1.
RM protocol
The diagnosis and evaluation of the extension of deep pelvic endometriosis is difficult employing only physical examination and conventional methods.
Exploratory laparoscopy considered as "gold standard" has limitations in assessing the subperitoneal space and under peritoneal adhesions,
which are frequent in this entity and prevent adequate visualization.
MRI due to its spatial resolution and its ability to tissue characterization is a precise technique in evaluating the extension of endometriosis and in detecting its complications.
Therefore,
provides information necessary to establish a map of the disease and plan an adequate surgical treatment.
As preparation,
the patient should be given a cleansing enema the night before MRI realization.
If there are artifacts by intestinal hypermotility a spasmolytic (iv.
or im.) should be administered.
We don't employ routinely vaginal or intestinal labeling with ultrasound gel because usually is not necessary and because it increases disconfort to the patient during the test.
In our center studies were performed on a high-field MRI (1.5 and 3 T,
GE,
Milwaukee) employing multichannel surface coil.
High resolutionT2-weighted series in transversal,
sagittal and coronal planes are acquired.
T1-weighted fat-suppressed series is also obtained,
generally LAVA in transverse plane and sagittal T1 (in phase or LAVA).
Optionally we add a STIR series in coronal plane.
High-resolution T2-weighted sequences provide great anatomical definition and are much more useful in the detection of fibrous lesions.
The combination of T1 and T2 sequences is useful in detection of endometriomas.
T1-weighted sequences with fat suppression are helpful in studies of female pelvis,
allowing differentiate adnexal mass with fat content of those with hemorrhagic content.
They also improve identification of T1 hyperintense structures that do not contain fat,
providing higher sensitivity in detection of small endometriomas or implants with hemorrhagic content.
It is also important to note that the loss of signal on STIR is not specific for fat and affect other lesions with short T1 (hemorrhagic cysts and endometriomas may decrease its signal).
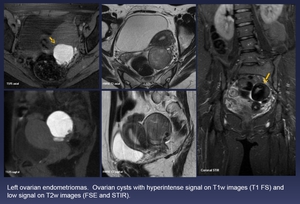
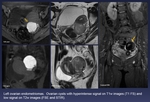
Fig. 2: Left ovarian endometriomas. Ovarian cysts with hyperintense signal on T1w images (T1 FS) and low signal on T2w images (FSE and STIR).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
We do not administer iv.
gadolinium routinely as part of the protocol in our center.
Although it has been described that implants enhances after contrast administration there is also an enhancement of another pelvic structures,
which can lead to confusion and false positives.
The assessment of contrast enhancement is facilitated by subtraction.
The use of contrast and subtraction sequences are useful in case of suspicion of malignant transformation.
2.
Imaging Findings
2.1 Endometriomas:
The ovaries are the most common site of involvement by endometriosis.
In most of revised cases,
endometriomas were observed alone or associated to other manifestations.
The endometriomas or chocolate cysts are ovarian cysts secondary to cyclic bleeding of endometrial tissue in this location.
They have high concentration of protein and blood in different developmental stages.
Therefore,
they characteristically exhibit high signal in T1 weighted sequences,
that not decreases with fat suppression,
and intermediate or low signal in T2 (shading effect).
If there are multiple endometriomas or large endometriomas,
it is common to find a secondary ovarian enlargement.
When endometriomas present large size,
they may contain clots or septa.
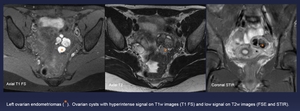
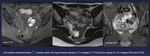
Fig. 3: Left ovarian endometriomas (orange *). Ovarian cysts with hyperintense signal on T1w images (T1 FS) and low signal on T2w images (FSE and STIR).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
Paraovarian adhesions because of small and superficial implants could also be found but are less frequent.
2.2 Superficial lesions or noninvasive peritoneal implants
Superficial endometriotic implants are composed of glands and stromal components inside fibromuscular tissue.
In T2-weighted images superficial implants are seen as ill-defined hypointense areas with variable millimetric hyperintense foci,
according to the presence of glandular component,
while in T1-weighted images hyperintense foci represent the presence of haemorraghic component.
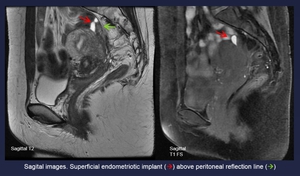
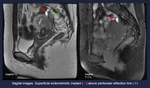
Fig. 4: Sagital images. Superficial endometriotic implant (red arrow) above peritoneal reflection line (Green arrow)
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
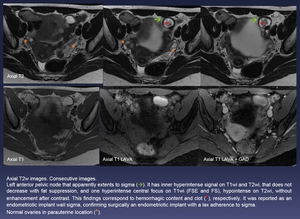
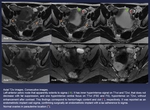
Fig. 5: Axial T2w images. Consecutive images.
