A.
Before scanning
When,
why and how to perfom paranasal sinus CT
Paranasal sinus CT imaging is a fundamental component of caring for paediatric patients with sinonasal and skull base pathology to confirm diagnosis and eliminate the need for unnecessary medication and surgery [4].
CT scans provide information about patients’ anatomy and disease,
which may not be appreciated on physical examination,
and are used to help otolaryngologists to develop a treatment plan for patients.
Indications for paranasal sinus CT include trauma,
malignant disease (tumors,
masses),
systemic congenital pathologies like cystic fibrosis and primary ciliary dyskinesia,
complicated or recurrent acute sinusitis and chronic sinusitis - especially when medical management and/or surgery have failed to control symptoms.
Chronic inflammatory diseases account for the major part of CT examinations which add valuable information regarding the extent and severity of inflammation and exquisitely demonstrates sinus anatomy and surgically relevant anatomic variants.
Given the relatively small diameter of the head in comparison with the trunk and the high intrinsic contrast of the evaluated structures,
CT can be performed in this district with adapted dose parameters depending on the indication.
Dose descriptors and paediatric diagnostic reference levels
Two dose descriptors are commonly used in computed tomography,
Computed Tomography Dose Index (CTDI) and Dose Length Product (DLP) [5].
CTDI is internationally recognized as the most important measure of the radiation output from the CT scanner.
Its units are milligrays (mGy).
It is measured by using a pencil ionization chamber (100 mm length) placed in a standard circular plastic phantom (a 16-cm diameter phantom for adult head and paediatric head and body calculations,
and a 32-cm diameter phantom diameter for adult body measurements).
As the dose distribution in CT is highest at the surface and lowest at the centre of the object,
CTDI is expressed in terms of CTDI weighted (CTDIw) and it is calculated by taking the properly weighted average of the measured dose at the surface and at the center of the specific phantom.
CTDIw is given by:
CTDIw = 1/3 CTDIcenter + 2/3 CTDIedge
CTDI may easily be considered as the radiation dose delivered to the patient during one complete revolution of the tube-detector system with no movement of the CT table (sequential mode).
Due to the fact the majority of CT scan protocols are now performed in spiral mode,
CTDIw must be replaced with another CT dose descriptor called CTDI volume (CTDIvol) that takes into account the pitch value of the scan.
CTDIvol is given by:
CTDIvol = CTDIw/pitch
CTDIvol represents the dose within the scan volume from a particular scan protocol for a standardized phantom and it is most suitable descriptor to compare the dose of different CT protocols.
Recently,
the American Association of Physicists in Medicine in its Task Group 204 Report (AAPM-TG204) has proposed the calculation of size-specific conversion of CTDIvol in order to get a more accurate descriptor of absorbed dose in patients with a variety of body habitus,
including children [6].
The formula to estimate the patient dose for a specific patient size (Size-Specific Dose Estimate,
SSDE) is:
SSDE = CTDIvol x f
where f is the correction factor that takes into account the patient size.
Due to the variable size of children,
it is clear that such a parameter is better than any other,
taking into consideration the patient’s real size instead than an approximation.
However,
SSDE is not currently displayed on CT scan console,
so it must be calculated manually.
Therefore,
although SSDE will probably replace CTDI in the near future,
at the present it is not possible to use it.
Dose-length product (DLP) is a second key patient dose descriptor in CT and it is an indicator of the total radiation dose for an entire CT exam.
DLP is simply obtained multiplying the CTDIvol value per the scan length as given by the following formula:
DLP = CTDIvol x scan length
DLP is expressed in mGy x cm.
These two dose descriptors allow to monitorize radiation dose while performing a CT examination,
but how to know when the dose is too much? A cut-off level is needed,
something to guide everyday practice and to improve it continually.
For this purpose,
Diagnostic Reference Levels (DRLs) have been recommended by the International Commission on Radiological Protection (ICRP) [7] as a measure to improve optimization of patient protection and a practical tool to promote the assessment of existing protocols and appropriate development of improved protocols at each CT centre by facilitating the comparison of doses from present practice.
According to the ICRP,
a DRL is a form of investigational level,
applied to an easily measured quantity,
and intended for use as a simple test for identifying situations where the levels of patient dose are unusually high or low [8].
The objective of DRLs is to help avoiding radiation dose that does not contribute to the clinical purpose of a medical imaging task [9].
