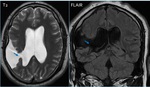
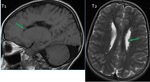
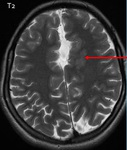
1. Close-lipped schizencephaly ( Fig. 1 Fig. 2 ) :
Schizencephaly may be close or open-lipped,
uni or bilateral,
and typically manifests with seizures and mental retardation.
It is a disorder of neuronal migration caused by second-trimester insult or genetics leading to a full-thickness communication between the pia and ventricular system ependyma.
The cleft is lined by abnormal gray matter,
which may act as a seizure focus.
If the abnormal gray matter is closely apposed,
the anomaly is close-lipped; if unopposed,
it is open-lipped.
The diagnosis is evident in open-lipped schizencephaly (a wide CSF communication exists between the lateral ventricle and the subarachnoid space).
Close-lipped schizencephaly is more subtle,
it may manifests as an ectopic gray matter extending from the ventricular wall to the brain surface and outlining a thin,
sometimes invisible cleft.
The diagnosis may also be suggested by gyri radiating into the cleft,
associated sub ependymal heterotopias,
and an indentation in the wall of the lateral ventricle.
Multiplanar imaging is necessary,
as isolated imaging in just one plane may miss the defect.
Associated anomalies include absence of the septum pellucidum (90%) and septo-optic dysplasia (1,2).
2. Gray-matter heterotopia:( Fig. 3 Fig. 4 Fig. 5 Fig. 6)
Gray-matter heterotopia is a relatively frequent congenital anomaly that results from an in utero arrest of neuronal migration.
The abnormally located gray matter is usually found in the subependymal region of the lateral ventricles,
around the trigones in particular.
Occasionally,
the heterotopias are more peripherally located near the cerebral cortex.
Patients come to clinical attention because of seizures and developmental delay.
Heterotopias may be classified into three groups: subependymal heterotopia,
focal subcortical heterotopia,
and diffuse (band) heterotopia.
The imaging sign is the isointensity with cortical gray matter on all sequences and the lack of enhancement after injection of contrast media.
These features allow the radiologist to distinguish this abnormality from the subependymal nodules observed in tuberous sclerosis (1,3,4).
3. Alobar holoprosencephaly:
Three subtypes of holoprosencephaly have been proposed according to the severity of cerebral and facial anomalies.
These include alobar,
semilobar,
and lobar holoprosencephaly.
A rare middle interhemispheric variant has also been described.
In the most severe subtype,
alobar holoprosencephaly,
there is complete or near complete lack of hemispheric cleavage.
The typical imaging features include a primitive midline mono-ventricle,
fused thalami,
absence of the falx cerebri,
and interhemispheric fissure; Midline facial anomalies (single maxillary central incisor,
hypotelorism,
cyclopia) are frequently present.
A multitude of extracranial and chromosomal abnormalities may occur in association with alobar holoprosencephaly,
frequently leading to a stillborn infant or death in the neonatal period (1,
5,
6).
4.Dysgenesis of the corpus callosum with associated interhemispheric cyst ( Fig. 9 Fig. 10 :
The corpus callosum develops in an anterior-to-posterior form,
with the posterior genu and anterior body forming first,
followed by the posterior body,
splenium,
and rostrum.
Abnormalities of the corpus callosum are frequently associated with other midline abnormalities such as cysts,
lipomas,
or abnormal anterior or hippocampal commissures.
In approximately 75% of cases,
callosal abnormalities are also associated with other malformations of cortical development such as heterotopia or abnormalities of sulcation; so we must carefully evaluate the brain when an abnormality of the corpus callosum is detected.
(7,8).
Complete agenesis of the corpus callosum lead to the development of abnormal parallel white-matter tracts called Probst bundles that run anteroposteriorly along the medial aspect of the bilateral lateral ventricles.
Additionally,
the lateral ventricles are frequently widely spaced and parallel in configuration.
5.
Craniopharyngioma ( Fig. 13)
Craniopharyngiomas are benign tumors that arise from epithelial remnants and ectopic embryonic cell rests of the Rathke's pouch.
They exhibit a bimodal age distribution with most cases occurring either between 5 and 15 years or after 50 years of age.
Clinical symptoms are generally insidious due to the relatively slow rate of tumor
growth.
The most frequent symptoms at presentation are headache of the early morning,
vomiting,
visual field defects,
and endocrine abnormalities.
