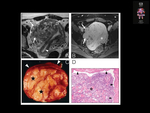
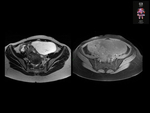
Leiomyomas are neoplastic nodular and/or diffuse proliferation of histologically benign smooth muscle cells (figure 1).
By the way,
extrauterine intravenous leiomyomatosis are rare and represent intraluminal growth into either venous or lymphatic vessels outside the limits of myoma.
Intravenous leiomyomatosis may result from the invasion of myometrial sinuses by either leiomyomatosis cells or proliferating smooth muscle cells,
arising directly from the venous wall of the uterine or pelvic veins and are associated with high estrogen levels.
First described by Birch-Hirschfeld in 1896 and defined by Norris and Parmlyin in 1975,
are more commonly seen between 40 and 50 years old women and are diagnostic dilemma of benignity,
once the most of patient are asymptomatic despite extensive intravenous extension and benign metastasizing leiomyomatosis (most often with metastatic lesions within the lungs) and with disseminated peritoneal leiomyomatosis following surgery have been reported.
Commonly extends into the pelvic veins and manifests as worm-like protrusions of tumor emanating from veins at the parametrial margins.
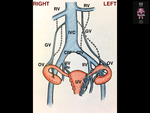
Initial route outside the uterus for “intrauterine” intravenous leiomyomatosis may include the broad ligament veins and then enter the systemic venous circulation via the uterine or ovarian veins into two (unilateral) routes (figure 2):
- uterine vein following the internal iliac vein,
followed by common iliac vein,
then inferior vena cava
- ovarian vein following common iliac vein,
gonadal vein then inferior vena cava (or renal vein then IVC,
if left side)
Bender et al describes that extrauterine cases are 30-80% and has cardiac involvement on 10-30%.
MRI and histopathologic study have important role on this entity during description and involvement evaluation,
and on differential diagnosis,
respectively,
like:
•Leiomyosarcoma
•Parasitic leiomyoma
•Pedunculated leiomyoma
•Endometrial stromal sarcoma
•Disseminated peritoneal leiomyomatosis (figures 3 and 4)
At MR imaging,
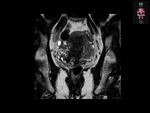
the intravascular tumor manifests as a mass of distended and folded tubular structures (“sausage-like”,
figure 5).
Iso to mildly hyperintense on T1-weighted images and mildly to markedly hyperintense on T2-weighted images,
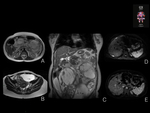
depending on the number of smooth muscle cells and fibrous tissue–containing hyalinized vessels the tumor contains with enhancing (figures 6 to 10).
Successful clinical management is dependent on total surgical excision,
including resection of the affected veins,
hysterectomy,
and bilateral oophorectomy.
Preoperative and postoperative antiestrogenic therapy facilitate debulking surgery and to prevent or delay recurrence,
respectively.
Recurrence occur up to 15 years after surgery.
Bender et al suggests 3,
6 and 12 months,
then annually imaging control with doppler US (including inferior cava vein and iliac veins study) and MRI.
If complete excision of the tumor is not possible,
ligation of the upstream vein is suggested.