OBSTRUCTIVE CAUSES
1.
CHOLEDOCHOLITHIASIS
Choledocholithiasis is the presence of gallstones within bile ducts,
typically the common hepatic duct and the common bile duct.
It is termed uncomplicated,
when asymptomatic,
or complicated when presenting as acute pancreatitis or cholangitis.
Management involves removing the stones (endoscopically or surgically) and treating any associated complications such as liver abscess or septic thrombophlebitis.
Diagnosis and treatment has traditionally been with endoscopic retrograde cholangiopancreatography (ERCP),
however it is associated with complications such as pancreatitis and is operator and experience dependent.
Therefore,
endoscopic ultrasound (EUS) and magnetic resonance cholangiopancreatography (MRCP) are becoming more widely accepted as modalities of choice for diagnosis,
with ERCP reserved for therapeutic purposes [1,2].
IMAGING OF CHOLEDOCHOLITHIASIS
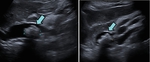
On ultrasound,
stones may be visualized as echogenic foci with posterior shadowing.
Dilatation is diagnosed when the common bile duct is greater than 4mm + 1mm per decade above the age of 40 (Fig. 1).
On CT,
the sensitivity for choledocholithiasis is 65-88% and typically is seen as a target sign (central rounded density with surrounding low attenuating bile or mucosa) in addition to biliary dilatation [2].
On CT cholangiography,
the sensitivity raises to 93% with a specificity of 100%.
However,
decreased contrast excretion may occur due to obstruction limiting utility.
On MRCP,
which is considered the gold standard,
both sensitivity and specificity approach 100%,
replacing ERCP as the diagnostic tool of choice.
On T2-weighted magnetic resonance (MR) images,
stones are visualized as filling detects [2].
2.
CHOLANGIOCARCINOMA
Cholangiocarcinoma (CC) is a malignant biliary adenocarcinoma,
and is the most common tumor of the biliary tract,
although its overall incidence in Western countries ranges from 2 to 6 cases per 100 000 people [3].
It is divided into intrahepatic and extrahepatic subtypes,
of which the perihilar cholangiocarcinoma (or “Klastkin” tumor) account for the majority of cases.
Extrahepatic CC typically cause occlusion of bile ducts [4].
Patients are usually above the age of 65 and present with pruritus and jaundice.
There is increased risk of cholangiocarcinoma in patients with primary sclerosing cholangitis (PSC) and choledochal cysts due to chronic biliary inflammation.
Surgical resection is the only curative option,
although adjuvant or neoadjuvant chemotherapy may also be necessary.
Even following complete resection of perihilar tumors,
the subtype with the best prognosis,
5-year survival rate is 40% [3].
The median survival rate is 17-27.5 months for those ongoing resection with chemoradiation [3-5].
IMAGING OF CHOLANGIOCARCINOMA
Ultrasound is often the initial imaging modality demostrating biliary dilatation and a poorly defined hypoechoic mass.
When cholangiocarcinoma is suspected,
further workup is required and assessement made for resectability.
Options include multiphasic MDCT and ERCP and may be followed by MRI and MR cholangiography.
Imaging should ideally be completed prior to procedures,
such as drainage or stenting to reduce artifact.
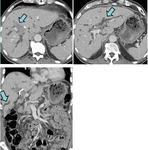
Arterial and portal venous phases are important for assessing the relationship of tumor to the hepatic artery and portal veins.
Key imaging features of cholangiocarcinoma include dilated segmental bile ducts with lack of communication between right and left hepatic ducts,
crowding of bile ducts,
ductal wall thickening and lobar atrophy (Fig. 2).
The subtypes of cholangiocarcinoma (intrahepatic,
perihilar and distal) can further classified according to morphology by the American Joint Committee on Cancer (AJCC),
into mass-like,
periductal and polypoid types each with its own specific imaging features [4,5].
Typical features of mass-like CC are peripheral enhancement in the arterial phase and centripteal enhancement in the portal venous phase with increased enhancement in the delayed phases in majority of cases [4,5].
3.
