Multi-parametric magnetic resonance imaging (mpMRI) has been validated in the detection and characterization of prostate tumours and can help in risk stratification and treatment selection.
In 2012,
the European Society of Urogenital Radiology (ESUR) established the Prostate Imagingd Reporting and Data System (PI-RADS) guidelines with the aim of standardising the acquisition,
interpretation and reporting of prostate MRI.
In 2014,
a joint committee formed by the ACR,
ESUR,
and the AdMeTech Foundation announced an updated version (PI-RADS v2).
Prostatic mpMRI indications:
- Local staging
- Cancer detection (with negative biopsies and clinical suspition)
- Better characterization of known alteraions
- Biopsy guidance
- Active vigilance
- Suspicion of recurrence
Main differences between PI-RADS v2 and v2 ( Fig. 1 )
For PI-RADS v1,
it was not specified exactly how to combine the scores from each MRI sequence,
therefore variable approaches were used.
PI-RADS v2,
proposes a specific algorithm for deriving an overall assessment on the basis of a lesion’s individual scores on T2WI,
DWI,
and DCE.
MR spectroscopic imaging,
which is an optional parameter in PI-RADS v1,
does not influence the assessment of lesions in PI-RADS v2.
On the other hand,
the interpretation of DCE-MRI was simplified and is now a binary assessment of the presence or absence of focal early enhancement.
Kinetic curves,
pharmacokinetic analysis,
and contrast washout are no longer included.
Normal 0 false false false PT JA X-NONE
A size threshold of 15 mm is suggested for differentiating between T2WI and DWI scores of 4 and 5.
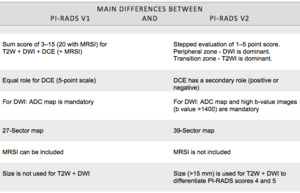
Fig. 1: Main differences between PI-RADS v1 and v2
PI-RADS v2 has more specific criteria and the classification is based on the individual characteristics of each lesion,
simplifying and reducing the variability of its application.



