RECIST CRITERIA
The original Response Evaluation Criteria In Solid Tumors (RECIST version 1.0) were proposed on 2000 jointly by several scientific Organizations,
including WHO [8]. They evaluated modifications of longest diameter of the neoplasm,
and progressive disease was defined as the appearance of new lesions or a greater than 20% increase in the longest diameter.
Due to the lack of assessment of lymph nodes and to the development of newer imaging technologies,
the RECIST Working Group revised the original criteria in 2009 [9].
According to RECIST 1.1,
complete response is defined when we have disappearance of all target lesions or lymph nodes dimensions <10 mm in the short axis; partial response with a >30% decrease in sum of longest diameters of target lesions; progressive disease with >20% increase in longest diameters of target lesions,
and stable disease for all the other cases.
Unfortunately,
RECIST 1.1 criteria for the evaluation of PDAC response to chemotherapy present some limits: first,
a spherical shape of the tumor is assumed; as demonstrated by several studies,
PDAC presents ill defined and irregular margins,
and very rarely a spherical shape.
In addition,
tumor density is not evaluated; as reported by Ferrone et al.
[10],
PDAC could respond to therapy only with modifications in attenuation at CT,
maintaining the same dimensions.
CHOI CRITERIA
In 2007 Choi et al have developed new criteria for evaluating treatment response of gastrointestinal stromal tumors (GIST),
which assess a change in size or a change in density of target lesions [11].
These criteria were developed because of the introduction of new molecular drugs,
competitive inhibitors of tyrosine kinase receptor (for example,
Imatinib).
During the course of treatment with inhibitors of tyrosine kinase receptor tumor size usually decreases,
but in some cases size may remain stable or even increase,
secondary to internal hemorrhage or necrosis.
So,
for GIST or other hypervascular neoplasms,
like hepatocellular carcinoma or neuroendocrine tumor,
Choi criteria appear to be better predictors of clinical response than RECIST.
In particular,
partial response is defined when we have ≥10% decrease in tumor size or ≥15% decrease in tumor attenuation at CT.
Unfortunately,
PDAC is a hypo-vascularised neoplasm,
hypodense at CT and hypointense at MRI images in the arterial-pancreatic phase.
So,
as reported by Cassinotto et al.
[12],
and as we have observed in our Institution,
several responder patients affected by PDAC present an increase of tumor density both in basal conditions and in the arterial-pancreatic phase.
Consequently,
Choi criteria are not indicated for the evaluation of PDAC treatment response.
PERCIST CRITERIA
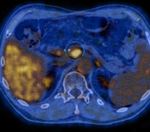
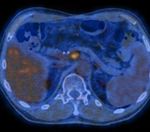
PET Response Criteria in Solid Tumors (PERCIST) were first proposed in 2009 [13] to assess tumor response with Positron Emission Tomography (PET) in a standardized and objective way.
Because many newer cancer therapies present a cytostatic effect instead of a cytocidal,
good response may be associated predominantly with a decrease in metabolism,
without a significant reduction in tumor size.
Response to therapy is expressed as a percentage change in the peak of SUL (lean body mass–normalized SUV) for the most active lesion between the pre- and post-treatment FDG-PET/CT studies.
A complete metabolic response is defined as disappearance of all metabolically active tumors.
A partial metabolic response is defined as a >30% decline in SUL peak.
A >30% increase in SUL peak or the appearance of a new lesion is classified as progressive metabolic disease.
It is fundamental to perform a standardized PET/ CT scanning protocol,
with accurate injected dose,
post-injection delay,
reconstruction parameters and SUV normalization technique.
There are difficulties regarding the high costs,
imaging standardization across PET centers and concerning the timing of assessment relative to treatment,
so the use of quantitative measurements of FDG uptake for evaluating response remains difficult.
However,
according to several studies and to the experience developed in our Institution,
FDG PET is becoming the gold standard technique for assessing tumor response of most solid tumor,
including PDAC.
MRI WITH DIFFUSION WEIGHTED IMAGING
Nowadays,
diffusion weighted imaging (DWI) is part of every abdominal MRI examinations,
thanks to its high sensitivity to depict small,
ill-defined lesions.
It is a form of MR imaging based upon measuring the random Brownian motion of water molecules within a voxel of tissue; densely cellular pathological lesions (like PDAC) exhibit lower diffusion coefficients compared with surrounding healthy parenchyma.
According to several studies and to our experience,
DWI is very helpful to distinguish between residual/recurrent tumor and fibrotic tissue,
after chemotherapy but even after pancreatic surgery [14].
It is also important for the depiction of pathologic lymph nodes,
brightly hyperintense in high b-values images.
Even if it is possible to perform quantitative evaluations on automatically reconstructed Apparent Diffusion Coefficient (ADC) maps,
it is difficult to attribute to the neoplasm specific ADC values,
due to the presence of several confounding parameters (different MR units,
coils,
sequence parameters,
vascularization,
temperature of the room).
So,
DWI evaluation has a cardinal role in assessing treatment response,
but it remains essentially a qualitative technique.
TEXTURE ANALYSIS
The texture of images refers to the appearance,
structure and arrangement of the pixels/voxels within the image.
Texture features are mathematical parameters computed from the distribution of pixels,
which characterize the texture type and thus the underlying structure of the organs shown in the image.
Although changes in density can be easily observed during the course of chemotherapy,
changes in tumor heterogeneity,
as measured with texture analysis,
are often more apparent.
This method measures changes in tumor appearances,
using established image processing tools to quantitatively describe the homogeneity or heterogeneity of gray-scale images.
Several ongoing studies are investigating the prognostic role of texture analysis in tumor response [15].
In our Institution we have performed some analysis with MaZda 4.6 freeware software,
to assess changes in mean and variance density values,
but also in kurtosis values and skewness histograms.
We have observed that after FOLFIRINOX in responders PDAC patients there is reduction of tumor volume and longest dimension,
but also increase of mean and variance density values,
increase of kurtosis and modifications of skewness histograms.
These changes are not significant in non responder patients.
So,
although this method is still experimental and requires a specific training,
it is very promising for its low costs and the possibility of retrospective evaluations.