Left anterior pelvic node that apparently extents to sigma (green arrow). It has inner hyperintense signal on T1wi and T2wi, that does not decrease with fat suppression, and one hyperintense central focus on T1wi (FSE and FS), hypointense on T2wi, without enhancement after contrast. This findings correspond to hemorrhagic content and clot (red *), respectively. It was reported as an endometriotic implant wall sigma, confirming surgically an endometriotic implant with a lax adherence to sigma.
Normal ovaries in parauterine location (orange *).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
2.3 Deep pelvic endometriosis:
Deep pelvic endometriosis corresponds to subperitoneal implants deeper than 5mm under peritoneal surface,
as mentioned before.
Bleeding episodes produce an inflammatory and desmoplasic reaction with fibromuscular proliferation surrounding the focus of endometriosis,
this leads to nodule formation,
sometimes with disappearance of the glandular component,
persisting only the fibrous one.
Radiologically this changes are observed as nodules,
masses or fibrous thickening of normal structures,
often with irregular borders and/or adhesions,
because of the presence of fibrosis.
They present hypointense signal on T2 and T1 weighted images,
sometimes with hyperintense foci on T1 according to the presence of blood.
Occasionally the predominant disturbance is morphological,
by secondary anatomical distortion to the retraction of structures that occurs.
Depending on its location,
we divide the deep pelvic endometriosis in anterior,
middle and posterior compartment.
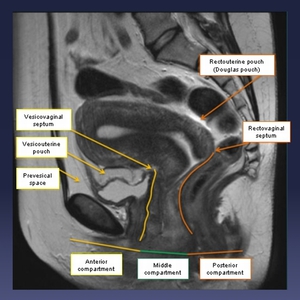
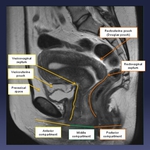
Fig. 6: Depending on its location, we divide the deep pelvic endometriosis in anterior, middle and posterior compartment.
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
A.
Posterior compartment: Includes Douglas pouch,
uterine torus,
uterosacral ligaments,
posterior vaginal fornix,
rectovaginal septum,
rectum and sigmoid.
The retrocervical region including uterosacral ligaments and uterine torus is the most frequent location of deep pelvic endometriosis.
It can be observed thickening and irregularity of ligaments,
and can lead to deformity or even to retroverted uterus by fibrosis.
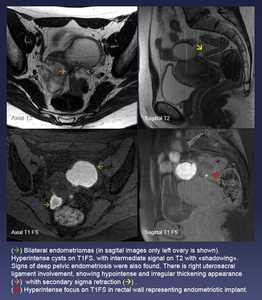
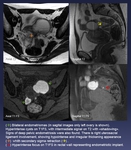
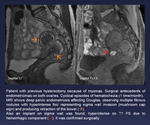
Fig. 7: (Green arrow) Bilateral endometriomas (in sagital images only left ovary is shown). Hyperintense cysts on T1FS, with intermediate signal on T2 with «shadowing».
Signs of deep pelvic endometriosis were also found. There is right uterosacral ligament involvement, showing hypointense and irregular thickening appearance (orange arrow) whith secondary sigma retraction (yellow arrow) .
(red arrow) Hyperintense focus on T1FS in rectal wall representing endometriotic implant.
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
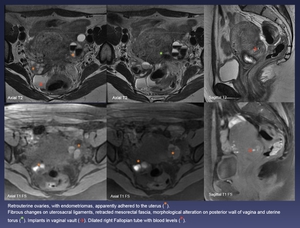
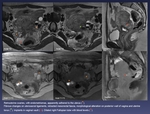
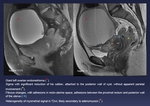
Fig. 8: Retrouterine ovaries, with endometriomas, apparently adhered to the uterus (orange *).
Fibrous changes on uterosacral ligaments, retracted mesorectal fascia, morphological alteration on posterior wall of vagina and uterine torus (green *). Implants in vaginal vault (red arrow). Dilated right Fallopian tube with blood levels (red *).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
Sometimes peritoneal adhesions cause retraction of the ovaries to the retrouterine region standing together between each other; this is known as "kissing ovaries sign".
