In this section we are going to demonstrate on the example of computed tomography how nanotechnology is currently applied in CT imaging.
More specifically,
we are going to give a general overview of some important nanobased contrast agents used in CT pointing out some of their interesting features as well as some of the concerns researchers have to face in their manufacture.
It is worth mentioning that nanoparticles of larger sizes (larger than 400nm) tend to be taken up by macrophage cells through opsonization.
So,
organs with high concentrations of phagocytic cells such as the liver and spleen as well as the lymphatic tissues show a high CT contrast enhancement when imaged with large nanobased contrast agents.
This can sometimes cause flu- and allergy like symptoms under the term : CARPA (complement activation related pseudoallergy)
Currently “hot” nanobased computed tomography contrast agents include the following:
I.Iodine-based contrast agents
a)Liposomes:
They are spherical vesicles lined by a double layer of phospholipids.
They can be used as carriers of both hydrophilic agents,
such as iodinated contrast agents,
encapsulated in their inner aqueous core as well as of hydrophobic agents dissolved in their phosholipid bilayers .
Their main advantage is their biocompatibility.
Their main disadvantage is their relatively low stability.
Free liposomes,
other than PEGylated liposomes,
are rapidy taken up by the ReticuloEndothelial System. This has already be used in mice for the detection of liver metastases since in contrast to the normal liver metastases do not contain phagocytic Kupffer cells.
Liposomes are also cleared from the blood not via renal filtration but rather via the RES which could lower the risk of nephrotoxicity,
especially in patients who undertake multiple CT examinations.
On the other hand,
the leakage of the encapsulated interior out of the liposomes could cause damage to the kidneys.
In the case of liposomes it has been possible to demonstrate the importance of the use of nanobased contrast agents for the real time observation of the effects of an applied therapy.
More specifically liposomes were used to detect pulmonary embolism in a rabbit model an monitor the resolution in real time after the injection of tissue plasminogen activator.
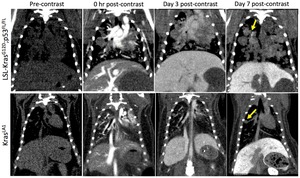
Fig. 1: Representative coronal micro-CT images of LSL-KrasG12D;p53FL/FL and KrasLA1 mice before liposomal contrast-agent injection and at 0 hr, Day 3 and Day 7 post-contrast injection.
Note the differential enhancement of tumors at Day 7 post-contrast time point in the LSL-KrasG12D;p53FL/FL lesions only.1
References: Badea et al. in conjuction with reference 1:This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
b)
Nanoemulsions:
A mixture of two fluids that are normaly immiscible (that do not mix if put together) is called emulsion.
Nanoemulsions form droplets at the nanoscale.
The interesting thing about nanoemulsions is that they have a larger surface to volume ratio than ordinary emulsions.
Their advantages are that they are thermodynamically stable,
simple to decorate and allow the encapsulation of a high amount of an iodinated contrast agent.
The classical example of a nanoemulsion is lipiodol.
Nanosuspensions:
Colloidal dispersions of pure (water-insoluble) drug particles stabilized by surfactants.
Colloids are mixtures in which particles ranging between 1 and 100 nm in size are evenly distributed in a medium; just like dust is dispersed in air.
Surfactants are compounds that lower the interfacial tension between two liquids or between a liquid and a solid.
Nanocapsules:
They are relatively stable nanoparticles consisting of a crosslinked polymeric membrane enveloping a payload-material that is often insoluble/immiscible with the surrounding solvent.
The polymeric shell confers the enhanced water solubility
c)Polymeric nanoparticles:
They are fabricated through the incorporation of iodine containing compounds into macromolecular structures.
This structures prevents the iodine comounds from leaving the nanoparticle allowing in this way the formation of stable nanoparticles of the desired size.
Two special kinds of polymeric nanoparticles are the dendrimers and the amphiphilic polymers.
The dendrimeric contrast agents (<greek δένδρο=tree+μέρος= part of it) are highly branched polymeric macromolecules.
The consist of an inner core which is connected to branching patterns.
Unlike classical polymers,
dendrimers are show low polydispersity values.
The number of the branching patterns is related to the properties as well as to the shape of the dendrimer.
Low generation dendrimers generally represent open structures while high generation dendrimers are usually globular in shape.
Amphiphilic contrast agents are as the name implies contrast agents which comprise a hydrophilic part,
usually PEG,
and a hydrophobic part.
Dendrimers?
Fig. 3: Schematic diagram
References: Albertazzi et al. In conjunction with reference 9:This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
II.
Gold nanoparticles (AuNPs)
Gold nanoparticles show a high biocompatibility due to their inert nature.
They are of interest,
because their use could lower the radiation dose delivered to the patient.
What makes them excellent CT contrast agents is their app.
2,7 times greater contrast per unit weight in comparison to iodine.
They are manufactured in various different shapes like sphere,
rods,
shells and cages.
Although the different shapes are not related to a different CT contrast effect,
the different shapes are related to some differences regarding the behavior of gold nanoparticles,
eg.
gold nanorods manage to escape from the RES phagocytosis more easily than spherical gold nanoparticles.
What do nanoparticles look like in the Transmission Electron Microscope?

Fig. 2: Transmission electron micrograph of GNPs having approximately 12 nm cores
References: In conjunction with reference 8: This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Important potential applications of CT contrast agents described in the literature include:
a)Blood pool imaging
Nanobased contrast agents could be used as blood pool imaging tools due to their high circulation time which could help in the delination of blood vessels in cardiovascular imaging settings.
The size of (spherical) nanoparticles should exceed 15 nm so as to prevent rapid excretion through the kidneys or liver uptake and smaller than app.
200 nm in order to prevent the filtration through the spleen.
b)Imaging of the ReticuloEndothelial System
The uptake of nanoparticles by the ReticuloEndothelial System suggests their potential use in the imaging or macrophage rich organs such as the liver,
the spleen and the lymphatics as well as in depicting rupture prone plaques that contain an increased density of macrophages.
The latter can be enhanced through the modification of the surface of the nanoparticles through ligands in such a way that it specifically targets the rupture prone plaques.
c) Cancer
The afore mentioned EPR effect as well as the biofunctionalization of nanoparticles for the active targeting of tumor cells represent fields of currently undergone research.
Limitations in the use of nanobased contrast agents in computed tomography
a)Potential toxicity.
b)Price (gold nanoparticles)




