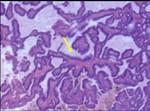
A papillary lesion consists of a frond-forming growth pattern composed of fibrovascular stalks (Fig. 1) covered by a layer of epithelial cells with or without an intervening myoepithelial cell layer.
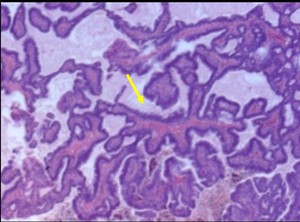
Fig. 1: Core biopsy:Photomicrograph. Branching papillary structures with fibrovascular cores (arrow) and lined by benign columnar cells
| 2.
Clinical presentation. |
Patients with papillary lesions may present with:
- Nipple discharge. Suspicious discharge is spontaneous,
serous or bloody and is coming from a single duct.
Cytology may depict papillary groups (Fig. 2 ).
- Palpable mass.
Most frequently central.
- Sometimes they are asymptomatic.
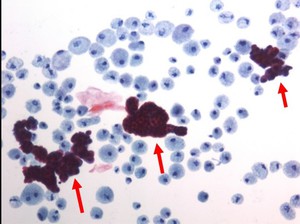
Fig. 2: Cytology of bloody nipple discharge: Papillary groups (arrows)
| 3.
Pathology classification. |
The classification for papillary lesions includes a wide range from benign to malignant lesions,
and frequently different areas within the same lesion offer features from several entities.

Table 1: Pathology classification
Lesions may present with different appearances,
usually non-specific for benign,
atypical or malignant lesions.
4.1.
Mammograms.
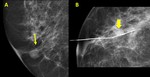
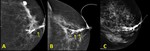
- Masses. Usually a solitary round or ovoid,
well-circumscribed retroareolar mass (Fig. 3 ).
Multiple papillomas may be peripheral masses.
- Calcifications are uncommon: either coarse,
dense calcifications or microcalcifications may appear.
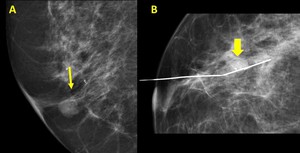
Fig. 3: A.- Papilloma. Round retroareolar mass without calcifications (arrow).
B.- Papilloma. Round retroareolar mass with calcifications ( arrow).
4.2.
US.
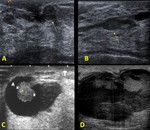
Benign and malignant lesions represent a solid growth within the duct with variable solid and cystic component,
presenting with different appearances also non-specific (Fig. 4 ):
- Well-circumscribed hypoechoic solid mass.
- Solid mural nodule within a dilated duct or within a cyst.
- A complex cyst
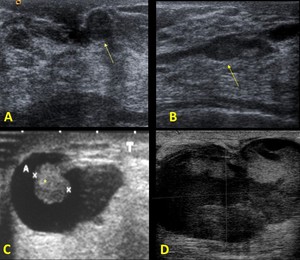
Fig. 4: US: A.- Well-circumscribed hypoechoic solid mass (arrow). B.- Solid mural nodule (*) within a dilated duct (arrows) C.-An intracystic mass (*). D.- A complex cyst
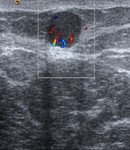
On US Doppler ( Fig. 5 ) the mass is usually highly vascular,
sometimes with a vascular pedicle.
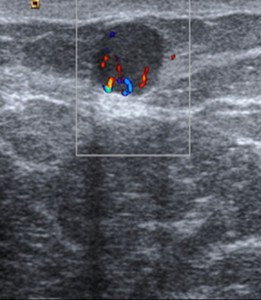
Fig. 5: US. Doppler.
Highly vascular lesions, sometimes a nodule with a vascular pedicle
4.3.
Ductography.
-Technique.
The technique consists in direct cannulation of the discharging duct and contrast agent injection through the cannula obtaining duct opacification.
Mammograms are then obtained in orthogonal views in order to search for ductal abnormalities,
most frequently papillary lesions.
-Indications for ductography.
Spontaneous unilateral single-pore nipple discharge of any kind with bloody or serous types being more suspicious.
-Findings.
