Ultrasound
Pelvic ultrasound,
both abdominal and transvaginal is useful for the initial evaluation of infertility. It provides information on the echogenicity and size of the uterus,
yielding information about the anatomy.
US can detect uterine anatomical abnormalities,
which can be further evaluated with MRI.
Fibroids or leiomyoma may be detected on ultrasound.
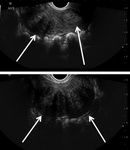
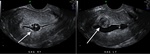
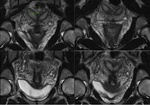
Figure 1 illustrates two large hypoechoic lesions in the fundus and body of the uterus,
consistent with intramural fibroids.
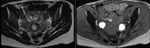
Structural information about the ovaries can be obtained,
including sonographic evidence of polycystic ovarian syndrome (PCOS).
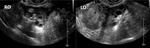
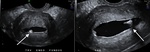
The classic appearance on US in PCOS is enlarged ovaries,
with echogenic stroma and multiple peripherally placed follicular cysts.
Figure 2 illustrates ovaries with multiple small peripherally located cysts.
However,
it must be noted that patients with these features may not have a diagnosis of PCOS and patients with PCOS may not exhibit these sonographic features.
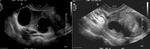
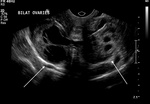
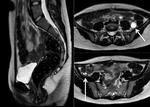
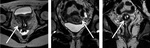
Endometriosis is defined as the presence of endometrial glands outside the uterus.
Evidence of endometriosis can be detected at ultrasound such as haemorragic cysts,
called endometriomas or kissing ovaries Figure 3 and 4.
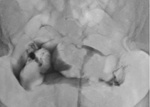
Hysterosalpingogram:
This is performed by placing a small sterile catheter through the cervix into the endometrial cavity.
Radio-opaque contrast material is injected into the endometrial cavity under direct fluoroscopic observation.
Contrast medium is hand injected slowly,
with close observation of the fallopian tubes looking for active spillage of contrast material.
HSG contraindications include active pelvic inflammatory disease and pregnancy.
The fallopian tube appears as a very thin,
curvilinear structure on HSG that gradually widens in the ampullary portion and HSG easily depicts the course,
size,
and contour as well as patency of the tubes.
Tubal spasm is a cause of a false positive tubal occlusion and an antispasmodic such as glucagon is usually administered to reduce it.
Peritubal abnormalities due to pelvic adhesions or endometriosis may be detected.
HSG is useful for diagnosing endometrial and uterine abnormalities such as adhesions,
polyps,
fibroids,
and müllerian duct anomalies by detecting filling defects or contour irregularities.
It is useful for evaluation of tubal abnormalities and occlusion.
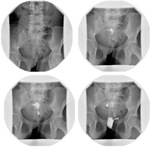
An example of a T-shaped uterus is illustrated in Figure 5.
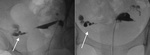
Tubual occlusion at HSG with no flow into the fallopian tubes and filling of lymphatics on delayed imaging is shown in Figure 6.
Tubal abnormalities such as hydrosalpinx can be detected on HSG.
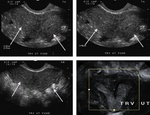
An example of this is in Figure 7 where the left fallopian tube is widely patent but there is a hydrosalpinx of the right tube.
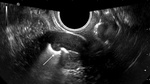
HyCosy and hysterosongram
This is performed in a manner similar to HSG but it does not involve the use of ionizing radiation.
Images of the endometrium are obtained transvaginally.
The endometrial cavity is distended with echogenic contrast material and the fallopian tubes are assessed for patency.
Figure 8 depicts echogenic contrast material in the right fallopian tube with peritoneal spillage.
It is useful for detecting tubal occlusion as a cause for infertility.
Intrauterine filling defects observed at HSG are best evaluated with hysterographic US,
which can help confirm the presence and characteristics of uterine synechiae,
endometrial polyps,
and submucosal fiborids.
Figure 9 are static images from a hysterosonogram illustrating a fibroid with minimal extension into the endometrial cavity.
An endometrial polyp evaluated with hysterosonogram is depicted in Figure 10.
MRI
Magnetic resonance remains the imaging modality of choice for the detection and characterization of endometriosis.
While pelvic US may be helpful for further evaluation of uterine contour abnormalities,
MR imaging is especially useful for differentiating between adenomyosis and uterine fibroids.
