1) Introduction
Intracranial dural arteriovenous fistulas (DAVFs) are pathologic shunts between dural arteries and dural venous sinuses,
meningeal veins or cortical veins.
Their true incidence is unknown,
but they are estimated to account for 10%–15% of all intracranial arteriovenous malformations (AVMs),
comprising arround 6% of supratentorial and 35% of infratentorial AVMs.
DAVFs tend to present later in life than AVMs and are distinguished from parenchymal or pial AVMs mostly by the presence of a dural arterial supply and the absence of a parenchymal nidus.
Althouth clinical presentation is diverse,
patients may develop intracranial haemorrhage (overall risk ~ 1.5% per year),
sinus thrombosis or venous infarct as their first presentation (fig.
4,
fig.
5).
As so,
early diagnosis and management of DAVFs may prevent serious complications,
as the occurrence of stroke.
While invasiveness of conventional cerebral catheter angiography dificults regular investigation of patients with suspected DAVF,
more studies have attempted to use ultrasonography in order to detect early hemodynamic changes sugestive of DAVF.
2) Etiology and Patophysiology
Most the lesions are considered idiopathic but some associated conditions have been reported:
- dural sinus thrombosis;
- venous hypertension;
- previous craniotomy;
- head trauma;
- ear infection;
- secondary to aneurysmal rupture;
- heritable risk factors for venous thrombosis (such as antithrombin,
protein C and protein S deficiencies,
factor V Leiden);
The pathophysiology of DAVFs is believed to derive from venous thrombosis of a dural sinus.
The two most accepted hypotheses are:
- Venous thrombosis promotes venous hypertension,
which may induce the opening of physiologic arteriovenous connections (microscopic dural sinus’s vasa vasorum or the dural venous plexus embryonic remnants).
Maturation of these channels,
secondary to venous stenosis or occlusion,
results in the development of direct pathologic shunts.
- Venous hypertension,
due to outflow obstruction,
causes decreased cerebral perfusion with the subsequent release of angiogenic factors (vascular endothelial growth factor,
basic fibroblast growth factor,
etc.) promoting neoangiogenesis and the development of a DAVF.
Generally,
the arterial supply to DAVFs is by meningeal arteries and the fistulous connection is contained in the leaflets of the dura mater.
Occasionally,
as the fistula grows or becomes larger,
pial recruitment from parenchymal vessels and dilatation of cortical veins can occur.
3) Classification
The two commonly used classification systems are the more user-friendly Borden classification (fig.
1-3) and the more detailed Cognard system.
While the Borden classification system stratifies lesions based on the local and direction of venous drainage,
and the presence or absence of cortical venous drainage (CVD),
the Cognard classification adds the venous outflow architecture to the equation.
The DAVF venous drainage pattern determines the severity of symptoms and provides the foundation for both classification schemes and both of them associate CVD with increased risk of intracranial hemorrhage and nonhemorrhagic neurologic deficits.
3.1) Borden classification system
| |
Description |
CVD? / High risk lesion? |
| Borden 1 |
Drainage of meningeal arteries directly into dural sinus or meningeal vein,
with anterograde flow. |
|
| Borden 2 |
Anterograde drainage into dural venous sinus + retrograde flow into cortical subarachnoid veins (with CVR). |
YES |
| Borden 3 |
Direct retrograde flow from meningeal arteries into cortical veins or an “isolated” sinus segment (CVR only). |
YES |
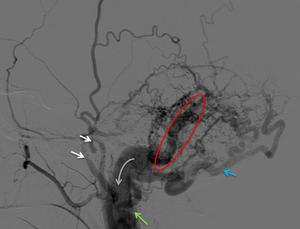
Fig. 1: A 58 year-old woman with a Borden 1 DAVF. Left ECA angiogram, lateral projection showing a osteodural AVF fistulous area on the left sigmoid sinus (red circle). We can see enlarged occipital artery with multiple transosseous branches feeding the dAVF (blue arrow), feeding branches from the superficial temporal and middle meningeal arteries (white arrows) and feeding branches from the posterior auricular artery (green arrow). The venous drainage is anterograde throw the jugular vein (grey arrow).
