Patients and Enrollment Criteria
The study protocol was approved by the institutional review board of Samsung Medical Center,
and all patients gave written informed consent before being enrolled.
From June 2014 to September 2014,
20 patients who referred to our department for planning US of biopsy or radiofrequency ablation (RFA) of focal hepatic lesions were enrolled in our study.
The inclusion criteria were as follows: a) patients referred for planning US of RFA for hepatocellular carcinoma (HCC) or for US-guided biopsy of focal hepatic lesions; b) patients with focal hepatic nodules with 1 – 3 cm in diameter detected at contrast-enhanced MRI with gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid (Gd-EOB-DTPA); and c) patients with Child-Pugh score A.
The diagnosis of HCC was based on the typical imaging features (arterial enhancement followed by portal or delayed washout) according to the American Association for the Study of Liver Disease (AASLD) guideline.
17
Exclusion criteria were as follows: a) patients expected to have poor sonographic window due to abundant omental fat (thickness more than 0.5 cm) or the colon surrounding the right liver,
b) patients with previous history of right hemihepatectomy,
c) aged less than 20 years old or more than 80 years old,
or d) patients who denied to participate in our study.
When each examiner performed total ten cases,
the study was terminated.
The patient population included 19 men and 1 woman (mean age ± SD,
58.9 ± 11.8 years; age range,
34 –77 years).
Eight patients were included for planning US of RFA and the remaining 12 patients were included for biopsy of focal hepatic lesion (metastasis from rectal cancer,
n=3; metastasis from gastric cancer,
n=2; metastasis from pancreatic neuroendocrine tumor,
n=2; HCC,
n=2,
metastasis from lung cancer,
n=1; eosinophilic abscess,
n=1; and hepatic involvement of leukemia,
n=1).
Nine patients had liver cirrhosis due to hepatitis B viral infection and the other 11 patients had normal liver.
All 20 patients had normal liver function (Child-Pugh A class).
The diameter of the target lesions were 1.8 ± 0.6 cm (mean ± SD) and their locations were as follows: segment 5 (n=8),
segment 8 (n=6),
segment 6 (n=3),
segment 7 (n=2),
and segment 4 (n=1).
Operators
Two radiologists participated in this study to perform fusion imaging.
One (A.Y.K) was a less-experienced radiologist in the first year of fellowship-training with about 50 cases of US-guided biopsy but less than 20 cases of fusion imaging-guided procedures,
at the starting point of our study.
The other (M.W.L) was an expert radiologist with 10 years of experience in abdominal intervention (more than 1,500 cases of US-guided biopsy or RFA of focal hepatic lesions,
including more than 500 cases under fusion imaging guidance).
Each examiner performed more than 10 training sessions of both manual and automatic image fusion before enrolling patients in our study.
Image Fusion Technique
Equipment
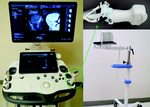
A RS80A US system (Samsung Medison,
Seoul/Korea),
capable of fusion imaging (S-Fusion,
Samsung Medison,
Seoul/Korea) was coupled with a magnetic field generator.
Two electromagnetic position sensors were connected with a position sensing unit (three-dimensional guidance driveBAY tracker; Ascension Technology Corporation) and were attached on a convex-array CA1–7A transducer through a bracket (Fig. 1).
Manual Image Fusion
Before image fusion,
Digital Imaging Communications in Medicine data of up to 6 sequences of MR images were uploaded to the US machine.
Among the 6 sequences,
20 minutes hepatobiliary phase was selected as a fused imaging sequence because the target lesion as well as landmark hepatic vessels,
including the portal and hepatic veins,
were relatively well visualized in this phase.
After image fusion,
the hepatobiliary phase image can be switched to other sequences of MR images.
Each consecutive patient who met the inclusion criteria assigned randomly to either the less-experienced or expert radiologist.
During the study period,
the order of image fusion method (manual method followed by automatic method or vice versa) was alternatively changed at each examination to minimize bias.
Manual image fusion consists of orientation lock and point lock.
For the orientation lock,
a radiologist usually places an US transducer in the sagittal plane to let the US system know the direction of US transducer relative to the patient’s position.
Although an US transducer can be placed on any location near a patient,
usually it was placed on top of the solar plexus (junction between sternum and xiphoid process).
