The most frequent pancreatic cystic lesions are: pseudocysts,
Serous cystadenomas (microcystic type),
mucinous cysts and solid pseudopapillary tumor.
There are other,
less frequent,
pancreatic cystic lesions such as: cystic endocrine tumors,
cystic pancreatic adenocarcinoma,
cystic metastasis,
cystic teratomas and lymphangiomas.
In this EPOS we will only discuss the most frequent pancreatic lesions.
Most frequent pancreatic cystic lesions:
Pancreatic pseudocysts
Pseudocysts are a complication of interstitial edematous pancreatitis.
They are filled with pancreatic juices (rich in amylase and lipase) surrounded by a wall formed by granulation tissue.
Pseudocysts have a communication with pancreatic ducts.
These cystic formations arise from acute peripancreatic fluid collections,
taking about four weeks to achieve that well-defined fluid collection appearance that we are used to recognize (four weeks being the time necessary for the wall to be formed).
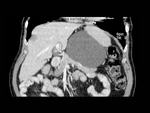
In CT scans ( Fig. 1 ) they appear as homogeneous collections with fluid attenuation,
round or oval-shaped,
with a well defined wall that can look like a thick rim.
Pseudocysts often occur adjacent to the pancreas and don’t have a non-fluid component.
MRI could be necessary to exclude the presence of necrotic tissue within the pseudocyst (up until 5% is acceptable).
They have an homogeneous hypo-signal in T1-weighted images and also homogeneous hyper-intensity in T2-weighted images.
Necrotic/proteic or blood debris have hyper-signal in T1-weighted images.
Serous cystadenomas
These are also called “grandmother’s tumor”,
as they often (> 80%) affect women with ages around 60-70 years old.
They’re only 1% to 2% of all exocrine pancreas’ tumors.
There are three types of serous cystadenomas,
classified according to number and dimensions of the cysts: microcystic,
oligocystic and unilocular.
The most frequent type is the microcystic which has imaging hallmarks.
It has multiple cysts (> 6) with less than 2 cm each - which gives the lesion a lobulated contour -,
that are distributed around a fibrous core (central scar) in a sunburst pattern,
with various septa coming out of the center.
This distribution of the multiple cysts give microcystic serous cystadenomas a sponge or irregular honey- comb appearance.
The fibrous core can have calcifications.
However this fibrous scar is present only in 30% of the cases and calcifications in 18%.
The other types of serous cystadenoma are rare and it’s differentiation from other cystic tumors can be challenging,
as will be described further ahead.
In the oligocystic type there are less than 6 cysts that have 1-2 cm in length (but can be much larger),
there is no central scar and the septa usually have a irregular distribution.
Even rarely serous cystadenomas can be unilocular (unilocular type).
Whatever the type,
these cysts are filled with serous fluid and rarely cause bleeding.
Serous cystadenomas don’t have any communication with the pancreatic ductal system.
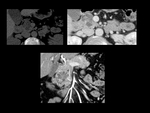
In CT microcystic serous cystadenomas ( Fig. 2 ) have a lobulated contour and are hypodense.
When present,
calcifications are in the core of the lesion and have a punctiform hyperdense appearance,
with a stellate pattern.
After administration of iodinated contrast material the septa and fibrous portions of the lesions enhance,
specially in the latter scans.
CT is a great imaging method for detection of inner calcifications and,
if the cysts are large enough,
to assure their fluid content.
However calcifications aren’t always present and the cysts are rather small.
In these lesions MRI has an important role.
MRI will identify the cystic component as well as depict the internal architecture and to exclude any communication with pancreatic ducts.
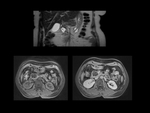
Cysts will be hyperintense and septa will be hypointense comparing to the adjacent pancreatic tissue in T2-weighted images ( Fig. 3 ).
In T1-weighted images cysts are hypointense but may have areas of hyperintensity if there has been previous intracystic hemorrhage.