Craniopharyngiomas are most commonly located within the suprasellar cistern that extend inferiorly into the sella.
But they can occupy only the suprasellar space,
the sella in isolation,
or even the infrasellar region.
It exists two histopathologic subtypes of craniopharyngiomas: adamantinomatous and squamous-papillary. Adamantinomatous craniopharyngiomas occur in children (and occasionally adults) and demonstrate these typical imaging features: cyst formation,
calcification,
and enhancement.
On MRI,
cystic components are usually multiple,
associated,
and express varied signal characteristics which depend on their relative protein content-with greater concentrations of protein engendering increased T1 signal and concomitant T2 hypointensity.
Typically,
the cysts
are hyperintense to CSF on fluid-attenuated inversion recovery (FLAIR) sequences.
Solid components of the mass are usually isointense to brain parenchyma on T1-weighted images,
iso-to hyperintense on T2-weighted images,
and demonstrate heterogeneous enhancement.
On CT,
the cystic constituent exhibits hypoattenuation.
CT is very sensitive for the detection of areas of calcification.
The squamous-papillary subtype arises in adults and is primarily solid with occasional mixed solid and cystic constituents. In this subtype,
calcifications are much less frequent (9,10).
6. Primary panhypopituitarism with translocation of the pituitary bright spot (i.e.,
ectopic posterior pituitary lobe) ( Fig. 14)
The posterior pituitary bright spot is thought to be associated to the inherent signal intensity of neurosecretory granules in the neurohypophysis; therefore,
an ectopic bright spot may be found in any process that interrupts the transport of antidiuretic hormone from the hypothalamus to the posterior pituitary lobe.
Primary panhypopituitarism arises when the pituitary is congenitally hypoplastic or absent.
A high correlation exists between the hormonal disorder and ectopia of the neurohypophysis.
Growth disturbances may dominate the clinical picture.
Characteristic MR findings of primary panhypopituitarism include a small sella turcica,
diminutive pituitary gland,
atretic pituitary stalk,
and ectopic posterior pituitary bright spot (11,12).
7.
Alexander's disease (infantile form) ( Fig. 15)
Alexander's disease is a rare anomaly characterized by a mutation to the gene GFAP that causes massive deposition of Rosenthal fibers in astrocytes leading to demyelination and rarefaction of the subependymal,
subpial,
and perivascular white matter with a frontal predominance.
There are three distinct clinical subgroups: infantile,
juvenile and adult.
The infantile form is most frequent and is characterized by early onset of macrocephaly,
developmental delay,
and seizures.
Rapid evolution of the disease typically leads to death within the first 2 to 3 years of life.
Establishing a definite diagnosis typically needs brain biopsy or autopsy.
Typical imaging findings in the infantile form of Alexander disease include a predilection for the bilateral frontal lobe white matter,
which manifests as hypoattenuation on CT and corresponding T2 hyperintensity on MRI.
It progresses posteriorly to involve the parietal white matter as well as the internal and external capsules.
Similar signal abnormalities can interest the bilateral caudate heads.
The typical finding is T1 hyperintensity and T2 hypointensity of the periventricular frontal rim,
which is hyperdense on CT and shows avid enhancement.
Further distinguishing features include involvement of the subcortical U fibers early in the disease process.
Alexander disease is one of the few metabolic disorders that shows abnormal enhancement.
Involvement of brainstem and cervical cord is rare.
Cysts may be present in affected zones of the brain in later stages of the pathology (13,14).
8.
Dysembryoplastic neuroepithelial tumor ( Fig. 16)
Dysembryoplastic neuroepithelial tumor (DNET,
DNT) is a benign (WHO grade I), mixed glial neuronal tumor that arise from the supratentorial cortex.
The most frequent clinical presentation include long-standing,
drug-resistant partial complex seizures in a child or a young adult.
DNETs are most commonly found in the temporal lobe.
Treatment implies surgical resection,
which can be curative,
even if incomplete.
Imaging findings are characteristic and can be diagnostic in many cases.
DNETs are generally well-delimited,
multi-lobulated,
multicystic appearing T2 hyperintense masses that arise from the cortex.
Uniquely characteristic properties of DNETs are their wedge or triangular shape with the apex pointing toward the ventricles and internal "bubbly" appearance.
DNETs typically do not enhance after injection of contrast media.