Then,
the radiologist performs the point lock a few times by marking corresponding anatomic landmarks (i.e.,
cysts,
calcifications,
vessel bifurcations or target lesion) within the liver between real-time US and MR images at the end expiratory phase of the patient’s breathing.
For the initial point lock,
any plane of US scans can be used.
That is,
not only orthogonal axial,
sagittal,
or coronal plane; but also oblique plane such as intercostal scan can be used for the initial point lock.
On the other hand,
corresponding MR images are always displayed in orthogonal axial,
sagittal,
or coronal plane.
Therefore,
if a target lesion is conspicuous enough to be easily detected with US image,
the center of the target lesion can be marked at any US plane including oblique plane.
When the target lesion cannot be easily localized on US image due to its small size or many other focal hepatic lesions,
we usually use orthogonal axial,
sagittal,
or coronal plane at US since it is easier to correlate both real-time US and MR images in these planes.
Among them,
we prefer the coronal plane for the initial point lock where both inferior vena cava (IVC) and right hepatic vein are visualized well as anatomic landmarks and the vessel bifurcation site was used for the initial point lock.
8 Then,
additional point locks is performed to refine the registration near the target lesion by marking corresponding anatomic landmarks on the real-time US and fused MR images.
This step can be repeated around the target lesion,
up to maximum five times as much as it is needed and the last point lock should be performed at the center of the target lesion.
Automatic Image Fusion
It also requires aforementioned orientation lock.
Unlike manual image fusion,
orientation lock in the automatic method should be always performed on the top of the solar plexus.
Then,
intrahepatic IVC is marked on the uploaded hepatobiliary phase MR image to notify the system the location of the IVC.
Hence,
the automatic image fusion is named one-click registration.
Then,
the radiologists select a right intercostal space in which an US scan volume can cover both the liver dome and intrahepatic IVC.
After positioning the transducer at the optimal intercostal space,
a 3-dimensional (D) US volume is obtained by sweeping the liver of the patients in the end expiratory breath-holding state with an US transducer.
Hence,
this method is also called sweeping auto-registration.
It takes about 10 seconds for the system to execute the calculation.
After the initial registration is completed,
initially fused real-time US image and MR image are displayed simultaneously on a split-screen display.
Then,
point locks are added to refine the registration using the aforementioned method.
During the study period,
the automatic image fusion method was updated once from version 1.1 to version 1.2 to reflect the improvement in the diaphragm extraction from the 3D US volume.
This updated version of automatic image fusion was applied in the latter cases of the study populations.
Assessment of Registration Error and Time Required for Image Fusion
After image fusion,
the radiologists graded and recorded the quality of image fusion by comparing and correlating anatomic structures around a target lesion between a real-time US image and a fused MR image using a 4-point scale: 1,
poor; 2,
fair; 3,
good; and 4,
excellent.
Grading of the quality of image fusion depended on the individual radiologist’s judgment at the time of US examination.
After image fusion,
the radiologists were also asked to obtain 3D volume data by sweeping the liver containing the target lesion.
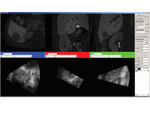
For the registration error assessment,
the 3D US volume data should be aligned to MR data through the previously saved registration information in image fusion.
In both modality,
we selected a pair of corresponding points in the target lesion and then 3D Euclidean distance was automatically calculated between the two selected points in an in-house software (Accuracy Measurement Tool; version 1.0) (Fig. 2).
Total three pairs of corresponding points were used to calculate the average value of registration error.
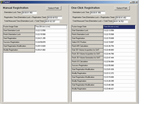
Time required for image fusion was automatically recorded in the US machine and was calculated using an in-house software (Elapse Time Calculation Tool; version 1.0) (Fig. 3).
It was defined as the sum of the elapse time during orientation lock and the time interval between starting registration and the last point lock.
Statistical Analysis
The quality of image fusion,
the number of point lock,
registration error of image fusion,
and time required for image fusion were compared between manual and automatic image fusion according to the level of operator’s experience using the Wilcoxon signed-rank test.
These variables were also compared between the two radiologists using Mann-Whitney U test.
Time required for initial image fusion immediately after sweeping the liver in the automatic method was compared between the two radiologists using Mann-Whitney U test.
P values less than 0.05 were considered to indicate a statistically significant difference.
Since automatic image fusion was updated once during the study period,
we also compared registration error of initial image fusion immediately after sweeping the liver with an US transducer between version 1.1 and version 1.2 using two-way ANOVA.