Core calcifications are very difficult to observe,
but if present have low signal intensity in both T1 and T2-weighted images.
After gadolinium administration there will be enhancement of the septa and fibrous core in delayed phases of imaging,
just like in CT scans.
In conclusion CT and MRI complete each other in an accurate characterization of serous cystadenomas,
as calcifications will better depicted in CT scans and the cystic component is better detailed by MRI.
These tumors don’t have a malignant nor pre-malignant features and are,
until certain sizes,
rather indolent.
When they have 4 cm or more in length usually become symptomatic (usually by compressing adjacent structures) and have a rapid growth rate.
In some institutions when they reach 4 cm in length are surgically removed,
but this way of proceeding is not consensual.
Mucinous cysts:
This group of pancreatic cystic lesions include lesions with different histological background but they share their mucinous content.
They have multiple classifications which are often based on histologic criteria and their imaging correlation is many times difficult.
In a pragmatic,
simplistic point of view we decided to divide them into:
- Mucinous cystadenomas
- Cystadenocarcinomas
- IPMN
This is often called “the mother's tumor” as it often affect women in their 50’s.
We must consider it whenever we are confronted with a cystic lesion in the pancreatic tail in a perimenopausal woman.
These tumors are formed by ovarian-like epithelium being almost exclusive (99,7%) of the female gender.
They often occur in the pancreatic body and tail (95%).
Pancreatic mucinous cystadenoma are uni or multilocular lesions composed by large cysts with a mean size of 6 to 11 cm,
with may have internal debris.
They have septa separating the cysts.
In CT they are well defined,
fluid-filled lesions (hypodense).
After administration of iodinated contrast material there is a wall enhancement (capsule) as well as septal (if present) enhancement.
In MRI these cystic lesions have high signal in T2-weighted images surrounded by a hypointense capsule.
In T1-weighted images,
if we have a protein-rich cystic component it can be iso or even slightly hyperintense.
After gadolinium administration there is septal enhancement.
Like serous cystadenomas these tumors do not show any communication with the pancreatic ductal system.
These lesions are considered pre-malignant as they can have a malignant transformation into mucinous cystadenocarcinomas.
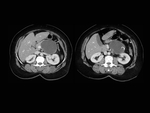
If there are peripheral/septal calcifications,
thickened and nodular walls/septa within the same lesion there is a predictive positive value of it being a malignant tumor of 94,5% ( Fig. 4 ).
If only one of these three features are observed in a mucinous cyst they are worrisome,
but don’t have the same predictive positive value of being a malignant lesion as all three features in the same lesion.
- Intraductal papillary mucinous neoplasms (IPMN):
This is a recently described entity and although the names are similar,
that they look much alike and that both of them are mucin producing tumors,
from an pathologically point of view are completely different from mucinous cystadenomas which we just described above.
IPMNs are the transformation of pancreatic ductal epithelium into mucin producing cells - with papillary projections observed in pathology studies.
As we mentioned earlier these are mucin producing tumors and their imaging hallmark is that they show communication with the pancreatic ductal system.
IPMNs have a male predilection,
with a mean age of 65 - so they are often called “the grandfather’s tumor”.
They are classified in 3 different groups,
according to their relationship with the pancreatic ducts:
- main branch;
- side branch;
- combined.
In CT studies the main branch lesions ( Fig. 5 ) show a segmental or diffuse dilatation of the main pancreatic duct.
A discrete lesion may or may not be observed.
Enhancement is generally only visualized in lesions with nodular foci and it’s a malignant feature.
CT scans are also useful to depict microcalcifications,
which also are a malignant feature.
A side-branch IPMN at CT is a pleomorphic,
hypotenuse lesion,
adjacent to the pancreatic duct.
Its classical site is the pancreatic uncinate process.
There isn’t a mass pancreatic duct ectasia and its communication with the side branch ducts is hard to identify.
