1.
Lung volume reduction surgery (LVRS)
The surgical removal of emphysematous tissue has the purpose of revert ventilation in residual healthy parenchyma [2],
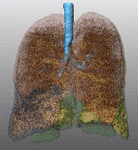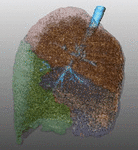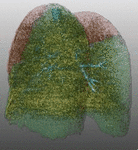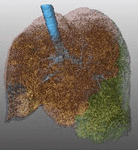
specifically in patients suffering from upper-lobes predominant emphysema (Fig.
1); therefore,
the pre-operative assessment of emphysema through visual scoring or by quantitative analysis of CT images is needed; the latter precisely calculates the percentage of emphysematous tissue,
thus guiding the correct strategy [3].
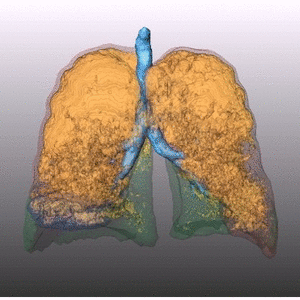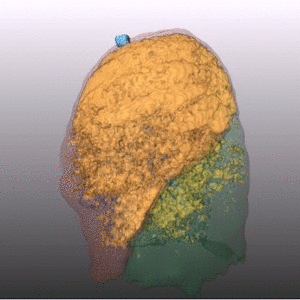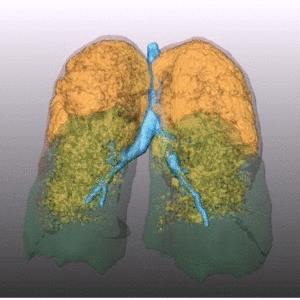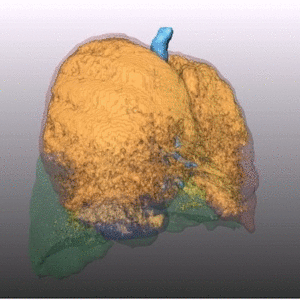
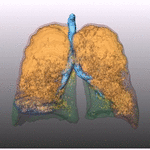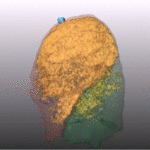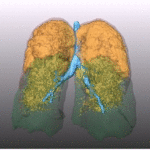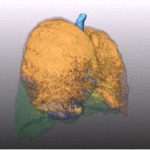
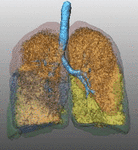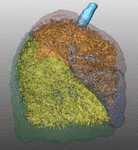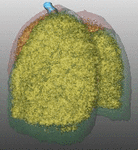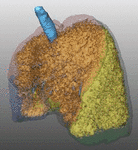
Fig. 1: Pattern of emphysema: the quantitative evaluation of emphysema by dedicated semi-automatic software provides three-dimensional display of emphysema distribution. In quantitative imaging, emphysema is defined by voxel density <-950 HU. Volume rendering reconstruction of volumetric CT shows upper lobe predominant emphysema, which can be treated by either surgical or bronchoscopic approach (EBV or LVRC). The evaluation of fissures and other parenchymal anomalies, namely bronchiectasis or bullae, is mandatory to guide the treatment decision. Notably, fissure integrity reflects absence of major collateral ventilation and is a necessary selection criteria for EBV positioning. Conversely, fissural defects (>10% on axial plane) and a residual amount of healthy tissue can be treated by LVRCs.
Two different surgical approaches can be performed,
namely the video-assisted thoracoscopic surgery (VATS) and the median sternotomy (MS) surgery (Fig.
2).
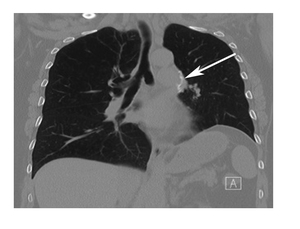
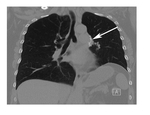
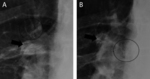
Fig. 2: Post operative finding after lung volume reduction surgery (LVRS): coronal chest CT after LVRS shows increased opacity in the left hilum with metallic staple lines (white arrow) that are usually positioned during the surgical procedure. These are normal findings that should not be misdiagnosed as complications.
References: Anand Devaraj, Royal Brompton Hospital, London, UK
The mortality-rate during the first three months is about 5% [4] and the most common complications are air-leak (in 90% of cases within 30 days of thoracotomy) and pneumonia (18.2%) [5]; chest x-ray or CT are useful for diagnosis and monitoring of these complications.