They also show high apparent diffusion coefficient (ADC) values and low relative cerebral blood volume (rCBV) on perfusion imaging.
Since the tumor is slow growing,
it can lead to remodeling or scalloping of the inner table of the adjacent calvarium in about 50% of cases,
which is best depicted on CT.
The DNETs have a strong association with focal cortical dysplasia (15,16).
9.Sturg-Weber syndrome ( Fig. 17)
Sturg-Weber syndrome is a neurocutaneous syndrome that occurs sporadically.
The hallmark of this disease is the vascular angiomatous lesion touching the face in the distribution of the trigeminal nerve and ipsilateral brain and meninges.
Imaging findings include cortical calcifications,
lobar atrophy with secondary calvarial changes,
lepto-meningeal enhancement,
angiomas of the choroid plexus,
and venous abnormalities.
The full extent of the cortical vascular lesions may not be apparent on unenhanced imaging and needs therefore systematic injection of the contrast media (1,17,18).
10. Neurofibromatosis type 2 ( Fig. 18 ):
Neurofibromatosis type 2 (NF 2) is an autosomal dominant disorder that is recognized as a distinct form of disease separated genetically,
clinically,
and radiographically from neurofibromatosis type 1 (NF 1).
Bilateral vestibular schwannomas are the hallmark of this disease,
generally presenting during or soon after puberty.
Schwannomas of other cranial and spinal nerves,
intracranial and spinal meningiomas,
and spinal ependymomas may also be found in these patients (1,19,20).
11.Tuberous sclerosis with subependymal giant cell astrocytoma ( Fig. 19)
Tuberous sclerosis complex (TSC) is the second most common of the neurocutaneous syndromes and is characterized by the formation of hamartomatous lesions in several organs.
Many patients have evident signs at birth,
whereas others remain undiagnosed for many years.
The classic clinical triad (adenoma sebaceum,
seizures,
and mental retardation) is present only in a minority of patients with TSC.
The CNS lesions include astrocytic hamartomas of the retina,
cortical tubers,
and subependymal nodular hamartomas.
Approximately 15% of patients present subependymal giant-cell astrocytomas (GCA),
which are defined as grade 1 neoplasms by the WHO.
The GCAs virtually always arise near the foramen of Monro and can induce obstructive hydrocephalus.
By CT,
GCA appears as an enhancing soft-tissue mass with variable calcification.
MR imaging expresses hypointensity to isointensity on Tl-weighted images,
hyperintensity on T2-weighted images,
and uniform enhancement after injection of gadolinium.
Even subependymal hamartomas usually enhance on MRI,
and interval increase in size is considered the only reliable sign of GCA development (18,21,22).
Lesions associated with TSC outside of the CNS include renal angiomyolipomas (AML), lymphangiomyomatosis (LAM) of the lung,
and cardiac rhabdomyomas.
12. Cerebellar hemangioblastomas in a patient with von Hippel-Lindau syndrome (Fig. 20)
Hemangioblastomas are unusual tumors,
representing less than 3% of all intracranial neoplasms,
and typically arise in patients aged between 20 and 50 years old.
Hemangioblastomas occur sporadically or as a manifestation of von Hippel-Lindau
(VHL) syndrome.
VHL syndrome is inherited as an autosomal dominant disorder.
In addition to hemangioblastomas of the cerebellum,
brainstem,
spinal cord and other intracranial structures,
lymphatic duct adenocarcinomas and non-CNS manifestations (cysts of the kidneys,
pancreas,
and liver,
as well as renal cell carcinoma,
microcystic adenoma of the pancreas,
and pheochromocytomas) may be seen in patients with VHL syndrome.
Hemangioblastomas in VHL syndrome are multiples in about 40% of cases and typically manifest at least 10 years earlier than those occurring in patients without the syndrome.
The diagnosis of VHL syndrome requires multiple hemangioblastomas or one hemangioblastoma with other visceral manifestations of the disease.
More than 50% of hemangioblastomas are cystic,
with a mural nodule that presents intense enhancement after injection of contrast media.
Due to hypervascularity, flow voids are commonly seen in the mass or at its periphery.
On perfusion imaging,
hemangioblastomas typically show increased CBV.
Hemangioblastomas also show increased diffusion.
(18,23,24).
13. Periventricular leukomalacia ( Fig. 21)
Periventricular leukomalacia (PVL) is used to describe brain injury in premature neonates in relation with the susceptibility of the periventricular white matter to ischemic or infectious inflammatory processes.