CT as a limited role in these lesions’ characterization and MRCP is often mandatory.
In MRCP main-duct IPMNs show an ectasia of the main pancreatic duct and,
just like in CT,
a discrete lesion may or may not be observed - as a nodular enhancing (after gadolinium administration) area that provokes an irregularity of the main pancreatic duct wall.
Side-branch IPMNs,
in MRI ( Fig. 6 ),
show dilatation of the side-branches with high signal in T2-weighted images and low signal in T1-weighted images.
They can have an honeycombing pattern or appear as unilocular or multilocular lesions.
It’s mandatory to assess its communication with the pancreatic side-branches that allow us to make a confident distinction between them and mucinous or serous cystadenomas.
Combined-type IPMN show dilatation of the main pancreatic duct as well as side-branches dilatation.
The risk of a malignant transformation in a main-branch IPMN is 63% at 5 years and for a side-branch IPMN is 15% (also at 5 years).
Risk factors for malignancy depicted on imaging include:
- Main duct width greater than 5 mm;
- increasing size of side-branch lesions;
- side-branch lesions larger than 3 cm;
- presence of calcifications;
- common bile duct dilatation;
- thick septations,
- mural nodules.
- Mucinous cystadenocarcinomas:
Mucinous cystadenocarcinomas ( Fig. 4 ) is pathologically defined as the presence of carcinomatous elements within a mucinous cyst surrounding ovarian-like stroma.
It usually affects women with a greater age than those who have mucinous cystadenomas.
They occur as cystic pancreatic lesions that are large and complex with solid wall nodular areas.
When mucinous cystadenocarcinomas are diagnosed they often have associated hepatic metastasis and locoregional adenopathies.
Solid pseudopapillary tumor (SPT)
Also known as “the daughter’s tumor”,
since it most often affects women (9:1) with 20 to 30 years old.
It’s a rare low-grade malignant tumor with an excellent prognosis after surgical resection.
Rarely,
dissemination has been described and,
if present,
is to the liver,
omentum and peritoneum.
Their symptoms occur due to compressive effect on other structures: epigastric mass sensation and abdominal pain.
SPT are usually large,
encapsulated large masses with a slow growth rate.
The capsule is formed by compressed pancreatic tissue or reactive fibrous tissue.
They tent to displace surrounding structures rather than invade them,
so rarely obstruction of biliary ducts can be observed.
SPT can show peripheral calcification up to 30% of the times.
As SPN have a vascular friability internal hemorrhage and cystic degeneration are hallmarks of these lesions.
So in CT scans ( Fig. 7 ) they will have an heterogeneous appearance as solid well encapsulated masses with cystic areas.
After contrast administration SPN usually have an heterogeneous peripheral enhancement in arterial phase with a progressive enhancement of the solid components in next phases.
At MRI SPN are also heterogeneous lesions,
with high signal in T2-weighted images of its cystic components and in T1-weighted imagens its hemorrhagic component will have a high signal.
The enhancing patter is similar to the one observed at CT.
Although SPN are low-grade lesions they should be surgically resected as they can reach large sizes and have compressive effect in the surrounding structures.
Cystic lesions classification
There are morphological classifications of pancreatic cystic lesions that are intended to help us reach a diagnosis.
One (of the most known) classification divides these lesions in 4 categories:
- Unilocular lesions: pseudocysts,
mucinous cystadenomas; lymphoepithelial cysts; small IPMNs; small serous cystadenomas;
- Microcystic lesions: serous cystadenomas; lymphoepithelial cysts;
- Macrocystic lesions: mucinous cystic neoplasms; oligocystic serous cystadenomas; IPMNs;
- cystic lesions with solid component: solid pseudopapillary tumors; cystic neuroendocrine tumors.
The practical use of this classification is debatable.
Group 1) can basically correspond to any pancreatic cystic lesion; groups 2) and 3) can be very difficult to distinguish.