MR imaging provides optimal and accurate characterization of müllerian duct anomalies.
Müllerian duct abnormalities can be divided in seven classes.
Class I,
uterine agenesis and hypoplasia,
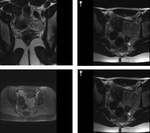
is illustrated in Figure 11,
where an 18 year old girl presented with primary amenorrhoea and a uterus could not be detected on US.
MR confirms the absence of the uterus,
cervix and vagina.
Two normal ovaries are identified lateral to the psoas muscles in a superior location.
Class II,
Unicornuate uterus includes a laterally placed uterine horn with a single fallopian tube.
Class III is Uterus Didelphys is characterized by two widely divergent uterine horns and two cervices.
Figure 12 includes axial and coronal images depicting two uterine horns and 2 cervices.
Bicornuate uterus (Class IV) is due to incomplete fusion of the müllerian ducts,
with two asymmetric but divergent horns.
In bicornuate bicollis uterus there are two cervices.
This can be differentiated from a uterus didelphys by increased myometrial fusion between the uterine horns inferiorly.
The MR features of a bicornuate bicollis uterus are shown in Figure 13,
where there are 2 uterine cavities with divergent uterine horns and 2 cervices.
A bicornuate unicollis uterus is illustrated in Figure 14,
where are 2 divergent uterine horns and a single cervical canal.
Septate uterus (Class V) is the most common anatomical uterine abnormality.
It is similar to bicornuate uterus but has a normal convex,
flat,
or minimally concave (< 1-cm-deep) external fundal contour.
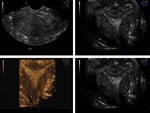
Figure 15 is a TVUS illustrating splaying of the endometrial cavity at the fundus,
with a smooth uterine fundus with minimal superior indentation.
In Figure 16,
an MR of the uterine,
there are divergent uterine horns with an intercornual distance approximately 3 cm.
There is slight concave outer fundal contour (<1cm).
An arcuate uterus (Class VI) is the mildest form of uterine anomaly,
where there is near complete septal resorption resulting in a shallow,
smooth,
broad-based impression on the uterine cavity.
Figure 17 illustrates an arcuate uterus on US where is a normal fundal contour with minimal indentation of the fundal endometrial canal.
Class VII includes DES abnormalities which are best depicted on HSG (Figure 5).
MR imaging features of endometriosis include cystic masses with internal high signal intensity on T1-weighted images and low signal intensity on T2-weighted images.
Other high- signal-intensity features seen on T1-weighted images may include hydrosalpinx (which may include blood products) and endometrial implants.
An example of endometriosis on MR is highlighted in Figure 18,
where there are bilateral well-defined,
thick walled pelvic lesions which display high signal on T1,
low signal on T2 and do not enhance post contrast.
At MR imaging,
the ovaries in patients with polycystic ovary syndrome classi- cally demonstrate a low-signal-intensity central stroma surrounded by small peripheral cysts on T2-weighted images.
An example of this is illustrated in Figure 19.
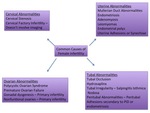
Common Causes of Female Infertility (Figure 20)
Uterine Abnormalities
Endometriosis (Figure 3,4,
18)
Endometrial polyps (Figure 10)
Uterine adhesions or synechiae
Adenomyosis
Leiomyomas (Figure 1,9)
Anatomical Abnormalities
Class I – Uterine agenesis and hypoplasia (Figure 11)
Class II – Unicornuate uterus
Class III – Uterus Didelphys (Figure 12)
Class IV – Bicornuate uterus
Bicornuate bicollis (Figure 13)
Bicornuate unicollis (Figure 14)
Class V – Septate Uterus (Figure 15,16)
Class VII – Arcuate Uterus (Figure 17)
Class VII – DES related uterine abnormalities (Figure 5)
Cervical Abnormalities
Cervical Stenosis
Cervical Factor Infertility – Doesn’t involve imaging
Ovarian Abnormalities
Polycystic ovarian syndrome (Figure 2,
19)
Gonadal dysgenesis - Primary
Nonfuctional ovaries - Primary
Premature ovarian failure
Tubal Abnormalities
Tubal Occlusion (Figure 6)
Spasm
Infection
Prior Surgery
Hydrosaplinx (Figure 7)
Tubal irregularity
Salpingitis isthmica nodasa
Peritubal abnormalities
Peritubal adhesions