Deep periventricular white matter is particularly susceptible to injury because glial cells in this zone are differentiating into astrocytes and oligodendrocytes,
with some in the process of myelination.
Ultrasound examination can show echogenic foci in the “watershed" zones of the periventricular trigones and frontal horns.
Although helpful in the neonatal intensive care unit,
ultrasound is not sensitive and has a high false-positive rate and low positive predictive value,
with only about 26% of infants who have prolonged periventricular echogenicity corresponding to significant changes on MR.
As PVL evolves and the water content of the infant brain decreases,
CT and MRI can be very useful.
The examiner may see hemorrhage,
small cyst formation (2-3 weeks),
large cyst formation (i.e.,
"Swiss cheese appearance"),
coalescence (1-3 months),
and deep white-matter hypoplasia.
Although MRI can show all of these findings,
CT may miss small cysts.
The imaging important sign of late PVL (synonymous with white-matter hypoplasia) is deep cortical sulci prolonging inward to enlarged lateral ventricles with diminished volume of white matter.
The size of the head is normal (1,25).
14. Leptomeningeal cyst,
growing skull fracture( Fig. 22 Fig. 23 ):
Leptomeningeal cysts (LMCs) are infrequent lesions that are typically a consequence of childhood cranial fractures and may become symptomatic years later.
Seizures are the most common symptom.
LMCs are extra-axial masses having CSF intensity on all MR sequences and typically induce erosion of the adjacent inner table of the skull and herniate into the bone.
The proposed pathogenesis includes dural tear under an acute fracture,
followed by herniation of the arachnoid tissue through the defect.
The subarachnoid adhesions then isolate the arachnoid fluid and form an encapsulated cyst,
which keeps enlarging by CSF pulsations toward both the overlying fracture and the underlying brain.
Gliosis in the adjacent brain parenchyma is also frequently found,
best seen as hyperintensity on FLAIR images (26-28).
15. Arteriovenous malformation( Fig. 24 ):
AVMs are a tangle of abnormal blood vessels in which arteries connect directly to veins without an intervening capillary network.
Typical clinical manifestations include headache and hemorrhage (50%),
seizure (25%),
and focal neurologic deficit (20%-25%).
The peak age of presentation is between 20 and 40 years of age.
Most are located supratentorally.
AVMs have a characteristic appearance on T2-weighted MR images,
with a nidus of abnormal flow voids,
and enlarged draining veins.
After injection the AVM is strongly enhanced.
AVMs may be seen on MR angiography,
but the gold standard for characterizing AVMs is DSA to dynamically evaluate the feeding arteries,
the nidus,
and the draining veins (29-31).
16. Hypertrophic olivary degeneration (HOD) ( Fig. 25 ):
HOD arises secondary to damage to the dentatorubro-olivary pathway (or Guillain-Mollaret triangle) comprising white-matter tracts prolonging between the dentate nucleus of the cerebellum,
the contralateral red nucleus of the midbrain,
and the ipsilateral inferior olivary nucleus (ION) of the medulla.
Brainstem lesions disrupting this pathway include the sequelae of ischemic infarct,
demyelination,
hemorrhage,
and surgery.
The most common clinical symptoms associated with HOD are rhythmic involuntary movement of the soft palate,
uvula,
pharynx,
larynx,
and upper extremity.
Typical imaging findings in association with a suggestive clinical presentation can establish the diagnosis.
MRI is the imaging modality of choice.
MR findings are dependent on the time passed since the causative insult.
During the first 6 months,
HOD manifests as T2 hyperintensity in the affected ION without definitive enlargement.
Beginning at about 6 months after the insult,
the ION hypertrophies and retains its T2 hyperintensity.
This characteristic appearance typically persists for up to 4 years after which there is resolution of the hypertrophy
with retention of the T2 hyperintensity.
There is no associated abnormal enhancement after injection of contrast media and lesions may also be bilateral (32,33).
17. Cerebral venous thrombosis( Fig. 26 Fig. 27 ):
Cerebral venous thrombosis is an infrequent disorder in which the diagnosis and appropriate management are often delayed because of the nonspecific clinical manifestations.
This pathology is most frequent in infants with dehydration or septicemia.
Etiologic factors associated with venous thrombosis in adults include Oral contraceptives,
pregnancy,
diabetes,
malignant neoplasms,
and hematologic disorders.