2. Endobronchial valve (EBV)
Endobronchial valves,
positioned under bronchoschopic guide into lobar,
segmental or subsegmental bronchi,
reduce the flow of air into the target lobe during inspiration and let air and secretions out during expiration,
thus producing a stable volume decrease in the target lobe [6] (Fig.
3 – Fig.
4).
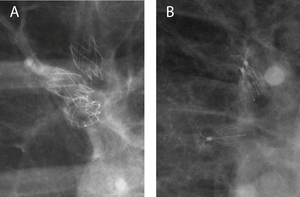
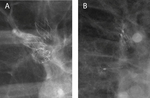
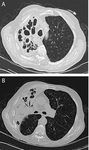
Fig. 3: Radiographic evaluation after positioning of endobronchial valves (EBV) currently available for treatment as for Conformitè Européenne (CE) qualification: (A) magnification of a chest X-ray in a patient treated with the duckbill EBV (Zephyr; Pulmonx, Inc., Palo Alto, CA, USA). (B) Magnification of a chest X-ray in a patient treated with the umbrella-shaped intrabronchial valve (Spiration; Olympus Respiratory America, Redmond, WA, USA).
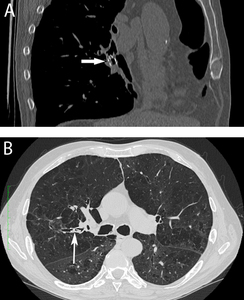
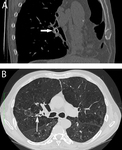
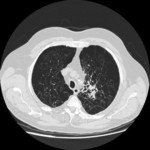
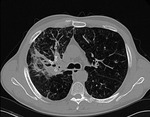
Fig. 4: Endobronchial valves: computed tomography (CT) multiplanar reformatted image (A) of oblique sagittal plane, in a patient treated with endobronchial valve (Zephyr; Pulmonx, Inc., Palo Alto, CA, USA), visible as a metallic structure in the bronchus lumen (white arrow). Axial chest CT with lung window reconstruction (B) after valve positioning in posterior segmental bronchus of the right upper lobe (Spiration; Olympus Respiratory America, Redmond, WA, USA). The valve is detectable as high-density linear structure within the bronchial lumen (white arrow).
Although atelectasis is associated with the most favorable outcome [7,
8],
it is infrequent or even transient [9] (Fig.
5 – Fig.
6).
Nevertheless,
there may still be clinical benefit because air is directed towards less damaged parenchyma [10].
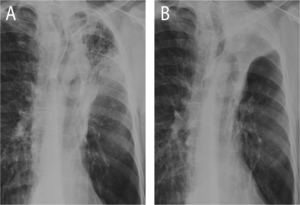
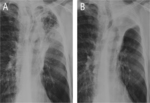
Fig. 5: Atelectasis after endobronchial valve treatment: chest X-ray 1 day after valve positioning in upper lobe predominant emphysema with consequential irregular consolidation (A). Follow up after 5 weeks show progression of consolidation towards sharply demarked opacification reflecting atelectasis (B), a sign of successful treatment after EBV positioning.
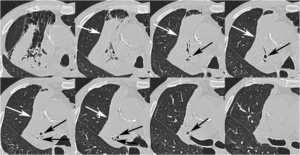
Fig. 6: Atelectasis after endobronchial valve treatment: axial chest CT scans after positioning of EBV (black arrows) show volume reduction of the right upper lobe.
Pre-operative selection of the lobes more severely affected and the evaluation of the degree heterogeneity of emphysema are mandatory.
Subjects with either upper or lower lobe predominant emphysema can undergo EBV positioning (Fig.
1 - Fig.
7).
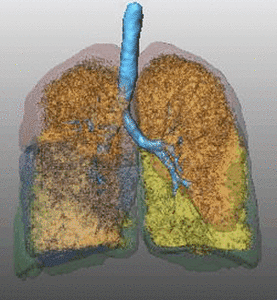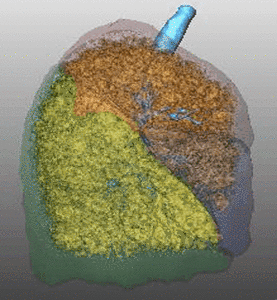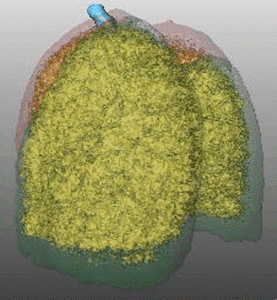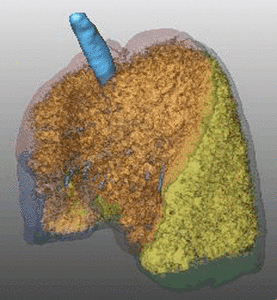
Fig. 7: Pattern of emphysema: volume rendering reconstruction of volumetric CT shows lower lobe predominant emphysema. Such distribution is better treated by EBV and LVRC. Likewise upper lobe predominant emphysema, fissural evaluation is mandatory during the pre-operative planning to guide bronchoscopic treatment.
