History
- 1961 – Sarles (Fig.
2) et al observed “chronic inflammatory sclerosis of the pancreas” with hypergammaglobulinaemia1

Fig. 2: Dr Henri Sarles - first described chronic pancreatitis with hypergammaglobulinaemia
References: www.histoire L'inferm.fr
- 1989 – Dreiling et al described pancreatic involvement in patients with known autoimmune collagen disease 2
- 1995 – Yoshida et al described a patient with an autoimmune aetiology of chronic pancreatitis in the absence of other another established autoimmune disease and hence defined the concept of autoimmune pancreatitis (AIP) as chronic pancreatitis with respect to specific clinical,
histological and serological findings 3
- 2001 – Hammano et al discovered increased serum IgG4 levels in AIP 4
- Now well recognised as systemic disease in it’s own right
AIP Definition
Fig. 3: Clinical presentation/Histology/Treatment
References: Kamisawa et al. (2012) Recent advances in autoimmune pancreatitis. Front Physio 3:374
Pathological Findings
Two subtypes of AIP
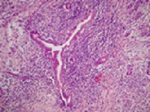
Type1: pancreatic manifestation of IgG4-related disease - lymphoplasmacytic sclerosing pancreatitis (LPSP) (Fig.
4)
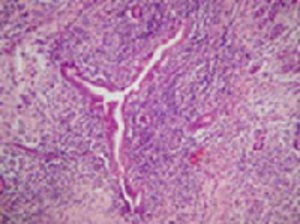
Fig. 4: Type 1 AIP: Medium sized duct showing typical periductal lymphoplasmacytic inflammation and narrowing of the lumen
References: Kloppel G. (2007) Chronic pancreatitis, pseudotumours and other tumour-like lesions. Modern Pathology 20:113-131
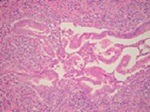
Type 2: pancreas-specific disease - idiopathic duct centric pancreatitis (IDCP) (Fig.
5)
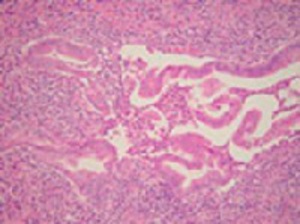
Fig. 5: Type 2 AIP: pancreatic duct showing a granulocytic epithelial lesion (GEL) - destruction of the epithelium by granulocytes
References: Kloppel G. (2007) Chronic pancreatitis, pseudotumours and other tumour-like lesions. Modern Pathology 20:113-131
Type 1 AIP
- Also known as Lymphoplasmacytic Sclerosing Pancreatitis (LPSP)
- Majority of cases in Asia
- Middle aged/elderly males
- Most patients exhibit systemic disease with multi-organ involvement- IgG4 Related Disease
- Affecting pancreas,
biliary tree,
kidneys,
salivary/lacrimal glands,
thyroid,
lungs,
prostate,
retroperitoneum,
GI tract,
CNS,
lymphatics.
Presenting symptoms depend upon organs involved