CT findings include hyperdense thrombus within the deep and/or cortical intracranial venous structures on noncontrast studies,
which is not always present.
Hypodensity in relation with edema or infarction may also be seen.
Hemorrhagic infarcts are frequent,
and bilateral involvement is frequently seen,
especially of the thalami.
Angiography identifies nonfilling of the cerebral veins with enlarged collaterals.
MRI may be used as the definitive investigation.
The thrombosed vessel becomes hyperintense on T1 and T2-weighted images as the thrombus becomes replete with extracellular methemoglobin.
Clot may also express loss of signal on T2*-weighted images; however,
none of these signs is completely specific for diagnosis of venous
thrombosis.
Absence of contrast filling,
best seen after injection of contrast media is the most reliable finding.
Antithrombotic therapy is considered to be safe and effective treatment,
even when hemorrhage is present (34,35).
18. Posterior reversible encephalopathy syndrome (PRES) ( Fig. 28)
PRES manifests with headache,
seizures,
altered mental status,
and blindness.
It is most often associated with rapid increases in blood pressure (eclampsia and pre-eclampsia,
acute glomerulonephritis,
and hypertensive crisis).
Other situations
may also induce PRES,
most notably in transplant recipients,
which may in part be due to immunosuppresants.
If unrecognized,
PRES may progress to infarctions.
If detected and treated,
deficits are generally reversible.
Imaging has an important role in early diagnosis.
The target area for PRES is the posterior cerebral circulation.
The resulting bilateral occipitoparietal vasogenic edema,
predominantly subcortical in location,
manifests as hypodensity on CT,
hyperintensity on T2-weighted images,
and increased diffusion.
Cortical
involvement is not uncommon and contrast enhancement is occasionally observed.
The lesions are not necessarily posterior or reversible.
They are frequently located in the frontal lobes and may be found in deep cerebral white matter and even the brainstem.
Decreased diffusion may exist even in the early stages and indicates possible infarcts.(36-38).
19. Superficial siderosis( Fig. 29 ):
Superficial siderosis is an uncommon anomaly characterized by the deposition of hemosiderin in the leptomeninges covering the cerebrum,
cerebellum,
brainnstem,
cranial nerves,
and spinal cord.
Chronic subarachnoid hemorrhage is the cause of this disorder.
The clinical triad of sensorineural hearing loss,
cerebellar ataxia,
and pyramidal signs along with hemorrhagic or xanthochromic CSF allows a clinical diagnosis.
MRI easily confirms the hemosiderin deposition and gives a diagnosis at an earlier stage of the disease.
T2-weighted images show a rim of marked hypointensity along the surface of affected structures,
which is much better appreciated on T2*-weighted images.
CT occasionally suggests the diagnosis by demonstrating a rim of mild hyperdensity around the brainstem; however,
MRI shows this abnormality to a much better advantage.
(39,40).
20. Intracranial hypotension( Fig. 30)
Intracranial hypotension (lH) is a clinical and pathologic entity in which imaging plays a capital role.
The common clinical presentation is positional,
or orthostatic,
headaches.
There are several varieties of lH.
The most frequent is spontaneous intracranial hypotension (SIH),
which is typically due to exertional rupture of a congenital cervicothoracic dural diverticulum,
with persistent spillage of CSF into the paraspinal soft tissues.
Fluid often tracks cephalad and collects in the deep nuchal region,
providing a hint to the diagnosis.
Post-lumbar puncture headache is an iatrogenic version of SIH.
Chronic ventricular overshunting and post-neurosurgical or posttraumatic CSF loss are other known etiologies of the syndrome.
The typical imaging features of IH include expansion of the epidural venous plexus,
diffuse dural enhancement,
sinking of the brain,
subdural fluid collections,
and distention of the dural venous sinuses.
These are best demonstrated with gadolinium-enhanced T1-weighted images (41-43).
21. Cavernous angioma with associated developmental venous anomaly( Fig. 31)
Cavernous angiomas are vascular malformations that are considered benign hamartomatous lesions with thin-walled sinusoidal vessels.
Many are associated with developmental venous anomalies (DVAs).
They are frequently asymptomatic but may bleed and induce headaches and seizures.
These lesions are generally sporadic but may also occur in familial forms,
and in those cases tend to be multiple.
Noncontrast CT may show a high-density lesion.
MRI is the modality of choice for diagnosis,
with prominent loss of signal on T2* sequences.