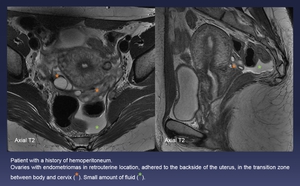
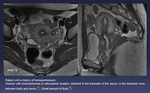
Fig. 9: Patient with a history of hemoperitoneum.
Ovaries with endometriomas in retrouterine location, adhered to the backside of the uterus, in the transition zone between body and cervix (orange *). Small amount of fluid (green *).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
When intestinal involvement exists,
implants initially adhere to the most superficial intestinal layer,
with subsequent muscle invasion and fibromuscular proliferation,
which causes stenosis,
obstruction and/or adhesions to neighboring structures.
Sometimes the fibrous muscle wall thickening of the rectosigmoid adopts a characteristic shape known as "mushroom cap sign".
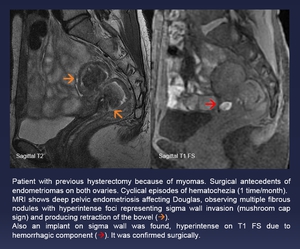
Fig. 10: Patient with previous hysterectomy because of myomas. Surgical antecedents of endometriomas on both ovaries. Cyclical episodes of hematochezia (1 time/month).
MRI shows deep pelvic endometriosis affecting Douglas, observing multiple fibrous nodules with hyperintense foci representing sigma wall invasion (mushroom cap sign) and producing retraction of the bowel (orange arrow).
Also an implant on sigma wall was found, hyperintense on T1 FS due to hemorrhagic component (red arrow). It was confirmed surgically.
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
B.
Middle Compartment: Fallopian tubes,
parametrium,
uterine ligaments (round,
broad) and ovaries.
The most frequent involvement on this compartment is in ovaries (endometriomas),
however,
this involvement is not classified as deep pelvic endometriosis.
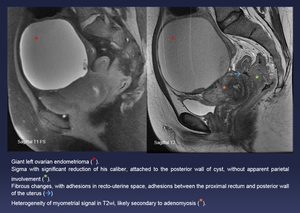
Fig. 11: Giant left ovarian endometrioma (red *).
Sigma with significant reduction of his caliber, attached to the posterior wall of cyst, without apparent parietal involvement (green *).
Fibrous changes, with adhesions in recto-uterine space, adhesions between the proximal rectum and posterior wall of the uterus (blue arrow)
Heterogeneity of myometrial signal in T2wi, likely secondary to adenomyosis (orange *).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
Adenomyosis,
defined as the presence of ectopic endometrial tissue in the myometrium,
is considered as a different medical condition due its characteristics and clinical implications.
In our patients was common finding foci of adenomyosis associated with endometriosis.
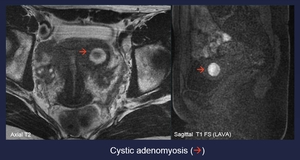
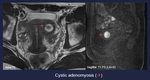
Fig. 12: Cystic adenomyosis (red arrow)
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
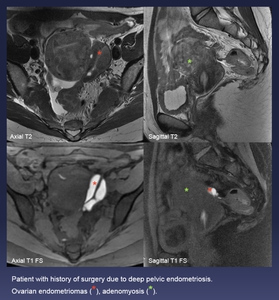
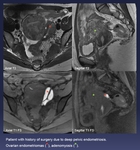
Fig. 13: Patient with history of surgery due to deep pelvic endometriosis.
Ovarian endometriomas (red *), adenomyosis (green *).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
Involvement of the fallopian tubes is associated with infertility and most often ocurrs in the subserosal layer.
It was the next area affected in frequency in our patients,
finding tubal dilation with hydrosalpinx,
and sometimes blood levels.
The presence of hematosalpinx although nonspecific is suggestive of endometriosis and occasionally it has been described as the only finding in MRI.
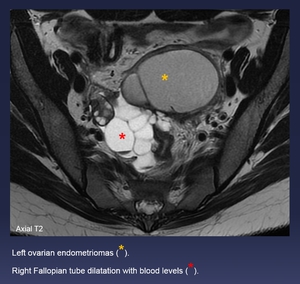
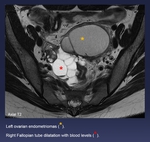
Fig. 14: Left ovarian endometriomas (yellow *).
Right Fallopian tube dilatation with blood levels (red *).
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
Uterine ligaments may be affected presenting thickening and nodularity.
C.
Anterior compartment: Vesicouterine space,
vesico-vaginal septum,
bladder and ureter.