Examination-specific DRLs for various patient groups can provide the stimulus for monitoring practice to promote improvements in patient protection.
Paediatric examinations and procedures are of special concern because,
compared to adults,
children have a higher risk from the detrimental effects of radiation.
Increased incidence of cancer after CT examinations in childhood has been reported in recent years [3,10].
Despite the recommendations and the clear need for DRLs for paediatric examinations,
few paediatric DRLs are available,
and only in a small number of countries within Europe.
Recently,
a workshop entitled ‘European Guidelines on DRLs for Paediatric Imaging’ was held in Lisbon (15th-17th of October),
focused on the establishment and use of DRLs in paediatric radiodiagnostic imaging so as to advance optimization of radiation protection of paediatric patients.
A methodology for establishing and using DRLs for paediatric radiodiagnostic imaging has been recommended,
and,
in addition,
European paediatric DRLs have been updated and extended [11].
Dose optimization strategies
The most important strategy to respect appropriateness criteria and to reduce as much as possible the radiation dose during paediatric CT examinations is to increase awareness about radiation risk and knowledge about radiation protection issues by means of specific training events.
Training must be performed on a regular basis by professionals with certified expertise,
focusing on dosimetry concepts and optimization measures,
dose reference levels,
radiation protection rules,
new research studies and relevant publications.
Information campaigns such as Image Gently®[12],
Image Wisely®[13],
and the most recent Eurosafe campaign have paid specific attention to the fundamental role of staff training in radiation protection,
emphasizing the role of strict cooperation among all radiological operators.
Another effective strategy for optimizing paediatric exams is the setting of a group of protocols with specific parameters for paediatric patients for each relevant CT scanner model in the department.
In fact,
paediatric exams may be performed on different CT scanners due to logistic problems: radiological staff,
with specific attention to radiographers,
must be trained to take full advantage of the best technologies available on each scanner.
So,
it is mandatory to set up dedicated training courses to allow users how to learn and understand the special features of each scanner.
B.
During scanning
Patient positioning
Patient should be placed supine,
head-first into the gantry,
with the head in the head-holder whenever possible,
with a slight reclination of the head to obtain parallel alignment of the upper jaw to the gantry minimizing impairment by dental prosthesis artifacts.
Table height should be set such that the external auditory meatus is at the center of the gantry.
Specific attention must be paid to the correct positioning of the head inside the gantry.
Indeed,
new CT scanners are equipped with specific beam filters (bow-tie filters) requiring a proper alignment of the patient in the gantry isocenter in order to avoid unwanted dose and noise increase.
Furthermore,
to reduce ocular lens exposure,
the scan angle should be parallel to a line created from the supraorbital ridge to the inner table of the posterior margin of the foramen magnum.
Immobilization strategies should be used to minimize patient motion artifacts.
Parameters tailoring
A scout view of the skull is obtained to plan CT data acquisition.
Scan range extends from the roof of the frontal sinus down to the maxilla.
Data is acquired in cranio-caudal direction.
Radiation dose depends on tube collimation,
tube rotation time,
pitch,
kVp,
mAs and the scan field of view (FOV).
Due to the different technical characteristics of the scanner and the physical trait of the patient (age,
weight,
height,
sex) CT parameters should be carefully adjusted for each specific exam.
Low-dose paranasal sinus CT allows the acceptance that diagnostic images could tolerate a certain amount of noise for the diagnostic purpose of the exam.
Thanks to the anatomical characteristics of sinus,
a great range of operative strategies for dose reduction can be implemented.
In particular,
the high intrinsic contrast of the structures (bone,
mucosa,
air) and the minimal presence of tissues with similar density allow the possibility of improving spatial resolution,
while contrast resolution is naturally already present.
As a consequence,
lowering kV values does not improve overall image quality,
though it is advisable in children in order to decrease radiation dose.
In-field and out-of-field shielding
When performing a CT of the paranasal sinus,
specific attention must be paid to the eye lens and thyroid gland,
as they are hit by direct or scattered radiation.
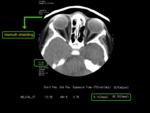
The radiation dose delivered to these two high sensitive organs can be lowered by using specific protection devices (such as bismuth shielding) or adapting scan protocols [14].
A bismuth shield primarily removes the lower-energy photons depositing energy and causing ionizations in tissues.