Clinical improvements is optimal when lobar exclusion is achieved,
namely when there is no collateral ventilation (CV) between the target and the adjacent lobe.
HRCT volumetric acquisition associated with multiplanar reconstructed images is needed for the detection of fissural defects,
as those anatomical defects are the main cause of collateral ventilation.
A fissure is complete when it is assessable over the 90% of the CT slices [11].
Fissural defects are difficult to detect in emphysematous lung because the anatomy is altered by hyperinflation and bullae.
Perihylar portions are the most difficult to assess,
and also the most frequent location of fissural defect.
Computed tomography measurement of the bronchial lumen diameter is needed to select the appropriate valve size (e.g.
5,
6 or 7 mm) [12].
The most common complications after EBV are pneumothorax and pulmonary infection [13] (Fig.
8 – Fig.
9 - Fig.
10); valve migration has been reported in about 5% of patients (Fig.
11 – Fig.
12).
Chest radiograph and CT can detect all of these complications [14].
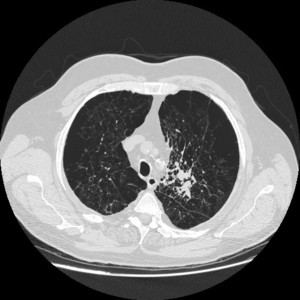
Fig. 8: Infectious consolidation after endobronchial valve positioning: axial chest CT with lung window reconstruction after EBV positioning in the left upper lobe shows ill-defined consolidation, reflecting infectious complication.
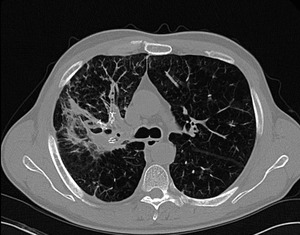
Fig. 9: Consolidation after endobronchial valve positioning: maximum intensity projection (MIP) reconstruction on the axial plane of a patient with severe emphysema treated with endobronchial valves in the right upper lobe. The reconstruction highlights the complex structure of the valves. The consolidation distal to the valves shows irregular margins and peribronchial distribution, which reflect infectious pneumonia rather than atelectasis.
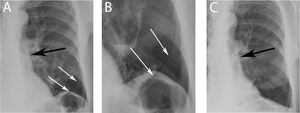
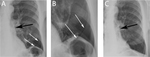
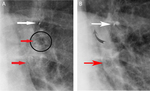
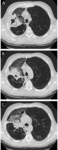
Fig. 10: Pneumothorax after EBV: chest X-ray (A) and its magnification (B) show hyperlucency in the left costo-phrenic sulcus (white arrows) after EBV positioning (black arrow), reflecting pneumothorax (prevalence 6% after treatment by EBV). Resolution of pneumothorax is documented by chest X-ray follow-up (C).
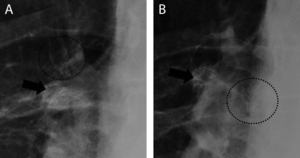
Fig. 11: Migration of an endobronchial valve: chest X-ray (A) performed during the follow-up after EBV positioning in the apical and anterior segmental bronchi of the right upper lobe. (B) The valve previously deployed in the apical bronchus has migrated into the right main bronchus.
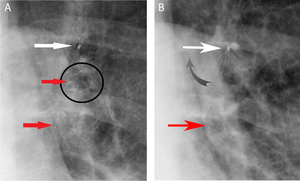
Fig. 12: Expulsion of an endobronchial valve: magnification of chest X-ray (A) shows three bronchial valves in the left upper lobe, respectively two Zephyr valves (red arrows) and one Spiration (white arrow). (B) Magnification of chest X-ray depicts the migration of a Zephyr valve (black circled in A), which cannot be seen; indeed, the patients reported the expulsion of the valve during cough.
3. Lung volume reduction with coils (LVRC)
Coils for lung volume reduction are wires that shrink the emphysematous lung parenchyma after their deployment into the selected airway,
resulting in a reduction of hyperinflation (Fig.
13 – Fig.
14).
