Computed tomography is useful not only for detecting signs of aneurysm´s rupture,
but also for planning surgical and/or endovascular repair.
Therefore,
there are some essential points our radiological report must include in order to help chose the most suitable treatment for the patients (Fig. 2):

Fig. 2: Main points an abdominal aortic aneurysm rupture must include.
1.
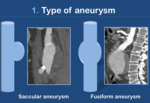
Type of aneurysm (Fig. 3)
a.Saccular: Those aneurysms that involve only part of a vessel wall.
These type of aneurysms are rare (5% of abdominal aortic aneurysms),
more frequently seen in infectious or inflammatory diseases,
and usually located in the suprarenal aorta.
b.Fusiform: Those aneurysms that involve the whole wall of a vessel.
They are by far the most common type of abdominal aortic aneurysm (95%).
2.
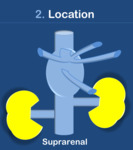
Location
a.Suprarenal (Fig. 4) : Aneurysms that include the level of the renal arteries and/or extend superiorly.
In the setting of a rupture,
these aneurysms are usually treated with open surgical repair,
as they often involve the visceral arteries and need custom-made branched or fenestrated endoprosthesis.
b.Juxtarenal (Fig. 5): Aneurysms that extend immediately below the level of the renal arteries with a normal diameter of the suprarenal aorta.
As in suprarenal aneurysms,
they may require custom-made endoprosthesis.
c.Infrarenal (Fig. 6): Aneurysms that arise below the renal arteries.
These are the most common type of abdominal aortic aneurysms,
and the most suitable type for endovascular repair.
3.
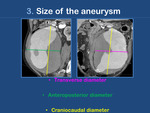
Size (Fig. 7)
The bigger the size of the aneurysm,
the higher the risk of rupture.
However,
only the transverse and anteroposterior diameters have an influence in the risk of rupture.
The craniocaudal diameter only helps in planning the surgery/EVAR.
4.
Signs of rupture:
Abdominal aortic aneurysm ruptures are more common in the posterolateral walls of the aorta.
Therefore,
it is above all in this location where we must look for signs of its rupture.
I - Primary signs ( Fig. 8 )
a.
Contrast extravasation: It is the most specific sign of a complete AAA rupture.
On arterial phases,
contrast can be seen scalping through the boundaries of the aneurysm wall into the retroperitoneal space.
If there are doubts of the existence of contrast extravasation in the arterial phase,
venous or delayed phases are recommended to be performed to confirm or exclude it.
b.
Peritoneal/retroperitoneal haematoma:
-Retroperitoneal haematoma: It typically affects periaortic regions,
perirenal and pararenal spaces,
but sometimes may not be evident.
In these cases,
indirect signs such as increasing volume of the psoas,
anterior displacement of the bowel towards the abdominal wall and anterior or displacement of the kidneys may be helpful.
-Peritoneal hematoma: It is seen in anterior or anterolateral rupture of the aneurysm wall and it is very important to mention in the report if present,
as it has a worse prognosis than a retroperitoneal hematoma.
-Aorto-enteric or Aorto-caval fistula: In some rare cases,
the ruptured aorta may drain into a small bowel loop (aorto-enteric fistula) or the inferior vein cava (aorto-caval fistula) (Fig. 9).
c.
Periaortic stranding: It represents edema of the periaortic fat,
and on CT,
it is seen as an area of high-attenuating fat surrounding a large aneurysm that may range from a discrete peripheral infiltration to a coarse linear pattern.
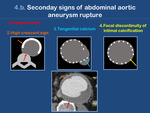
II - Secondary signs (contained rupture) (Fig. 10)
a.
Drapped aorta sign: It is the impossibility to differentiate the posterior wall of the aorta from adjacent structures,
or the presence of a posterior wall of the aorta that closely follows the contour of the spine on one or both sides.
b. Hyperattenuating crescent sign: It is a well-defined peripheral crescent of increased attenuation (higher than the psoas muscle in enhanced exams and higher than the aortic lumen in unenhanced exams) that represents an acute hematoma within the thrombus or in the aneurysm wall,
and is one of the first and most specific manifestation of impending rupture.
c.