[Patient A in Fig. 1]
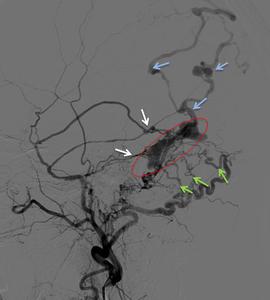
Fig. 2: A 44 year-old woman with Borden 2 DAVF. Left ECA angiogram, lateral projection (A) showing arterial feeding mainly from branches of the middle meningeal artery (white arrow) and transosseous branches of the occipital artery (green arrow). The fistulous area is despicable in a compartment of incarcerated left transverse sinus (red circle) with cortical venous reflux and venous ectasias (blue) present.
[Patient B in Fig. 2, Fig. 4 and Fig. 6]
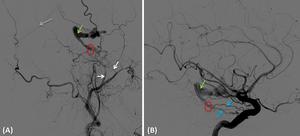
Fig. 3: A 76 year-old man with a Borden 3 DAVF. Right ECA angiogram, lateral projection (A) and right ICA angiogram, lateral projection (B) showing arterial supply to the fistula via the middle meningeal and accessory meningeal arteries (white arrows) and also from the tentorium cerebelli dural branches arising from the meningohypophyseal trunk of the ICA (blue arrow). The fistulous point seems to be located over the anterior and medial portion of tentorium cerebelli (red circle) and the venous drainage is made directly to an enlarged cortical vein (green arrows) that ultimately drains to the straight sinus (grey arrow).
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]
3.2) Cognard classification system
| |
Description |
CVD? / High risk lesion? |
| Type I |
Direct anterograde flow into a dural venous sinus |
|
| Type II a |
Drainage into a sinus with retrograde flow within the sinus |
|
| Type II b |
Drainage into a sinus with retrograde flow into cortical veins |
YES |
| Type II a+b |
Drainage into a sinus with retrograde flow within the sinus and cortical veins |
YES |
| Type III |
Direct drainage into a cortical vein without venous ectasia |
YES |
| Type IV |
Direct drainage into a cortical vein with ectasia >5mm and 3x larger than the diameter of the draining vein |
YES |
| Type V |
Direct drainage into spinal perimedullary veins |
YES |
4) Natural History
Even nowadays,
data on DAVFs natural history is limited.
Lack of CVD (Borden type I,
Cognard types I and IIa) is a favorable feature and is associated with a benign natural history and the presence of CVD (Borden type 2 and 3,
Cognard types IIb to V) is an aggressive feature that places DAVFs in a high-risk category.
Regarding high-risk lesions,
some values have been estimated:
- annual mortality rate of 10.4%;
- annual risk of intracranial hemorrhage of 8.1%;
- annual risk of Nonhemorrhagic neurologic deficits of 6.9%;
- annual hemorrhage risk is diferente between symptomatic (aprox 7.5%) and asymptomatic patients (aprox 1.5%).
- risk of rebleeding within 2 weeks after inicial hemorrhage of 35%
Although classifying DAVFs is helpful for risk stratification,
one should be aware that these lesions have a dynamic nature: the risk of conversion from a benign to an aggressive DAVF is small (~ 2%) but sufficient to warrant repeat investigation if the clinical picture changes.
5) Clinical Presentation
The majority of patients with DAVFs present in the fifth and sixth decades of life with symptoms related to lesion location and pattern of venous drainage.
The clinical picture may include:
- Pulsatile tinnitus (sometimes associated with an audible bruit).
A common symptom that results from high flow in AV shunts,
particularly those drainaging to the transverse or sigmoid sinus.
- Ocular/visual symptoms (such as ophthalmoplegia,
proptosis,
chemosis,
retro-orbital pain or decreased visual acuity),
more commonly found in cavernous sinus DAVFs.
