At the Center of Breast and Ovarian Familiar Tumors in Modena,
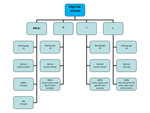
3306 women considered at increased familiar risk for BC were enrolled in a surveillance program from January 1994 to June 2014 and divided into four risk categories depending on familiar history or BRCA status according to Modena Criteria: BRCA mutation carriers (BRCA+),
High,
Intermediated and Slightly increased risk (Fig.1).
Clinical Breast Examination (CBE),
MMG,
US and MR are performed annually by experts radiologists of about 10-20 years of breast imaging experience at our dedicated clinic located at the Department of Radiology.
Clinical and instrumental monitoring begins at 25 years in BRCA+,
while at 30 years in the other groups.
In BRCA+ patients CBE,
MMG and MR are performed annually,
while in High risk and senior groups CBE,
MMG and US are performed every 2 years up to 36 years,
then annually by 40 years at the same place.
In these groups a clinical and instrumental follow-up with CBE and US runs every 6 months for BRCA+,
High and Slightly increased risk women by clinicians of the Center of Breast and Ovarian Familiar Tumors in Modena with at least 10 years breast US-experience.
Slightly increased risk women are subjected to CBE,
MMG and US every 18-24 months,
while the clinical follow-up US is annual (Fig.2).
MMG was conducted with two standard breast projections: one craniocaudal and one angled in a side view (medium-lateral).
Other projections,
compression views and magnifications may be included when necessary.
Breast US study was performed bilaterally exploring all breast glandular sectors by using a linear array transducer at high frequency (frequency of 7-13 MHz) in radial displacement of sensor and also studied retroareolar region and,
when necessary,
axillary recess too.
The average time necessary to perform a US examination was about 15-20 minutes.
All lesions US-detected were documented into two spatial planes,
localized with scanning in all clock positions and then classified according to Breast Imaging Reporting and Data System Ultrasound Criteria (BI-RADS US categories) in order to standardize terminology and clinical management [14,15].
No breast lesions were categorized as 6 BI-RADS US category.
Each suspicious lesion was verified cytologically with US-guided Fine-Needle Aspiration Cytology (FNAC) or histologically with 14-Gauge needle biopsy.