Because of the poor prognosis,
pancreatic adenocarcinoma has been described and studied since years.
A detailed imaging schema for the TNM-classification has been modified,
adapted and finally proposed in order to achieve better standardization in the therapeutic approach.
The diagnostic CT-parameters of resectability are well known and in common use of radiologists,
who describe the tumor.
[3,4]
Parameters of evaluation of resectability at CT.
[4,
5]
-
Tissue obliteration: gross macro infiltration.
-
Alteration of the tissue density (ROI in the region of interest): spread,
fine increased density or suspicion of micro infiltration
-
Irregularity of the medial margin of the uncinate process with focal bulge in the pancreatic contur --> micro infiltration soft tissue mass surrounding the area of involvement
-
Collateral vessels,
engorgement of collateral circles: secondary sing of vascular involvement
-
Venous thrombosis
-
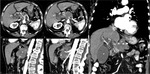
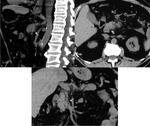
Contact with the vessels: Superior Mesenteric Artery (SMA),
Common Hepatic Artery (CHA),
Celiac Axis,
Gastroduodenal Artery (GDA),
Splenic Artery (SA ),Portal Vein (PV),
Inferior Vena Cava (IVC),
Superior Mesenteric Vein (SMV): <180° without deformity,
> 180° without deformity,
>180° with deformity,
>180° with tear drop deformity.
Attention: arterial variants.
(Figure 1)
-
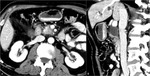
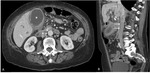
Involvment of the retroportal lamina,
retroportal fat tissue,
connective tissue ,
lymphatic and Retroperitoneal nerves: pain (Figure 2)
Non resectability criteria were: Distant metastases (liver,
lung,
pleura or peritoneum),
presence of distant metastastic lymphnodes (medistinal,
supraclavicular,
interaortocaval or paraaortal),
infiltration of extrapancreatic or extraduodenal viscerals and tumorous artrial vessels encircling of over 180° as described in the CT-parameters.[6]
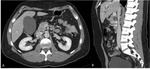
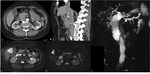
Invasion of the peripancreatic fat tissue,
lymphatic network and extrapancreatic neural plexuses (Figure 3) affects the staging and the patients prognosis,
because neoplastic tissue may remain in the resection margins,
resulting in postoperative recurrence.[7-10]
On CT examination a retroperitoneal fat tissue invasion appears as streaks extending into the soft tissues from tumor margin.
Coronal and sagittal multiplanar reformatted images are very useful to perform a correct assessment of these findings,
increasing CT performance in the evaluation of local extension,
useful for descriminate preoperativelly the best treatment-strategy.[9,
11-13]
Anatomical location of the retroportal fat tissue:
The retroportal lamina is roughly rectangular in shape.
It is located behind the portal vein,
behind the distal parts of the mesenteric vein and between the left margin and right proximal wall of the uncinate process.
It contains adipose tissue,
nervous structures,
lymphatic vessels and capillaries.
Because of the absence of fibrous sheath or fascia,
its complete removal in pancreatic head carcinoma is feasible by a subadventitial dissection of the superior mesenteric artery,
which can be consideres as the real limit of the mesopancreas.[14,
15].
Pancreatic adenocarcinoma generally grows in a less cohesive fashion.
Itis charecterized by “ tumor desmoplasia”,
a remarkable increase of connetive tissue that infiltrates and envelops the neoplasm.
More extensive fibroblastic cell proliferation in pancreatic carcinoma correlates with poor outcome.
The tumor cells produce and secrete proteolitic matrix-degrading enzymes,
angiogenic growth factors (f.e.
VEGF,
bFGF,
PDGF,
IGF,
TGF-beta) and lymphangiogenesis factors (VEGF-C).[16]
The resulting stimulation of neovessels formation in the stroma encourages the carcinoma progression by delivering nutrients and oxygen,
promoting local growth and cell-scattering around the extrapancreatic tissue,
resulting in a fibrotic thickening of adipose tissueand forming peripancreatic strands.
This creates an inhomogenity which can be seen on CT ,frequently described and identified as “reticular”,
“tubular”,
“soft tissue mass” along the lymphatic system,
including microvessels in peri-pancreatic fat tissue.
Talking about the retroportal lamina,
there is an imaging-classification of invasion:
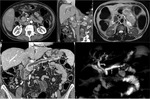
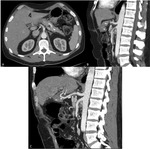
Grade 0: No retroperitoneal fat infiltration (Figures 4,
5)
Grade 1: Strands or streaks in peri-pancreatic soft tissues (<0,5mm) radiating from the tumor margins (Figure 6)
Grade 2: Strands or streaks in peri-pancreatic soft tissues (>0,5 mm) radiating from the tumor margins without abutting the major artery (Figures 7-8)
Grade 3: Strands or streaks structures (>0,5) radiating from the tumor abutting the major artery (Figure 9)
Surgical procedure:
After excluding intra-abdominal contraindications like peritoneal carcinomatosis and liver metastasis,
all patients underwent a pylorus-sparing pancreaticoduodenectomy (PPPD),
also known as Traverso-Longmire procedure,
with the removal of the duodenum,
the gall bladder and its cystic duct, the common bile duct,
the proximal jejunum,
the head of the pancreas and the regional lymph nodes.
Reconstruction of the anastomosis was performed in Blumgart´s technique (transpancreatic U-suture technique).
If the tumor does not involve vascular structure,
it does not performed any vascular resection.[17,
18]
Histopathological work up:
All histopathological specimens were assessed by size,
histological type,
differentiation (grading),
lymphovascular invasion and resection margins.
On patological examinations,
there may be two ways of peripancreatic invasion by pancreatic cancer:
-
tumor emboli,
that fill up the dilatated lymphatics and neovessels
-
tumor extension along the lymphatic channels and nerve plexuses (f.e.
posteriorly to the portal vein along the plexus pancreaticus capitalis ,
involving the right celiac ganglion; anteriorly along the gstroduodenal artery involving the neural plexus of the common hepatic artery and the plexus of the hepatoduodenal-ligament) appearing as confluent tissue with similar attenuation values of the primary pancreatic cancer,
extending along neural plexuses pathways in directed contyguity with the primary intrapancreatic adenocarcinoma.
It occurs even in relatively small cancers (<2cm).[8]
The current grading criteria are useful to descriminate the best treatment strategy preoperatively,
because the grades were significantly associated with overall survival,
as well as the incidence of surgical margin status and recurrence.
The posterior margin of the retroportal lamina was found to be the only aspect of a R1 resection margin that significantly influenced the disease free survival.
The invasion of the retroportal fat tissue (grade 3) is considered as T3 tumor,
and it may be managed as a systemic disease,
using for example with neoadjuvant chemo-radiotherapy before surgery.
Prognostically it was seen that intraoperative inability of complete resection of the involved retropancreatic fat tissue had the same prognosis as a non-operation.
On the other hand It must be considered that Whipple operation is a complicated and long procedure with a high risk of pre-and postoperative complications.
A positive involvment of the retroportal lamina means that the operation was unuseful and a recurrent of the disease is very likely.
[11]