- Intracranial hemorrhage (including subarachnoid,
subdural or intraparenchymal),
probably caused by rupture of draining veins,
as the source of hemorrhage is usually not the nidus but rather the distended leptomeningeal venous varices (fig.
6).
- Nonhemorrhagic neurologic deficits (seizures,
parkinsonism,
cerebellar symptoms,
apathy,
failure to thrive and cranial nerve abnormalities,
including rare cases of trigeminal neuralgia),
probably due to arterial steal.
- Dementia-like syndrome with cognitive decline (that may improve after treatment).
|
|
Common clinical presentation
|
Main arterial supply
|
Venous drainage
|
|
Transverse-sigmoid junction DAVFs
|
pulsatile tinnitus,
headaches.
|
Transmastoid branches of the occipital art,
branches of the middle meningeal art,
meningeal
branches of the ascending pharyngeal artery,
branches of the vertebral artery
|
Particular attention should be made to the direction of flow in
the vein of Labbé
|
|
Carotid-cavernous DAVFs
|
chemosis,
exophthalmos,
third and/or sixth cranial nerve palsies,
decreased visual acuity,
headache,
tinnitus and/or bruit.
|
Middle or accessory meningeal artery,
artery of the foramen
rotundum,
ascending pharyngeal artery
|
Superior ophtalmic vein or inferior ophthalmic vein into facial venous system and external jugular vein
|
|
Superior sagittal sinus DAVFs
|
intracranial hemorrhage,
progressive neurological deficits.
|
Bilateral supply from branches of the middle meningeal art,
anterior
falcine artery from the ophtalmic art
|
|
|
Tentorial DAVFs
|
non-specific symptoms headache,
ataxia,
trigeminal neuralgia,
etc.)
|
Tentorial artery
(Bernasconi and Cassinari),
petrosal and petrosquamosal branches of the middle meningeal artery
|
Superior petrosal sinus or pontine,
perimesencephalic and basal veins into the
galenic system
|
|
Anterior cranial fossa (ethmoidal) DAVFs
|
headaches,
seizures,
ocular symptoms (such as exophthalmos).
atypical symptoms (such as proptosis,
chemosis,
elevated IOP,
and loss of vision
high incidence of ICH
|
Posterior and anterior
ethmoidal branches of Oph art.
Secondary supply from the ethmoidal branches of the internal and middle meningeal arts or superior temporal artery
|
Main drainage into the frontal cortical veins and secondarily into thesuperior sagital sinus or the anterior
pontomesencephalic vein
|
|
Superior petrosal sinus DAVFs
|
headaches,
pulsatile tinnitus,
trigeminal neuralgia,
ocular symptoms,
intracranial hemorrhage
|
Tentorial artery
(Bernasconi and Cassinari),
petrosal and petrosquamosal branches of the middle meningeal artery
|
Superior petrosal sinus, pontine and perimesencephalic
veins.
It also drains retrograde to the vein of Galen and antegrade
down the internal jugular vein
|
Common clinical presentations by anatomical localization
Indicators for early treatment include:
- intracranial hemorrhage;
- increased intracranial pressure;
- increased intraocular pressure causing visual loss;
- neurovascular compression producing neurological deficits;
- presence of leptomeningeal drainage / CVD.
6) Diagnosis / Imaging
Because clinical and imaging features can be non-specific,
the diagnosis of a DAVF is often delayed or missed.
Occasionally plain films can demonstrate grooving within the skull vault due to chronic compression from enlarged middle meningeal vessels.
Cross-sectional imaging techniques (CT and MR imaging) aid in the diagnosis and mostly constitute the initial radiological evaluation.
Head computed tomography and magnetic resonance imaging studies have a reported sensitivity for detection of DAVFs of 70% and 81%,
respectively.
Conventional angiography remains the most accurate method for complete characterization and classification of DAVFs.
Doppler sonography represents a minimally invasive test that can be applied as a diagnostic screening tool,
and treatment monitoring in patients with DAVF,
as we will address in a separated topic under "Findings and procedure details".