( Fig. 6 )
Anatomy of ductal systems is variable from small systems to widely branching ones.
Ductal dilatation may be the cause of nipple discharge,
without associated papillary lesions.
Nevertheless some features may indicate the presence of a papillary lesion either benign or malignant.
- An intraluminal filling defect,
with meniscal sign is most frequently due to intraductal papilloma.
Sometimes multiple defects appear.
- Ductal wall irregularity,
strictures,
obstruction and distortion are more frequently caused by malignant lesions.
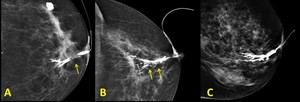
Fig. 6: Ductography A.- Intraluminal filling defect. B.- Ductal wall irregularity, and distortion. C.- Ductal dilatation
4.4.
MR-MR ductography.
MR with Gadolinium injection.
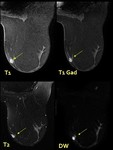
Usually round,
ovoid,
or lobulated well-circumscribed masses (Fig. 7) with or without ductal dilatation and contrast enhancement with variable enhancement patterns.
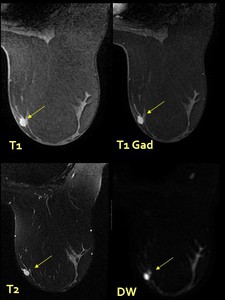
Fig. 7: MR.- Hyperintense lesion on MR T1,T2 and DW images
MR ductography.
Two different techniques may be performed:
- Direct: Cannulation and Gad injection within the duct.
- Indirect.
Heavily T2-weighted sequences and high spatial resolution.
4.5.
Interventional procedures.
Guidance of interventional procedures may be performed by mammography,
US,
or MR imaging,
but US is the preferred method.
Certain lesion types present peculiarities:
• Nipple or retroareolar lesions:
Difficulties: Puncture in this area is specially painful.
Tricks:
- Additional anesthesia should be used.
- The use of an US approach from a distant point sometimes changing patient positioning may help.
• Lesions within a duct or complex cysts.
Difficulties: If the cystic area disrupts and disappears with the first pass the mass may be difficult to identify.
Tricks:
- Accurate targeting of the solid area, trying to avoid the cystic component.
- The use of a vacuum-assisted directional device may help.
• MR guided biopsies
Difficulties: Lack of adequate compression of the anterior region of the breast makes this area difficult for biopsy.
Trick: second-look US may find the lesion allowing US-guided biopsy.
4.6.
Pathology.
The key for diagnosis is the presence of fibrovascular stalks (Fig 1).
The diagnostic criteria for papillary lesions are:
• Cell types.
Epithelial and myoepithelial.
Absence of myoepithelial cells indicates carcinoma.
• Stroma of papillae. Prominent fibrosis with epithelial entrapment.
Difficulties in biopsy are frequent and caused by:
• Lesion heterogeneity. A wide morphologic spectrum frequently appears within a single lesion with benign,
atypical and malignant areas in the same lesion.
• Partial sampling due to tissue fragmentation and undersampling together with lesion heterogeneity cause a certain degree of overgrading in surgery.
5.1.Intraductal papilloma.
Papillomas are located central,
within a few centimeters of the nipple and grow within the duct.
(Fig. 8 ).
- Mammogram depicts a well- circumscribed nodule.
- US.
A well-circumscribed nodule sometimes within a duct or cyst.
- Ductography.
A filling defect within a duct sometimes with a menisc-sign.
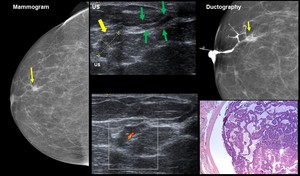
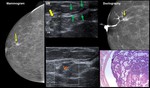
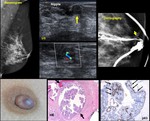
Fig. 8: Papilloma: 32-year old woman. Right breast single-pore bloody discharge
Mammogram. Partially circumscribed nodule. US Nodule (yellow arrow)
within a duct (green arrows) Ductography. Filling defect within a dilated duct (arrow). HE: Papilloma
5.2.
Papillomatosis.
Multiple papillomas show an increased risk of malignancy .