The bladder is usually described as the most frequently affected organ in this compartment,
although it was uncommon in our patients.
It is generally described a serosal involvement,
with subsequent infiltration of the muscular layer,
and protrusion into the lumen in the more advanced cases,
as explained before for the intestinal involvement.
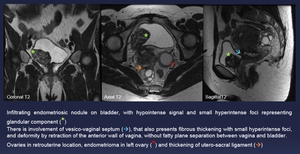
Fig. 15: Infiltrating endometriosic nodule on bladder, with hypointense signal and small hyperintense foci representing glandular component (green *)
There is involvement of vesico-vaginal septum (blue arrow), that also presents fibrous thickening with small hyperintense foci, and deformity by retraction of the anterior wall of vagina, without fatty plane separation between vagina and bladder.
Ovaries in retrouterine location, endometrioma in left ovary (red *) and thickening of utero-sacral ligament (orange arrow)
References: Department of Radiology. LluĂs AlcanyĂs Hospital. XĂ tiva/ES
The ureteral involvement when it occurs,
usually represents implant on adventitia which secondarily produces hyperplasia and fibrosis with stenosis of affected segment.
Stenosis can conduce to ureterohydronephrosis and lead to renal impairment.
It was also observed in our patients associated to other manifestations.
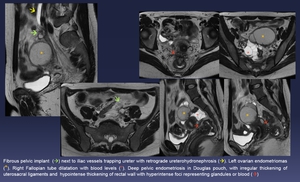
Fig. 16:
Fibrous pelvic implant (green arrow) next to iliac vessels trapping ureter with retrograde ureterohydronephrosis (yellow arrow). Left ovarian endometriomas (yellow *). Right Fallopian tube dilatation with blood levels (red *). Deep pelvic endometriosis in Douglas pouch, with irregular thickening of uterosacral ligaments and hypointense thickening of rectal wall with hyperintense foci representing glandules or blood (red arrow)
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
2.4.
Carcinomas related to endometriosis
Although malignant transformation of endometriosis is a rare complication,
women with endometriosis have a higher risk compared to the general population of developing endometrioid ovarian carcinoma and clear cell ovarian carcinoma.
In two of our patients carcinomas associated with endometriosis have been observed,
one of endometrioid subtype and one of clear cell subtype,
although we don't have MRI of the second case.
The MRI characteristics suggestive of malignant endometriomas include increase in size,
increased T2 signal and development of mural nodules that enhance with contrast.
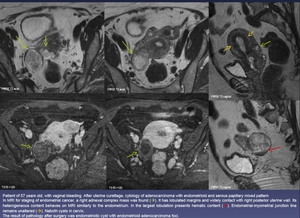
Fig. 17: Patient of 57 years old, with vaginal bleeding. After uterine curettage, cytology of adenocarcinoma with endometrioid and serous papillary mixed pattern
In MRI for staging of endometrial cancer, a right adnexal complex mass was found (green arrow). It has lobulated margins and widely contact with right posterior uterine wall. Its heterogeneous content behaves on MRI similarly to the endometrium. In the largest lobulation presents hematic content (red arrow). Endometrial-myometrial junction line remains unaltered (yellow arrow). Naboth cysts in cervix.
The result of pathology after surgery was endometriotic cyst with endometrioid adenocarcinoma foci.
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
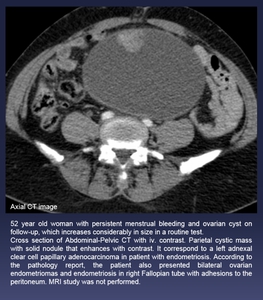
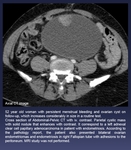
Fig. 18: 52 year old woman with persistent menstrual bleeding and ovarian cyst on follow-up, which increases considerably in size in a routine test.
Cross section of Abdominal-Pelvic CT with iv. contrast. Parietal cystic mass with solid nodule that enhances with contrast. It correspond to a left adnexal clear cell papillary adenocarcinoma in patient with endometriosis. According to the pathology report, the patient also presented bilateral ovarian endometriomas and endometriosis in right Fallopian tube with adhesions to the peritoneum. MRI study was not performed.
References: Department of Radiology. La Fe University and Polytechnic Hospital. Valencia/ES
3.
Surgical Correlation
In all revised patients,
MRI study was requested prior to surgical intervention,
and all of the observed findings were confirmed surgically.
Therefore MRI is an effective technique for diagnosis and staging of pelvic endometriosis.