6.1) CT
Non-contrast CT is limited to the identification of complications such as intracranial hemorrhage,
venous sinus thrombosis or edema due to venous congestion.
Linear bony defects formed by enlarged emissary veins,
similar to the grooving abnormality seen on plain film,
can also be found.
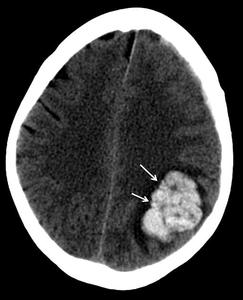
Fig. 4: CT scan of a 44 year-old woman with a Borden 2 DAVF, later confirmed by digital subtraction angiography, who presented with focal neurological signs secondary to the brain hemorrhage seen in the left parietal lobe (white arrow).
[Patient B in Fig. 2, Fig. 4 and Fig. 6]
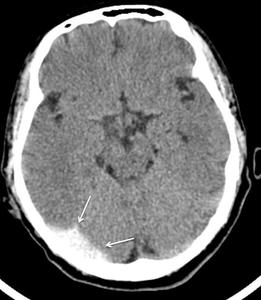
Fig. 5: Initial CT scan of a 53 year-old man revealing a hyperdense region in the right sigmoid and lateral sinus. Broader study with gadolinium injection revealed a large thrombus extending from the transverse, lateral and sigmoid sinus to the cervical portion of the internal jugular vein.
[Patient D in Fig. 5]]
6.2) MR imaging
An increasing number of DAVFs are being diagnosed incidentally due to the more frequente use of high-resolution MRI and the wider availability of the technique.
T2-weighted MRI is more sensitive to the white matter changes of venous congestion or infarction when compared to CT.
Findings vary with the type of DAVF and include:
- dilated cortical vessels,
appearing as flow-voids located within the cortical sulci without a true parenchymal nidus (originally referred to as a “pseudophlebitic” pattern);
- venous pouches;
- prominent occipital arteries;
- vascular enhancement;
- osseous abnormalities due to ecstatic or hypertrophied vessels;
- sinus stenosis or thrombosis;
- signs of venous hypertension (as white matter hyperintensity,
intracranial hemorrhage or venous infarction).
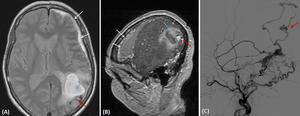
Fig. 6: MRI of a 44 year-old woman with Borden 2 DAVF, later confirmed by digital subtraction angiography (C). Axial DP (A) and Sagital contrast-enhanced T1 (B) showing parenchymal (blue arrow) and subdural hematoma (white arrow). We can clearly depict the flow void pouch on axial DP (A), enhancing after contrast (B), probably corresponding to the rupture point of the venous pouch of this cortical vein. Angiographic study (C) confirms dAVF with cortical venous reflux and venous ectasia (red arrow) as the likely point of rupture.
[Patient B in Fig. 2, Fig. 4 and Fig. 6]
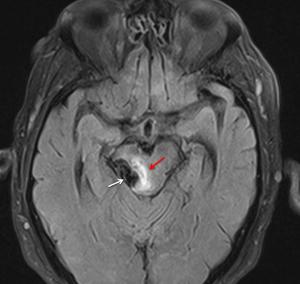
Fig. 7: A 76 year-old man with a Borden 3 DAVF, later confirmed by digital subtraction angiography. Axial FLAIR showing mesencephalic edema (red arrow) and flow voids corresponding to a venous ectasia.
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]
Lesions Borden type 1 and 2 are more likely to show flow-void clustering,
engorged ophthalmic veins or proptosis,
while aggressive type 2 or 3 lesions may reveal dilated vessels,
prominente vascular enhancement and hemorrhage.
6.3) CT angiography
Any suspicious flow void cluster around the dural venous sinus should prompt additional evaluation with dynamic CTA,
MRA,
or DSA. CTA is particularly useful in treatment planning by helping define the arteriovenous shunt relative to surrounding brain and skull.