These low-energy photons do not contribute to image formation but do contribute to dose.
However,
these shields may also reduce the photon number reaching the detector ring in some projections and may add noise or artifacts to the images,
especially if no standoff pads are used (figure 1).
Recent studies show that the reduction in organ dose can also be more efficiently achieved by adapting scan parameters (i.e.,
using automatic exposure control).
In addiction,
out-of-field shields (lead) may be used to avoid irradiation of nearby sensitive organs.
C.
After scanning
Use of reconstruction filters: filtered back projection (FBP) and iterative reconstruction.
CT raw data,
acquired in axial plane,
may be reconstructed in the scanner’s computer processor.
Raw data is available for manipulation into any tissue algorithm (bone,
intermediate and soft tissue) or field of view,
and can be reformatted into any plane of interest as well as into three-dimensional renderings for surgical consultation.
To this purpose voxel isotropy is pivotal,
which can be obtained with thin beam collimation.
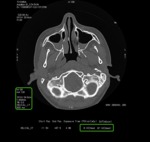
CT images reconstruction are usually performed by using filtered back projection algorithms (FBP),
to correct the blurring encountered in simple back projection.
This algorithm operates on several fundamental assumptions about scanner geometry but it is basically a compromise between reconstruction speed and image noise (figures 2-3).
One of the major problems with the FBP algorithm is noise.
Therefore,
many vendors have implemented new algorithms such as iterative reconstructions (IR) and model-based iterative reconstructions (MBIR).
These iterative reconstructions work on different assumptions about scanner geometry and noise statistics which are computationally more complex,
but their primary advantage is to greatly reduce image noise.
IR uses FBP native projections,
referred to as “measured projections”,
to create a new set of simulated projections which are compared with the former to determine differences in image noise.
Once these differences are determined,
it is applied to the simulated ones to correct for inconsistencies [15].
For this reason IR techniques allow to perform high quality images with lower photon energy than FBP (figures 4-6).
Dose monitoring
Many regulations and guidelines (such as the European directive Euratom 97/43) express the need to monitor radiation dose values for different types of procedures,
keeping radiation dose As Low As Reasonably Achievable (ALARA) [16].
Such efforts are easier,
and more likely to occur regularly,
when dose estimates are provided electronically.
Radiation dose tracking software facilitates the assessment of dose associated with individual CT protocols,
comparison with diagnostic reference levels (DRLs),
and identification of those exam protocols with high radiation exposure.
Radiation reporting systems can periodically query the procedures’ "dose objects" from an archive,
or receive them directly from the scanner.
The reporting system is expected to perform relevant dose quality assessment analysis and to produce related reports.
Dose details are recorded for each irradiation event,
allowing the evaluation of the cumulative radiation exposure of a specific patient.
This data could be useful to keep track of the day-to-day practice,
and to get to know if one procedure exceeds DRLs.
In our institution two radiation dose tracking software are available,
DoseWatch by GE Healthcare and TQM (Total Quality Monitoring) by Qaelum.
Using these software it is possible to track dose levels of all performed procedures and be alerted if some of them are beyond the third quartile range (the dose value used to set up DRLs) (figure 7).
European Workshop on Paediatric Imaging DRLs has stressed the importance of using dose monitoring software in all institutions managing paediatric patients,
as the most useful and precise way to control the amount of radiation dose and the good practice of all workers.
of using dose monitoring software in all institutions managing paediatric patients,
as the most useful and precise way to control the amount of radiation dose and the good practice of all workers.
Audit for quality assurance
An audit can be defined as "a systematic examination or review of radiological procedures which seeks to improve the quality and the outcome of patient care" [16].
A systematic evaluation of the patient dosimetry and image quality represent a fundamental aspect of any quality-control program in diagnostic radiology.
Image quality must be adequate for diagnosis and obtained with reasonable patient doses.
Clinical audits are carried out in accordance with national procedures and regulations; appropriate local reviews are undertaken whenever diagnostic reference levels are consistently exceeded and that appropriate corrective action is taken without delay.
DRLs can be used as a tool to perform audit in radiology.
Audits allow not only to identify exceeding dose values of specific procedures but also to outline unjustified variability among dose values of similar procedures.
Personalized audit feedback and education can change technologists' and radiologists’ attitudes about radiation and can lower patient radiation exposure from CT imaging.