They are usually located on distal ducts and presentation may be asymptomatic,
serous or bloody nipple discharge Fig. 9.
- Mammograms. Multiple circumscribed nodules.
- US.
Multiple small hypoechoic nodules,
central or peripheral.
- Ductography.
Multiple filling defects on ductography,
both central and peripheral.
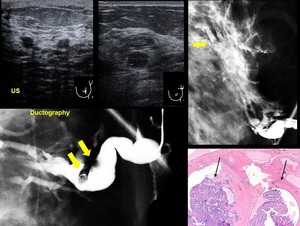
Fig. 9: Papillomatosis: 44-year old . Left breast one-pore bloody discharge
US. Multiple small hypoechoic nodules, two in the central area, one peripheral (Upper outer quadrant) Ductography. Multiple filling defects on ductography, both central and peripheral.
HE.- Papillomas (arrows)
5.3.Papilloma with atypia (atypical papilloma).
Variable criteria have been used for diagnosis of atypical papilloma.
No specific findings appear on imaging with a benign appearance Fig. 10 .
- Mammogram. Well-circumscribed nodule,
sometimes small difficult to detect.
- US. Well-circumscribed nodule.
- Ductography. Filling defect,
usually central.
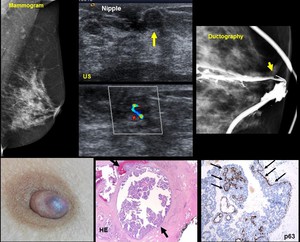
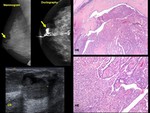
Fig. 10: 61-year old woman. Single-duct bloody nipple discharge for two weeks
Mammogram. Normal US. Well-circumscribed 5 mm nodule immediately beneath the nipple. Ductography. Central filling defect
HE.- Papilloma with atypia. Myioepthelial cells on P63
5.4.Papilloma with DCIS.
Findings are similar to a benign lesion ( Fig. 11 )
- Mammogram depicts a circumscribed nodule.
- US. Circumscribed nodule,
sometimes with cystic and solid area.
- Ductography. Filling defect within a duct.
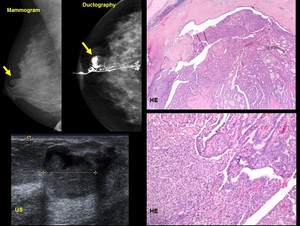
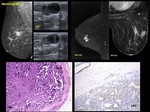
Fig. 11: Papilloma with DCIS.- 40-year old woman. Single duct bloody discharge
Mammogram depicts a circumscribed nodule
Cyst with solid area on US.
Ductography. Filling defect within a dilated duct
5.5. Papillary DCIS.
Findings are similar to a benign lesion and papilloma with DCIS.
Differentiation from papilloma with DCIS is based only on pathological criteria,
with different groups proposing different criteria,
in general: greater than 3 mm and DCIS comprising at least one third but less than 90% of a papillary lesion.
If it is smaller than 3 mm,
or involves less than 30% of papilloma,
the diagnosis should be atypical papilloma or papilloma with DCIS.
5.6.
Encapsulated papillary carcinoma
It is an invasive tumor with excellent prognosis.
Terminology has changed.
It used to be called “Intracystic or encysted papillary carcinoma”,
which is now deemed incorrect,
the correct term should be encapsulated papillary carcinoma Fig. 12 .
- Mammogram.
Well-circumscribed mass
- US.
Mixed solid and cystic lesion,
sometimes complex cysts.
Low-level echoes may be depicted within the cystic areas,
consistent with hemorrhage due to high vascular component in the solid area.
- MR.
Haterogeneous lesion,
with hyperintense component on T2 w image and contrast enhancement with type 2 or 3 curves.