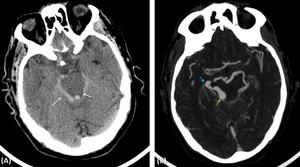
Fig. 8: A 76 year-old man with a Borden 3 DAVF. Initial CT scan (A) revealing subarachnoid hemorrhage in perimesencephalic cistern bilaterally (white arrow). CT angiography (B) showing the presence of venous ectasia with wall irregularities, located around the perimesencephalic cistern, more pronounced at the right (blue arrow), apparently converging to an aneurysmatic dilatation (green arrow). We cannot depict the arteriovenous nidus in this study.
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]
Multi-detector CTA can provide high resolution for vascular anatomy and,
in the investigation of tinnitus,
has the additional advantage of evaluating inner and middle ear abnormalities.
Arterialised venous blood within the veins draining a DAVF has increased density when compared to non-arterialised blood and that can be assessed by carefull evaluation using narrow window settings.
Recent publications on 4D CTA by using 320-section multidetector row CT angiography have highlighted its potential to diagnosis,
classification and treatment planning.
More reported experience is needed,
though.
6.4) MR angiography
Studies have reported that MRA has more sensitivity than CTA for the detection of DAVFs (50% vs 15.4%).
Due to current limitations of low resolution,
restricted FOV,
and saturation artifacts,
the negative predictive value of MRA is still inadequate to exclude DAVFs.
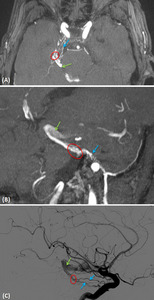
Fig. 9: A 76 year-old man with a Borden 3 DAVF. TOF-3D MPR axial MIP 4mm (A) TOF-3D MPR oblique MIP 7mm (B) and corresponding right ICA angiogram, lateral projection (C) showing a DAVF located over the anterior and medial portion of tentorium cerebelli (red circle). We can see and compare the arterial supply from the meningohypophyseal trunk of the ICA, via the tentorium cerebelli dural branches (blue arrows), and the venous drainage directly to an enlarged cortical vein (green arrows).
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]
3D TOF MRA is sensitive enough to detect flow signal in arterialized veins,
especially with reversal of flow direction,
but this technique currently has a high false-positive rate of 14%.
Time-resolved MRA techniques are promising and may be reliable for DAVF screening and surveillance in the future.
6.5) Digital subtraction angiography
Benign disease,
without cortical venous reflux can be missed using both CT and MRI.
Therefore,
conventional angiography remains the most accurate method for detection and classification of DAVFs (fig.
1-3,
fig.
11).
However,
their invasiveness and high cost make it less attractive as a screening tool.
The pretreatment angiographic evaluation must include:
- Bilateral ICA and ECAs,
and it can also include the vertebral arteries when evaluating DAVFs in the posterior fossa or tentorium.
- Subsequently,
superselective angiography is required to access the arterial supply (including anastomoses with arterial distributions to the brain,
orbit,
or cranial nerves),
and the venous anatomy (with respect to the pattern of venous drainage and the adequacy of normal venous flow from the brain).
Arterial and venous anatomy can affect the selection of the ideal treatment modality and it’s extremely important in order to avoid treatment complications.
|
Angiographic features associated with aggressive behaviour:
|
|
- leptomeningeal retrograde;
- venous drainage,
variceal or aneurysmal venous structures;
- galenic venous drainage.
|

![Fig. 1: A 58 year-old woman with a Borden 1 DAVF. Left ECA angiogram, lateral projection showing a osteodural AVF fistulous area on the left sigmoid sinus (red circle). We can see enlarged occipital artery with multiple transosseous branches feeding the dAVF (blue arrow), feeding branches from the superficial temporal and middle meningeal arteries (white arrows) and feeding branches from the posterior auricular artery (green arrow). The venous drainage is anterograde throw the jugular vein (grey arrow).
