Of 1800 CT & MR studies of the head over a period of six months,
five patients were found to have clinical & radiological evidence of cerebral hemiatrophy.
The Computerised Tomographic scans were performed on a 256-slice CT scanner,
while Magnetic Resonance studies were performed on a 1.5 Tesla MR System with multinuclear spectroscopic capabilities using standard head coils (circularly polarized phased array head coil).
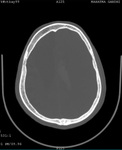
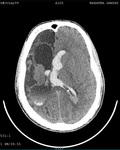
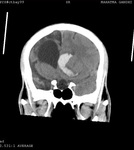
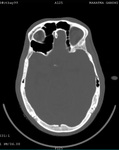
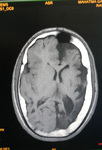
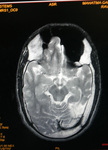
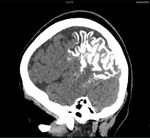
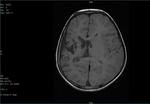
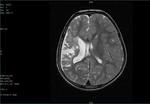
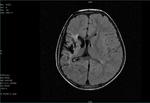
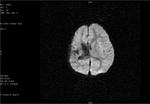
Two cases of Dyke-Davidoff-Masson Syndrome revealed cerebral hemi-atrophy,
leading to dilatation of sulci and cisterns as well as ex-vacuo dilatation of ipsilateral lateral ventricle with compensatory contralateral calvarial thickening and hyper-pneumatization of paranasal air sinuses (Figure 1 to 7).
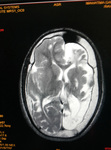
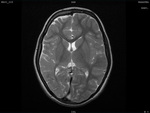
Sturge Weber Syndrome revealed right cerebral hemiatrophy with prominent right choroid plexus and sub-arachnoid spaces,
predominantly involving the fronto-parietal lobes with evidence of tram-track or gyriform calcification pattern in right parietal lobe (Figure 8 to 12).
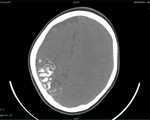
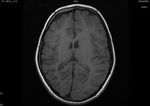
Rasmussen’s Encephalitis revealed diffuse atrophy involving the right cerebral hemisphere mainly in the cortical and sub-cortical regions with ex-vacuo dilatation of right lateral ventricle.
Multiple gliotic-encephalomalacic areas were seen in right fronto-parieto-temporal lobes with persistent cavum septum pellucidum and cavum vergae.
The encephalomalacic area appeared isointense to CSF on all imaging sequences while the gliotic area appeared hyperintense on FLAIR images (Figure 13 to 16).
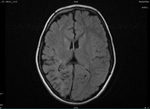
Hemiconvulsion-Hemiplegia-Epilepsy Syndrome revealed small corpus callosum with ipsilateral hemispheric volume loss with shift of midline structures.
CHA is an entity with diverse etiologies and pronounced asymmetry of cerebral hemispheres [17].
It may be a progressive spectrum or emanating from a single event [13].
Half and three-fourths of adult brain sizes are attained during the first and third year of life,
respectively.
With brain growth,
the brain presses outward on the calvarial tables underscoring the gradual enlargement and general shape of the adult head [14].
Identification of compensatory calvarial changes presuppose that cerebral abnormalities are the consequences of an atrophic or hypoplastic process from brain insults that started in early life (usually before 2 years).
These skull changes correlate with the amount of underlying brain atrophy.
These changes include thickening of calvarium with loss of convolutional markings of the inner table and flattening of calvarium.
Overpneumatisation of paranasal sinuses and mastoid air cells lead to elevation of the petrous ridge [9].
(1) Dyke Davidoff Mason Syndrome
In 1933,
Dyke,
Davidoff and Masson described skull radiographic and pneumatoencephalographic changes in a series of nine patients characterized clinically by hemiparesis/hemiplegia,
seizures,
facial-asymmetry,
and mental retardation [6].
The aforementioned are the hallmark clinical presentations of Dyke-Davidoff-Masson syndrome (DDMS).
DDMS is a childhood unicerebral atrophy or hypoplasia with compensatory ipsilateral calvarial hypertrophy.
This infrequent condition is secondary to brain insult in-utero or early childhood period.
Insult could be vascular,
congenital or acquired ischaemic disease,
trauma or inflammation [7,14].
In-utero gestational vascular occlusion involving middle cerebral vascular territory or coactation of mid-aortic arch may decrease carotid arterial flow [15].
In DDMS,
calvarial changes occur only when brain damage is sustained before three years of age but such changes may become visible as early as nine-months after brain damage.
Brain growth failure compels inward redirection of nearby calvarial growth accounting for the compensatory calvarial changes like enlargement of the frontal sinus,
increased width of the diploic space with elevations of the ipsilateral greater wing of sphenoid,
petrous ridge and planum sphenoidale.
CT is preferred to conventional radiography in DDMS as skull plain radiographs may be so subtle and overlooked [14].
MRI features in DDMS include variable extent of unilateral loss of cerebral volume (with or without enlargement of the cortical sulci and perimesencephalic cistern),
hypoplasia/atrophy of the cerebral peduncle,
abnormal myelination and contralateral cerebellar atrophy.