INTRADUCTAL PAPILLARY MUCINOUS NEOPLASM OF THE BILE DUCT (IPMN-B)
Intraductal papillary mucinous neoplasm of the bile duct (IPMN-B) is becoming recognized as its own distinct neoplastic entity.
Due to its similar histopathologic features to intraductal papillary mucinous neoplasm of the pancreas,
it has thus been refered to as such [6].
IPMN-B is unlike biliary mucinous cystic neoplasm (MCN),
since the former lacks ovarian-like stroma and communicates with the bile ducts.
Malignant IPMN-B is also unlike intrahepatic cholangiocarcinoma (ICC),
as it can be resected and demonstrates favorable prognosis.
Despite its relatively rare occurrence (2.9% to 8.9% of cholangiocarcinomas),
its specific features and treatability demand for correct imaging diagnosis for optimal patient management to ensue [6].
IMAGING OF INTRADUCTAL PAPILLARY MUCINOUS NEOPLASM OF THE BILE DUCT (IPMN-B)
Key diagnostic features of IPMN-B include the detection of mucobilia,
the distribution of dilated bile ducts,
the shape and detection of enhancement.
Unlike the mucin produced by biliary MCN which remains in a closed cyst and thus,
does not prevent bile flow; the opposite phenomenon is observed for the mucin found in IPMN-B.
Furthermore,
the majority of malignant IPMN-B demonstrates mural nodules or invasive tumors in the bile ducts,
where hyperenhancement on US,
CT and MRI can be an additional sign of malignancy [6].
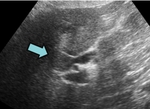
On ultrasound,
viscous mucin appears echogenic and occupies the bile ducts (Fig. 3),
although it is usually less viscous and anechoic,
such that irregular nodular intraluminal masses may be demonstrated.
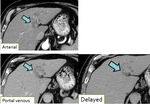
On CT & MRI,
a large mural nodule in the duct on contrast-enhanced studies is concerning for malignancy,
but small or flat tumors may be difficult to detect (Fig. 4).
ERCP however,
can depict mucin as an elongated filling defect in the bile duct which supplements information obtained on CT and MRI (Fig. 5 - Fig. 6).
Endoscopic ultrasound and intraductal ultrasound can provide precise information regarding continuation between the bile duct and the cystic IPMN-B [6].
NON-OBSTRUCTIVE CAUSES
1.
CAROLI DISEASE
Caroli disease is a congenital cystic dilation (usually large and saccular) of intrahepatic biliary ducts with a 1/100,000 incidence [7].
Pathologically,
it is derived from the malformation of the ductal plate,
a precursor to intrahepatic bile ducts,
causing bile ducts to be too numerous and ectactic.
The clinical presentation is with recurrent cholangitis,
portal hypertension and its derivatives (hematemesis from varices,
splenomgaly,
cirrhosis) in the pediatric population.
It is often associated with polycystic kidney disease [7].
The management of localized disease is segmentectomy or lobectomy,
while diffuse disease is managed conservatively,
although liver transplant is also considered.
Its prognosis is poor,
secondary to complications such as intraductal stones,
cholangitis,
abscess,
cirrhosis,
and an increased risk of cholangiocarcinoma.
IMAGING OF CAROLI DISEASE
The distribution of Caroli disease is segmental,
lobar or diffuse.
Its morphology is typically saccular (75% cases) dilatation of intrahepatic bile ducts as its hallmark feature,
although dilated extrahepatic bile ducts may also occur.
The sacculations appear as intrahepatic cystic water-density structures on CT and as anechoic areas on ultrasound.
The communication between the sacculations and bile ducts is important in distinguishing Caroli’s disease from polycystic liver disease [8].
One of Caroli disease’s characteristic signs is the “central dot sign” where a portal vein is surrounded by dilated bile ducts (Fig. 7).
On cholangiograms,
an alternating pattern of strictures and dilatations with possible stones are typifying features [9].
2.
CHOLEDOCHAL OR BILIARY CYSTS
Traditionally known as choledochal cyst,
which is a congenital dilatation of extrahepatic bile duct,
biliary cysts have now expanded to also include intrahepatic cysts [10].