Fig. 6: Various organs involved
- 10-20% do not have other organ involvement
Major histopathological features:
- Infiltration of IgG4 and plasma cells(>10hpf,
40% IgG4:IgG cells) and CD4+ T-lymphocytes
- Characteristic "storiform" fibrosis
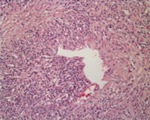
- Obliterative phlebitis (Fig.
7) caused by perivenular infiltration of plasma cells and lymphocytes causing lumen obliteration
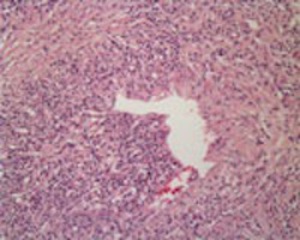
Fig. 7: AIP: Obliterative venulitis
References: Kloppel G. (2007) Chronic pancreatitis, pseudotumours and other tumour-like lesions. Modern Pathology 20:113-131
Other associated histopathological features:
Lumen preserving phlebitis
Increased number of eosinophils6
Type 2 AIP
- Also known as idiopathic duct centric pancreatitis (IDCP) or AIP with granulocytic epithelial lesion (GEL)
- Orginally described in USA and Europe
- No associated elevated serum IgG4 or other organ involvement
- Pancreas specific
- Characteristic granulocytic epithelial lesion causing immune complex mediated destruction of the pancreatic duct
- Complement C3c and IgG deposition at basement membrane of pancreatic ducts and acini7
- Association with ulcerative colitis
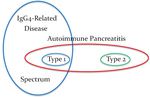
Terminology
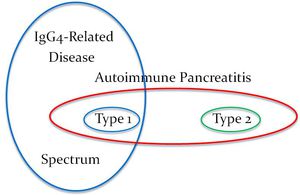
Fig. 8: Schematic representation of AIP terminology
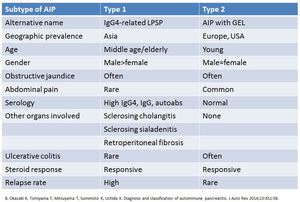
Fig. 9
References: Okazaki K et al. (2014) Diagnosis & classification of autoimmune pancreatitis. J Auto Rev 13:451-58
Diagnostic Criteria for AIP
-
Multiple kinds of organ-specific diagnostic criteria for AIP have been proposed
-
Japan Pancreas Society - 2002,
2006,
2011 - relies on the appearances on cross-sectional imaging and endoscopic pancreatogram for diagnosis
-
Mayo Clinic's HISORt - was developed to include characteristic histological criteria and response to steroid treatment,
to increase the sensitivity to diagnose the wide spectrum of manifestations of AIP
-
Korean (2007),
Asian (2008),
Italian (2003,
2009) and Manheim (2009) have also been proposed
-
International Consensus of Diagnostic Criteria (ICDC) - this has been formulated to incorporate the different practice patterns in the use of diagnostic test and differentiation of the two subtypes
International Consensus of Diagnostic Criteria (ICDC)
- Considers 5 cardinal features of AIP (both Types 1&2)
- Imaging of the pancreatic parenchyma (CT/MRI) and duct (ERP/MRP)
- Serology - IgG4 >x2 normal
- Other organ involvement
- Pancreatic histology
- Response to steroid treatment

Fig. 10: ICDC Original Guidelines of the International Association of Pancreatology
References: Shimosegawa T et al. (2011) International consensus diagnostic criteria for AIP: guidelines of the International Association of Pancreatology. Pancreas 40(3):352-8
- Each feature categorised as Level 1 or Level 2 based on diagnostic reliability
ICDC for Type 1 AIP
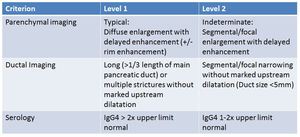
Fig. 11: ICDC
References: Shimosegawa T et al. (2011) International consensus diagnostic criteria for AIP: guidelines of the International Association of Pancreatology. Pancreas 40(3):352-8
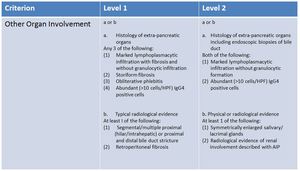
Fig. 12: ICDC
References: Shimosegawa T et al. (2011) International consensus diagnostic criteria for AIP: guidelines of the International Association of Pancreatology. Pancreas 40(3):352-8
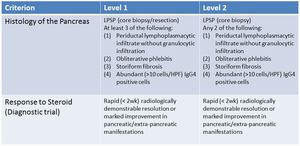
Fig. 13: ICDC
References: Shimosegawa T et al. (2011) International consensus diagnostic criteria for AIP: guidelines of the International Association of Pancreatology. Pancreas 40(3):352-8
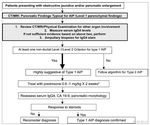
Algorithm to diagnose Type 1 AIP in patients presenting with obstructive jaundice +/- pancreatic enlargement
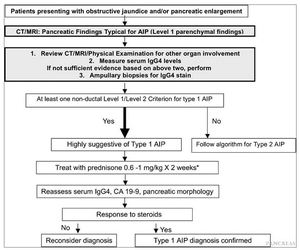
Fig. 14: Type 1 AIP Diagnostic Algorithm
References: Shimosegawa T et al. (2011) International consensus diagnostic criteria for AIP: guidelines of the International Association of Pancreatology. Pancreas 40(3):352-8
ICDC for Type 2 AIP
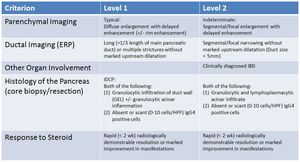
Fig. 15: ICDC for Type 2 AIP
References: Shimosegawa T et al. (2011) International consensus diagnostic criteria for AIP: guidelines of the International Association of Pancreatology. Pancreas 40(3):352-8