The lesions typically show both T1 and T2 heterogeneous internal “popcorn" signal intensities with a complete dark rim on T2-weighted images representing hemosiderin.
Some lesions may express subtle enhancement after injection,
especially if associated with a DVA (44,45).
22. Colloid cyst (Fig. 32)
Third ventricular colloid cysts are unusual masses ( <1% of all intracranial tumors).
Patients typically present with postural headaches induced by a ball-valve effect at the foramen of Monro,
with associated obstructive hydrocephalus.
Acute ventricular obstruction resulting in death has been reported.
Colloid cysts generally do not enhance after injection and are either isodense or hyperdense to brain on CT.
The signal characteristics on MR vary,
but the most frequent presentation is a hyperintense mass on T1-weighted images due to the high cholesterol content (46,47).
23. Hypothalamic hamartoma (HH),
or hamartoma of tuber cinereum( Fig. 33)
Hypothalamic hamartomas (HHs) are congenital,
nonneoplastic lesions classically associated with laughing seizures.
Patients may also present central precocious puberty (CPP) or be completely asymptomatic.
HH produces a specific type of epilepsy owing to a noncortical seizure focus.
Magnetic resonance imaging shows a mass,
that projects from the tuber cinereum or mamillary bodies into the suprasellar or interpeduncular cisterns.
HH follows signal intensity of gray matter on T1-weighted sequences,
is of iso-to-hyperintense T2 signal,
and does not enhance after injection of contrast media.
Hypothalamic astrocytoma may have the same imaging appearance.
Because of its often pedunculated and noncortical location,
transcallosal neurosurgical resection can produce cure with minimal morbidity (48,49).
24. Epidermoid cyst (primarycholesteatoma)( Fig. 34)
Epidermoid cysts arise from normal epithelial cells that are present during neural tube closure.
They are slowly growing lesions.
A frequent imaging enigma is the differentiation between arachnoid cysts and epidermoid tumors,
which have common anatomic distributions and imaging features by CT and conventional MRI.
Both entities have hyposignal on T1-weighted and hypersignal on T2-weighted images,
although epidermoids often have a "dirty" CSF appearance with internal heterogeneity on FLAIR images.
Both are relatively avascular on CT,
MRI,
and angiography.
DWI has been recognized as a convenient and reliable discriminator between these two lesions.
Epidermoids are bright on DWI while Arachnoid cysts predictably follow CSF,
becoming dark on DWI.
The finding of a smooth round lesion mimicking CSF on CT and “conventional" MR images but bright on DWI is diagnostic of epidermoid (5~52).
25. Medulloblastoma ( Fig. 35 ):
Medulloblastoma is a highly malignant neuroepithelial tumor found predominantly in the posterior fossa.
It is the most frequent malignant CNS tumor and one of the most common tumors seen in the posterior fossa in children,
after pilocytic astrocytoma (PA).
The cerebellum is the most frequent location,
and most are located in the vermis.
Lateral locations are more common in older children and adults.
Symptoms are generally brief and include headache,
truncal ataxia,
spasticity,
and sixth nerve palsy.
On noncontrast CT,
These masses are generally hyperdense.
On MRI,
they express relatively lower-signal intensity than the adjacent normal cerebral parenchyma on ADC maps.
This is in contrast with PAs,
which are very bright on ADC maps.
On T1-and T2-weighted images,
both tumors may express variable signal intensities and amounts of enhancement after injection of contrast media.
A diagnosis of medulloblastoma should indicate MR.
imaging of the entire spine to exclude drop metastases (53,54).
26. Brain abscess ( Fig. 36 ):
They may arise from hematogenous dissemination,
meningitis,
or direct extension from the paranasal sinuses or mastoids.
Symptoms are usually abrupt in onset and are related to mass effect from a rapidly expanding mass lesion.
MRI shows a mass with increased central T2 and decreased T1 signal.
As the abscess progresses,
a dark T2 and iso-to-bright T1 signal rim is often seen.
Abundant surrounding vasogenic edema is usually present.
There is ring enhancement of the lesion after injection of contrast media,
which is nonspecific.
The central portions of abscesses are very hyperintense on DWI and dark on ADC maps (thick,
proteinacious pus causes restricted diffusion).
Necrotic metastatic tumors,
conversely,
express central low signal on DWI,
comparable to CSF.