[Patient A in Fig. 1]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548603?maxheight=150&maxwidth=150)
![Fig. 2: A 44 year-old woman with Borden 2 DAVF. Left ECA angiogram, lateral projection (A) showing arterial feeding mainly from branches of the middle meningeal artery (white arrow) and transosseous branches of the occipital artery (green arrow). The fistulous area is despicable in a compartment of incarcerated left transverse sinus (red circle) with cortical venous reflux and venous ectasias (blue) present.
[Patient B in Fig. 2, Fig. 4 and Fig. 6]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548604?maxheight=150&maxwidth=150)
![Fig. 3: A 76 year-old man with a Borden 3 DAVF. Right ECA angiogram, lateral projection (A) and right ICA angiogram, lateral projection (B) showing arterial supply to the fistula via the middle meningeal and accessory meningeal arteries (white arrows) and also from the tentorium cerebelli dural branches arising from the meningohypophyseal trunk of the ICA (blue arrow). The fistulous point seems to be located over the anterior and medial portion of tentorium cerebelli (red circle) and the venous drainage is made directly to an enlarged cortical vein (green arrows) that ultimately drains to the straight sinus (grey arrow).
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548605?maxheight=150&maxwidth=150)
![Fig. 4: CT scan of a 44 year-old woman with a Borden 2 DAVF, later confirmed by digital subtraction angiography, who presented with focal neurological signs secondary to the brain hemorrhage seen in the left parietal lobe (white arrow).
[Patient B in Fig. 2, Fig. 4 and Fig. 6]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548606?maxheight=150&maxwidth=150)
![Fig. 5: Initial CT scan of a 53 year-old man revealing a hyperdense region in the right sigmoid and lateral sinus. Broader study with gadolinium injection revealed a large thrombus extending from the transverse, lateral and sigmoid sinus to the cervical portion of the internal jugular vein.
[Patient D in Fig. 5]]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548607?maxheight=150&maxwidth=150)
![Fig. 6: MRI of a 44 year-old woman with Borden 2 DAVF, later confirmed by digital subtraction angiography (C). Axial DP (A) and Sagital contrast-enhanced T1 (B) showing parenchymal (blue arrow) and subdural hematoma (white arrow). We can clearly depict the flow void pouch on axial DP (A), enhancing after contrast (B), probably corresponding to the rupture point of the venous pouch of this cortical vein. Angiographic study (C) confirms dAVF with cortical venous reflux and venous ectasia (red arrow) as the likely point of rupture.
[Patient B in Fig. 2, Fig. 4 and Fig. 6]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548609?maxheight=150&maxwidth=150)
![Fig. 7: A 76 year-old man with a Borden 3 DAVF, later confirmed by digital subtraction angiography. Axial FLAIR showing mesencephalic edema (red arrow) and flow voids corresponding to a venous ectasia.
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548610?maxheight=150&maxwidth=150)
![Fig. 8: A 76 year-old man with a Borden 3 DAVF. Initial CT scan (A) revealing subarachnoid hemorrhage in perimesencephalic cistern bilaterally (white arrow). CT angiography (B) showing the presence of venous ectasia with wall irregularities, located around the perimesencephalic cistern, more pronounced at the right (blue arrow), apparently converging to an aneurysmatic dilatation (green arrow). We cannot depict the arteriovenous nidus in this study.
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548608?maxheight=150&maxwidth=150)
![Fig. 9: A 76 year-old man with a Borden 3 DAVF. TOF-3D MPR axial MIP 4mm (A) TOF-3D MPR oblique MIP 7mm (B) and corresponding right ICA angiogram, lateral projection (C) showing a DAVF located over the anterior and medial portion of tentorium cerebelli (red circle). We can see and compare the arterial supply from the meningohypophyseal trunk of the ICA, via the tentorium cerebelli dural branches (blue arrows), and the venous drainage directly to an enlarged cortical vein (green arrows).
[Patient C in Fig. 3, Fig. 7, Fig. 8 and Fig. 9]](https://epos.myesr.org/posterimage/esr/ecr2014/119582/media/548611?maxheight=150&maxwidth=150)