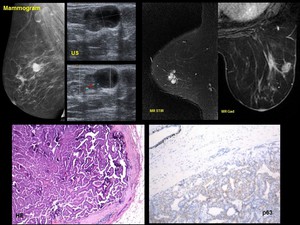
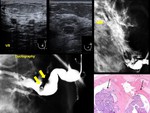
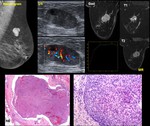
Fig. 12: Encapsulated papillary carcinoma.- 81-year old woman. Palpable mass
Mammogram. Well-circumscribed mass
US. Mixed solid and cystic lesion. Low-level echoes are depicted within the cystic areas, consistent with hemorrhage
US Doppler .Vessels are depicted on solid area
MR Hyperintense lesion on T2 w image.
Contrast enhancement.
5.7.
Solid papillary carcinoma.
It is a variant of DCIS.
Single or multiple circumscribed nodules of neoplastic cells,
with solid pattern and grossly well circumscribed frequently with mucin production.
It appears usually in older women (70 yo).
Fig. 13 .
- Mammogram. Usually well-circumscribed mass sometimes lobulated or partially ill-defined or occult margins.
- US. Solid highly vascular circumscribed mass sometimes with cystic component.
- MR. Usually well-circumscribed mass predominantly hyperintense on T1 and T2 sequences with hypo T1,
hyper T2 cystic areas.
Contrast enhancing curve type 2 or 3.
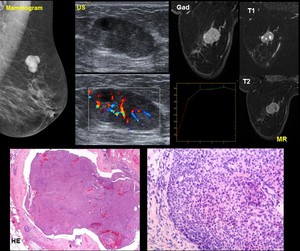
Fig. 13: 82-year old woman. Palpable mass
Mammogram. Well-Circumscribed lobulated mass
US.Solid highly vascular circumscribed mass with small cystic areas
MR. Lobulated mass predominantly hyperintense on T1 and T2 sequences with hypo T1, hyper T2 cystic areas. Contrast enhancing curve type 2
| 6.
Challenges in papillary lesions. |
Papillary lesions pose a challenge both in diagnosis and treatment.
When facing a papillary lesion some issues are clear,
but others are continuosly evolving with different theories.
6.1.
Challenges in diagnosis.
• Questions.
The first questions we should ask when facing a lesion which could possibly be papillary:
- Is it a papillary lesion?
- Is it benign or malignant?
• Difficulties.
Diagnosis is difficult as overlapping features appear clinically,
on imaging and even on core biopsy samples:
- Imaging may offer nonspecific findings in all techniques.
- Biopsy problems due to heterogeneity of lesions and partial sampling, tissue fragmentation and undersampling.
• Recommendations.
Features that suggest malignancy.
The features which are statistically most frequently associated with malignancy are:
- Size greater than 1 cm.
- Lesion location more than 3 cm from the nipple.
- Patient age greater than 50 years.
6.2.
Challenges in management.
Management of papillary lesions in also controversial in some points.
• Questions.
Should benign lesions be excised? The usual recommendation for benign papillary lesions has been excision due to the high rate of overgrading at excision.
• Difficulties.
Benign lesions may have an associated area of malignancy and they also have an increased risk of malignancy.
• Recommendations for management.
• Papilloma with atypia (atypical papilloma),
Papilloma with DCIS,
Papillary DCIS,
Encapsulated papillary carcinoma,
Solid papillary carcinoma.
Excision
• Lack of radiopathologic concordance.
Excision.
•Benign papilloma is still a matter of discussion.
Discussion: surgical excision was usually recommended but recently some authors recommend follow-up.
| 7.
Diagnostic work-up and recommendations for management. |
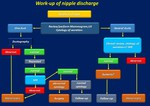
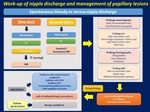
We propose our work-up of nipple discharge and diagnosis and management of papillary lesions in Fig. 14 and Fig. 15 .
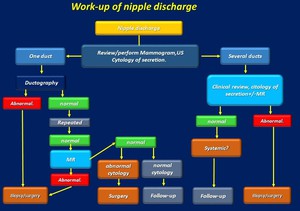
Fig. 14: Work-up of nipple discharge
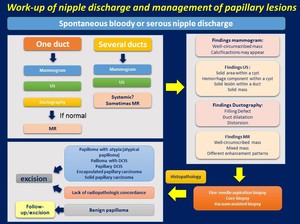
Fig. 15: Work-up of nipple discharge and management of papillary lesions