Choledochal cysts are caused by dilatation of an anomalous pancreatico-biliary junction,
where the contraction of the sphincter of Oddi causes pancreatic enzymes to flow back into the cystic duct.
Patients are usually diagnosed under the age of 10 with palpable mass and jaundice.
Choledochal cysts are associated with cholangiocarcinoma and due to the increased risk of cholangiocarcinoma,
especially for the most common types (Todani types I & IV),
resection is recommended [10].
IMAGING OF CHOLEDOCHAL OR BILIARY CYSTS
A choledochal cyst is a dilated cystic structure that communicates with the biliary system and is separate from the gallbladder [11].
Ultrasound and CT can be used to identify the ductal dilatation (Fig. 8); however,
ERCP and MRI,
are required for better visualization of communication between the cyst and biliary tree.
Classification of choledochal cyst is by the Todani classficiation where Types I and IV are most common.
Type I is a true choledochal cyst with focal dilatation of extrahepatic duct and Type IV is the dilatation of the entire extrahepatic duct with involvement of portions of intrahepatic ducts [10].
3.
PRIMARY SCLEROSING CHOLANGITIS (PSC)
Primary sclerosing cholangitis (PSC) is a chronic progressive biliary disorder of unknown etiology causing inflammation,
fibrosis and strictures of medium and large ducts and/or extrahepatic biliary tree.
It is associated with inflammatory bowel disease (IBD).
Patients typically present with fatigue,
pruritis,
jaundice (obstructive),
and IBD findings,
where imaging is key in its diagnosis,
such as identifying cholangiographic evidence of bile duct changes.
Its prognosis is dependant on complications such as cholestasis and hepatic failure [12].
IMAGING OF PRIMARY SCLEROSING CHOLANGITIS (PSC)
Key imaging features include mild intrahepatic bile duct dilatation with multifocal strictures (short segments of 3-5mm) and ductal irregularity.
On ultrasound,
thickening of bile duct wall is one of the earliest features.
On CT,
discontinuous dilatation,
bile wall thickening at the level of the porta hepatis and lymphadenopathy can be noted (Fig. 9).
On cholangiography,
which is used in the initial diagnosis of disease,
a certain typical features can be noted.
These include beading (alternating strictures and normal ducts),
pruned-tree appearance (distal bile ducts are narrowed),
mural irregularity (irregular luminal margin) and diverticula (specific for PSC) [13].
4.
RECURRENT PYOGENIC CHOLANGITIS
Recurrent pyogenic cholangitis is the recurrent infections of the biliary system with formation of intrahepatic pigmented stones,
where the recurrent infection is often of unknown etiology,
although suspected to be due to biliary parasites such as Clonorchis,
amongst others.
Patients present with a septic picture,
similar to recurrent cholangitis as a result of superadded bacterial infection.
The acute complications require urgent supportive management,
such as decompression of the infected biliary tree (which can be accomplished by percutaneous transhepatic biliary drainage or endoscopic sphincterotomy with stent placement),
and clearance of calculi (with percutaneous transhepatic cholangioscopic lithotomy (PTCSL) and peroral cholangioscopy).
However,
prevention of long-term complications requires surgical resection,
clearance of stones with minimally invasive techniques.
Long-standing infections lead to complications such as biliary cirrhosis and cholangiocarcinoma,
which can be responsible for increased patient mortality [14].
IMAGING OF RECURRENT PYOGENIC CHOLANGITIS
The initial evaluation is usually with ultrasound where ductal diltation and stones are demonstrated in 85-90% of cases.
Additional assessment may involve CT and cholangiography (with ERCP,
MRCP or PTC).
Features include intra- and extrahepatic duct dilatation with enhancement of duct walls,
straightened intrahepatic ducts with less acute or right-angled branching patterns (as a result of extensive periductal fibrosis),
decreased arborization,
acute tapering of the periheral ducts (classic “arrowhead”),
“missing duct” sign due to a complete obstruction of a bile duct,
hepatic abscesses,
bilomas,
and stones (Fig. 10 - Fig. 11) [15-17].
Clinical context is key,
as intrahepatic lithiasis with focal dilatation can be indistinguishable from focal Caroli’s disease and requires close correlation with history and patient demographics.