Perfusion imaging can also be used and demonstrate decreased CBV in abscesses,
as opposed to peripherally neoplasms which demonstrate increased CBV (55-57).
27. Creutzfeldt-jakob disease ( Fig. 37)
Creutzfeldt-Jakob disease (CJD) is a rare,
fatal disease categorized generally as a subacute spongiform encephalopathy and arising in sporadic,
familial,
and variant forms.
The cause of the pathology is thought to be abnormal proteins,
that are transmissible.
CJD is characterized clinically by rapidly progressive dementia and generalized myoclonus.
MRI is the single best diagnostic exam in the evaluation for CJD.
FLAIR and DWI are the most helpful sequences.
Imaging in the acute phase shows hyperintense signal on both FLAIR and DWI in the cortex,
striatum,
and thalamus.
Extensive cortical hyperintensity involving both cerebral hemispheres on DWI is essentially pathognomonic for CJD.
In later phases of the disease,
there is atrophy of the cortex and disappearance of the hyperintense signal on DWI (57-59).
28. Mesial temporal sclerosis (MTS) or hippocampal sclerosis( Fig. 38)
It is the most frequent etiology of temporal lobe epilepsy,
which is usually refractory to antiepileptic treatment and therefore managed surgically.
MRI can show volume loss with hyperintensity of the hippocampus on T2-weighted or FLAIR sequences.
Coronal,
thin-section inversion recovery images may be particularly helpful.
Functional MRI is helpful for mapping eloquent cortex,
whereas SPECT and PET show ictal hyperperfusion and interictal hypoperfusion.
MRS demonstrates a decrease in the NAA/Cho+ Cr ratio.,
(60-62).
29. Wernicke's encephalopathy ( Fig. 39)
The classic triad of ophthalmoplegia ,
ataxia,
and changes of the mental status is rarely seen in patients with Wernicke's encephalopathy,
who most frequently present with only altered mental status.
Early treatment (intravenous thiamine) is essential because mortality is >10% in adults.
Because of the vague clinical presentation and frequency of confounding factors such as alcoholism and Korsakoff's psychosis (i.e.,
amnesia and confabulation),
imaging can play an important role in early diagnosis.
Anatomically,
the zones touched in Wernicke's encephalopathy include the medial thalami,
mamillary bodies,
and periaquaductal gray matter.
CT may show hypodensity in these regions,
and occasionally microhemorrhage.
MRl shows symmetric hypersignal on T2-weighted,
DWI,
and FLAIR images.
The injection of contrast media has a limited role as lesions do not enhance in 50% of acute cases.
A late finding is mamillary body atrophy.
Wernicke's encephalopathy is underdiagnosed in the pediatric population,
where the mortality rate is 42 percent.
Children usually do not display the classic triad,
generally have an underlying malignancy,
and are almost always female (63,64)
30. Multiple sclerosis (MS)( Fig. 40)
It is an inflammatory,
autoimmune disease characterized by demyelination and axonal injury with multiple lesions of the CNS separated in both space and time.
Clinically,
it presents most frequently with weakness,
parasthesias,
vertigo,
and visual or urinary disturbances.
The natural history typically manifests as unpredictable relapsing and remitting symptoms.
The principles of MS diagnosis are based on demostrating dissemination of white-matter lesions in space and time.
MR is the most sensitive tool for detecting MS lesions.
MR typically shows abnormalities in hyposignal on T1WI and hypersignal on T2WI in the periventricular white matter,
which are typically oriented perpendicular to the ventricles,
ovoid lesions,
corpus callosum lesions,
and spinal cord involvement. Active lesions enhance after injection of gadolinium and may be bright on DWI,
dedicated coronal sequences are useful for evaluation of the optic nerves,
because optic neuritis may be the first presentation of MS.
McDonald's criteria should be used when assessing patients for MS.
Any three of the four following criteria constitute MR findings compatible with MS: (1) one gadolinium-enhancing lesion or nine T2-hyperintense lesions when there is no enhancing lesion,
(2) with at least one lnfratentorial lesion,
(3) at least one juxta-cortical lesion,
and (4) at least three periventricular lesions.
A spinal cord lesion may be substituted for a brain lesion.
Conforming to the revised criteria; dissemination in space needs at least one T2 lesion in at least two of four locations (juxtacortical,
periventricular,
infratentorial,
and spinal cord) and dissemination in time requires a new T2 lesion on a follow-up scan.
(65,66).