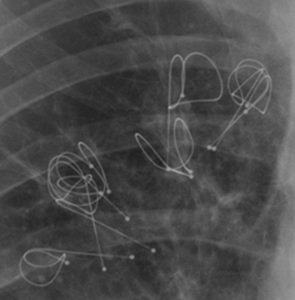
Fig. 13: Lung volume reduction coil (LVRC): chest X-ray magnification shows LVRC RePneu coils (PneumRx, Inc., Mountain View, CA, USA) in right upper lobe. LVRC are wires made of Nitinol, a nickel and titanium alloy that combines memory shape effects, strength, and elasticity. Up to 10 LVRCs can be positioned into the target lobe during a single procedure.
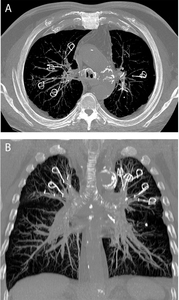
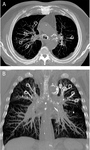
Fig. 14: Lung volume reduction with coils: maximum intensity projection (MIP) reconstructed images in the axial (A) and coronal (B) plane of a patient with severe upper lobe predominant emphysema treated with bilateral positioning of LVRC. Pulmonary volume loss was achieved without atelectasis, which is a rare finding after LVRC indeed.
Procedure is usually unilateral to limit the likelihood of complication and up to ten coils can be positioned in a single procedure [15] (Fig.
15).
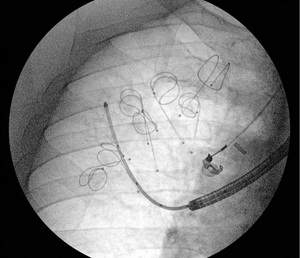
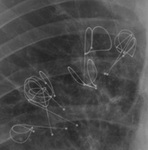
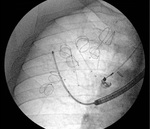
Fig. 15: LVRC positioning: fluoroscopic image shows seven LVRCs in the right upper lobe and the catheter that is used for their positioning. Before positioning, the coils are straightened and they regain their original shape once the catether is removed.
Coils are partially irremovable and can be efficacious in patients with either heterogeneous or homogeneous emphysema,
regardless of the presence of collateral ventilation [16-19] (Fig.
1 - Fig.
7 - Fig.
16).
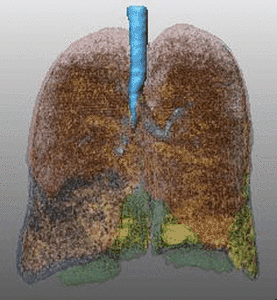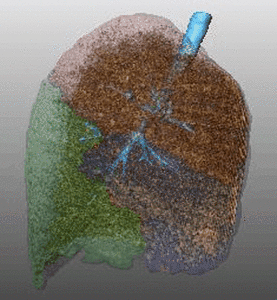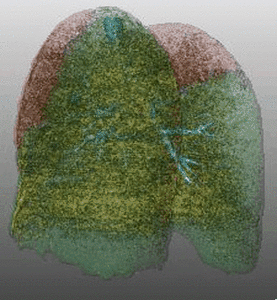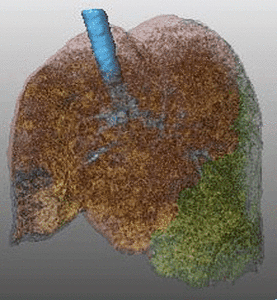
Fig. 16: Pattern of emphysema: volume rendering reconstruction of volumetric CT shows homogeneous distribution of emphysema, which can be treated by LVRC but EBV.
Bronchiectasis,
large bullae,
and massive destruction of lung parenchyma are specific contraindications for LVRC.
Frequent early complications after LVRC are pulmonary infection and pneumothorax [16]; coils do not migrate (Fig.
17 – Fig.
18).
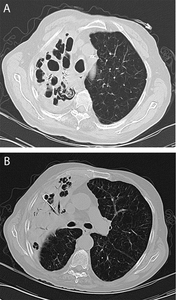
Fig. 17: Pneumonia after coils lung volume reduction: axial chest CT with lung window reconstruction (A, B) in the follow-up of LVRC shows subtotal opacification of the right upper lobe. Opacification after LVRC positioning is an uncommon finding after LVRC, that can reflect either atelectasis (extremely rare) or pneumonia. In this case, the irregular morphology of the consolidation and air collections are consistent with pneumonia.
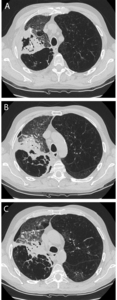
Fig. 18: Multiple synchronous complications after LVRC: axial chest CT images with lung window reconstruction (A, B, C) show synchronous consolidation and pneumothorax (white arrow) after LVRC positioning. Pneumothorax and infectious complications are the most frequent complications after LVRC or EBV.