While the later is due to postictal lobar sclerosis,
hypoplasia of the ipsilateral mesencephalon is due to distal axonal degeneration [1,5,14].
(2) Sturge Weber Syndrome
Sturge-Weber syndrome (SWS) or encephalofacial/encephalotrigeminal angiomatosis,
is a rare,
congenital neurocutaneous syndrome accompanied by unilateral facial cutaneous vascular malformation (nevus flammeus or port-wine stain {PWS}) and ipsilateral leptomeningeal angiomatosis [9,10,12].
Paucity of superficial cerebral cortical veins with malformed fibrotic veins usually ipsilateral to the skin lesion is the underlying anomaly [10].
The facial PWS is usually purple to pink in appearance and involves one side of upper face,
including the eye corresponding to ophthalmic divisions of the trigeminal nerve.
Other manifestations include progressive seizures,
ipsilateral glaucoma,
ipsilateral cranial thickening,
contralateral hemiparesis,
hemiatrophy,
hemianopia and mental retardation [9].
Cerebral atrophy in SWS are typically unilateral and confined to the parieto-occipital area,
but occasionally may extend to the entire hemisphere or bilaterally [10,12].
Abnormality of the cerebral parenchyma can be detected from birth in some patients and has a progressive character of atrophy and parenchymatous hyper density of the affected hemisphere,
as well as a decrease in arterial size,
especially during the first decade of life [11].
MRI usually shows evidence of ipsilateral tissue loss with hallmarks of tubular regions of flow void and gyriform hypointensities on T1- and T2-weighted MRI images due to venous collateralization and cortical calcification.
These hallmarks show on CT as “tram-line” or gyriform calcifications usually involving the occipital and parietal lobes underlying the leptomeningeal angiomatosis [12].
The cortical calcification may be as a result of chronic hypoxic injury [10].
Histology has revealed these intracranial lesions are leptomeningeal angiomatosis,
Leptomeningeal thickening,
gyriform calcifications,
neuronal loss,
astrogliosis in underlying brain tissue,
choroidal angioma,
and choroid plexus angioma [20].
Calcification is seen histologically in almost every case,
90% in CT and 50-60% in skull radiographs [9,10].
Bilateral calcifications occurred in 15% of cases with skull thickening seen on the side of more extensive calcifications.
However,
calcifications may be absent or minimal in neonates and infants [20].
MRI with contrast is the preferred imaging modality for the identification of structural brain abnormalities in SWS.
It shows enhancement of the leptomeninges overlying the involved cortex,
reflecting leptomeningeal angiomatosis,
or blood-brain-barrier damage related to chronic cortical ischemia.
Homogeneous enhancement of an enlarged ipsilateral choroid plexus may be seen due to a choroid plexus angioma.
Because of insufficient superficial cortical venous drainage,
collateralization develops in the deep venous system,
leading to enhancement of medullary and subependymal veins ipsilateral to the cortical lesion [20].
MR perfusion weighted imaging (PWI) indicates cerebral hypoperfusion predominantly due to impaired venous drainage,
with only the most severely affected regions in some patients also showing arterial perfusion deficiency [11].
(4) Rasmussen's Encephalitis
Rasmussen's encephalitis (RE) or chronic focal encephalitis was discovered in 1958 by Theodore Rasmussen [1].
It is a rare but severe immune-mediated,
chronic inflammatory,
progressive,
devastating brain disorder [4,11,19].
The onset is in childhood and is characterized by unilateral hemispheric atrophy,
abrupt appearance of focal,
persistent motor seizure activity (epilepsia partialis continua),
followed by hemiplegia and progressive cognitive deterioration [19].
Three features of the epilepsy in RE are polymorphism of seizures,
frequent occurrence and medical intractability [4].
RE is usually uni-cerebral and generally occurs in children under the age of 15 with median age of 6years [1].
Nevertheless,
adolescent and adult patients with milder course exist[4].
Aetiology of RE is unknown but autoimmunity associated with persistent viral infection has been implicated.
Preceding inflammatory episodes are tonsillitis,
upper respiratory tract infection,
and otitis media.
Unihemispherical atrophy results from brain cells inflammations and epilepsy [1,4,14].
RE is a diagnosis of exclusion and typically insidious in onset.
CT and MRI shows unihemispheric lesion.
CT reveals nonspecific atrophy.
MRI demonstrates the progression of RE and may suggest the diagnosis in the early stages,
prior to appearance of neurological deficits.
Apart from atrophy of the head of the caudate nucleus,
MRI may also show associated secondary changes such as atrophy of the contralateral cerebellar hemisphere,
ipsilateral hippocampus and brainstem [1].
There are four recognized stages of RE based on T2 weighted MRI criteria.
These are swelling with hyperintense signal (stage 1); normal volume with hyperintense signal (stage 2); atrophy with hyperintense signal (stage 3); and progressive atrophy and normal signal (stage 4) [1].
Gadolinium enhancement is very rare [12].
(4) Hemiconvulsion-Hemiplegia-Epilepsy Syndrome (HHE)
HHE reveals small corpus callosum with ipsilateral hemispheric volume loss with shift of midline structures.Parenchymal damage results from sustained ictal activity in HHE.