Finally group 4) is the most useful and exists to remind us that what appear to be cystic lesions can in fact correspond to solid lesions with a large cystic component.
What to do next?
The main radiologist’s concern when confronted with a pancreatic cystic lesion is this: Is it a neoplasm or will ever become a neoplasm?
A microcystic serous cystadenoma,
with all its imaging hallmarks doesn’t pose as challenge and can be diagnosed only with imaging tools.
The oligocystic and unilocular types of serous cystadenoma can’t be diagnosed by imaging as they are difficult to differentiate from a mucinous cystadenoma.
One (or more) simple cyst that is diagnosed 4 weeks after an acute pancreatitis diagnosis can be considered a pseudocyst.
An accurate clinical history helps to reach a diagnosis.
Multiple pancreatic cystic lesions are rare.
If they are simple cysts,
all with the same appearance,
and do not show any communication with pancreatic ductal system we must consider a syndromic background as Von Hipple-Lindau (VHL) syndrome.
If they do communicate with pancreatic ducts we should consider a side-branch IPMN (specially if located in the uncinate process).
A simple,
solitary cyst without a previous history of acute pancreatitis becomes a bigger challenge.
If it has communication with the pancreatic ductal system,
then it’s probably an IPMN.
If there’s no communication the hypothesis of it being a mucinous cystadenoma must be ruled out.
Endoscopy US-guided (EUS) fine needle aspiraion (FNA) of the fluid within the cyst may be a precious tool.
This examination gives us,
not only the lesion’s morphology (mainly if it has septa or wall nodules) but also an biochemistry analysis of the fluid ( Fig. 8 ).
In this analysis many elements can be measured,
such as amylase,
carcinoembryogenic antigen (CEA),
carbohydrate antigen CA 19.9 as well as identify the presence of mucin or neoplastic cells within the cystic fluid.
If we can’t achieve a lesion’s characterization,
we must rule out the possibility of it being a malignant or pre-malignant lesion.
We know that if a lesion has a size less than 3 cm and/or is asymptomatic only in very few cases can it be malignant,
unless they have worrisome features.
The morphologic features that should alert radiologists for a malignant lesion or a malignant transformation are:
- Presence of wall nodules;
- Wall or thickened septa;
- pancreatic ducts dilatation;
- lesion’s growth;
- Presence of lymphadenopathy.
A possible algorithm to characterize pancreatic cystic lesions:
The American College of Radiology (ACR) presents an algorithm for incidental pancreatic cystic lesions ( Fig. 9 ).
This algorithm was designed for asymptomatic lesions that don’t present worrisome features.
According to ACR all lesions that are larger than 3 cm should be surgically resected,
unless they are either a pseudocyst or a serous cystadenoma.
In this algorithm symptomatic lesions aren’t included and it doesn’t consider the possibility of resecting lesions with less than 3 cm.
We also present another algorithm from this paper: "Cystic tumors of the pancreas: Imaging and Management” Radiological Clinics of North America,
2012 ( Fig. 10 ) that’s based in the presence or absence of symptoms and lesion’s size,
which we consider more useful.
This algorithm is easier to follow in routine clinical practice and approaches this problem in a broader form (it includes both symptomatic and asymptomatic lesions).
A lesion is considered symptomatic if it occurs in a context of: hyperamylasemia; onset of diabetes; severe epigastric pain; weight loss; steatorrhea.
Finally we are faced with lack of consensus concerning the follow-up timing of these lesions.
It’s important to remember that 75% to 90% of all focal pancreatic cystic lesions incidentally found are stable at long-term follow-up.
Of those that do progress,
the mean time interval for significant growth (>20%) is longer than 2 years.
For this purpose we suggest the follow-up timings also presented in the paper
"Cystic tumors of the pancreas: Imaging and Management” Radiological Clinics of North America,
2012 ( Fig. 11 ).