Tangential calcium: The intimal calcification of the aneurysm points away from the aneurysm.
d.
Focal discontinuity of intimal calcifications: Aortic aneurysm wall usually contains calcifications in the intima that follow the circumference of the aortic lumen.
The presence of an area of discontinuity in mural calcifications,
above all if there are previous examinations to compare with and this area is new,
suggests contained ruptured.
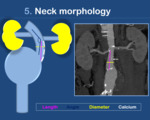
5.
Neck morphology (Fig. 11,
Fig. 12)
a.
Neck length: It is defined by the distance between the inferior border of the lowest renal artery and the upper border of the aneurysm.
It must be of at least 1.5 cm for endovascular repair.
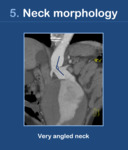
b.
Neck angle: It is the angle between the suprarenal aorta and the aortic neck.
Angles <120° are not suitable for EVAR.
c.
Neck diameter: Neck diameters lower than 28 mm are ideal for EVAR.
d.
Calcifications and thrombus: Their presence in the neck of the aneurysm may not allow for EVAR.
6.
Mural thrombosis and calcified atheromatosis (Fig. 13)
The assessment of mural thrombosis and calcified atheromatosis of the aneurysm sac must be included in the report,
as significant stenosis or occlusion may not allow for EVAR.
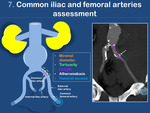
7.
Common femoral and iliac arteries (Fig. 14,
Fig. 15)
If in the setting of an abdominal aortic aneurysm rupture an EVAR wants to be performed,
the anatomy and morphology of the common iliac and femoral arteries needs to be assessed,
as they are important for vascular access and device introduction,
and for adequate fixation at the distal attachment site and maintenance of limb patency (Fig. 16).
a.
Common iliac arteries caliber: The minimal outer diameter of the iliac arteries must be of at least 7 mm for placing a device.
A dilated iliac artery (considered more than 22 mm,
and present in up to 20% of the patients with AAA) affects the distal seal zone,
preventing successful fixation of the graft limb (Fig. 15).
b.
Common iliac arteries tortuosity: Very tortuous iliac arteries make it more difficult for the devices to pass through them and may not allow for EVAR (Fig. 15).
c.
Common iliac arteries calcification: Stenotic or occlusive disease prohibits delivery of the stent-graft.
Calcifications greater than 50% of the circumference are considered severe and make the device delivery difficult.
In addition,
they also have a potential risk of embolization.
d.
Common iliac arteries length: Adequate length of the common iliac arteries is need to ensure the prosthesis´ positioning and distal sealing.
If the common iliac artery is too short,
extension of the aortic stent-graft into the external iliac artery,
which complicates the procedure,
may be needed.
A length of at least 3 cm is considered ideal for EVAR.
e. Transfemoral access: Limited transfemoral access (stenosis,
calcium,
occlusion…) may preclude the possibility of an EVAR.
Treatment
In general terms,
infrarenal ruptured AAA with favorable anatomy (points 5,
6 and 7) undergo endovascular repair,
whereas suprarenal and juxtarenal,
as well as infrarenal with unfavorable anatomy ruptured AAA undergo open surgery. However,
unfortunately,
because of differences in interventional experience levels amongst physicians,
anatomical inclusion criteria for EVAR still vary widely amongst medical centres.
Morover,
other factors such as the disponibility of an on call interventional radiologist and/or vascular surgeon,
and the availability of different types of endoprosthesis (e.g.,
there are some prosthesis that permit a higher degree of angulation of the neck of the aneurysm) must also be taken into account in order to chose the most suitable treatment for the patient.
( Fig. 17 ).