electrons (highly paramagnetic); heterogeneous distribution of methemoglobin; configuration of the molecule allows approach within 0.3 nm of the paramagnetic center
–Late subacute: extracellular methemoglobin is no longer heterogeneous;
–Chronic: hemosiderin contains iron in the ferric state (strongly paramagnetic) and is insoluble in water
Etiology:
- Hypertension associated intraparenchymal hemorrhage affects patients in the average age of 50-60 years.
Most commonly occurs in deep brain structures like basal ganglia,
specially putamen [2] (Fig. 2).
- Cerebral amyloid angiopaty hemorrhage commonly occurs in lobar regions and affects the frontal and parietal lobes and is associated with microbleeds.
Affects particularly elderly patients.
GRE or SWI sequences are very useful for the detection of small and diffuse hypointense regions (Fig. 3).
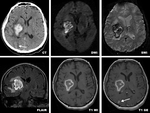
- Vascular malformations present large hematomas and usualy the presence of subarachnoid hemorrhage (Fig. 4).
- Hemorrhagic transformation secondary to ischemic stroke is a relatively common entity,
with comined features of a hematoma inside an ischemic stroke area.
- Venous hemorrhagic infarct
- Trauma
- Coagulation deficiency
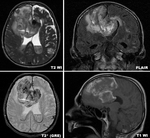
- Tumoral hemorrhage usualy involves more edema and mass effect compared to simple bleeding and the vasogenic edema is persistent on time (Fig. 5).
Also,
the hemosiderin rim has distinctive features,
as presented below:
The hemosiderin rim configuration is an important factor in delineating a simple intra-axial hematoma from an intratumoral hemorrhage.
In a simple intraparenchymal hematoma the hemosiderin rim is well defined,
and continuous,
when in the case of a hemorrhage associated with a tumor,
hemosiderin deposits are discontinuous or with a random distribution because the blood-brain barrier is not intact,
therefore hemosiderin-laden macrophages have access to the blood stream and the hemosiderin is resorbed [3].
- Others causes: venous stasis,
vasculitis,
eclampsy
CT and MRI findings [4]
CT appearance of bleeding:
–Hyperacute: slightly heterogeneous appearance with densities between 45-60 HU (similar to normal cerebral parenchyme areas)
–Acute and early subacute: increased density of the accumulation (80 HU) surrounded by an area of edema
–Late subacute: hematoma is isodense to normal cerebral parenchyme areas
–Chronic: hypodense appearance associating atrophy of surrounding parenchyme and ventriculomegaly
MRI appearance of bleeding:
–Hyperacute:hypo-/ or isointense on T1-weigthed images and high signal intensity on T2-weighted images.
–Acute: slightly hypo-/ or isointense on T1-weigthed images and low signal intensity on T2-weighted images.
–Early subacute: high signal intensity on T1-weigthed images and low signal intensity on T2-weighted images.
–Late subacute: Remaining high signal intensity on T1-weigthed images .
High signal on T2-weighted images.
–Chronic: Hemosiderin is slightly hypointense on T1-weigthed images,
and very hypointense on T2-weighted images.
Differential diagnosis:
On MRI,
hemorrhage is occasionally confused with other pathologies or conditions that cause hyperintensity on T1-weighted images [5].
Examples are lesions containing fat,
protein,
calcification,
and melanin.
- T1-weighted images can show a hyperintensity similar to that of intracellular and extracellular methemoglobin in metastases from melanoma,
however,
they less commonly display susceptibility on gradient recalled-echo images,
and they typically show some contrast enhancement.
- Lesions containing fat,
such as lipomas or dermoids,
are also hyperintense on T1-weighted images,
but the use of fat-suppression techniques,
such as chemical shift imaging or inversion recovery sequences (eg,
short-tau inversion recovery = STIR) can help differentiate fat from hemorrhage.
The presence of a chemical shift artifact may also indicate a fatty lesion.
- Hemorrhagic metastases usually show intense contrast enhancement,
which is not seen in bland hematomas.
- Calcification may mimic hemorrhage,
as both result in profound hypointensity on gradient-echo images.
However,
differences in the morphology and location of the abnormal signal intensity and in the clinical presentation suffice to distinguish the two.
CT may also help differentiate these entities,
as well as new and improved sequences from MRI machine developers.
- Residual gadolinium-based contrast material can resemble hemorrhage.
Prognosis
Important prognostic factors are:
-severity of stroke
-location of the hemorrhage
-size of the hemorrhage
The intracerebral hemorrhage score is used for evaluation of the outcome in hemorrhagic stroke [6]:
-GCS score 3-4: 2 points
-GCS score 5-12: 1 point
-GCS score 13-15: 0 points
-Age ≥80 years: yes=1 point; no=0 points
-Infratentorial origin: yes,
1 point; no=0 points
-Intracerebral hemorrhage volume ≥30 cm3: 1 point
-Intracerebral hemorrhage volume < 30 cm3: 0 points
-Intraventricular hemorrhage: yes=1 point; no=0 points
In a study by Hemphill et al,
all patients with an Intracerebral Hemorrhage Score of 0 survived,
and all of those with a score of 5 died [7].
Other prognostic factors:
-Nonaneurysmal perimesencephalic stroke has a better prognosis because of a lesser clinical impact
-The presence of blood in the ventricles is associated with a higher mortality rate
-Patients with oral anticoagulation-associated intracerebral hemorrhage have higher mortality rates and poorer functional outcomes