Appropriate evaluation of the pituitary gland requires knowledge of the normal and its variants.
While in young children the gland has an upwards convex shape,
in adults is flat.
Glandular height more than in 8 mm in males and 9 mm in women is rare under normal circumstances,
the infundibular diameter is less than 3 mm.
There is a physiologic size increase of approximately 0.08 mm per week during pregnancy but it rearly exceeds 10 mm,
the maximum diameter achived immediately postpartum and recovery within 6 months after delivery.
MRI is the cornerstone for imaging pituitary lesions and can often reveal characteristics that help establish a diagnosis in most cases.
Lesions other than adenoma such as craniopharyngiomas,
meningiomas,
germ cell tumors,
gliomas,
metastases,
lymphomas,
vascular malformations,
inflammatory and other less common lesions should be in the differential diagnosis.
However,
computed tomography still have some applications in delineating disease.
In the perisellar region,
evaluate size,
localization and extent of a lesion,
if it is lateral,
superior or posterior to the dorsum sellae or infrasellar (sphenoidal).
Look for invasion of either the sphenoid cavity or cavernous sinus.
Unilateral carotid artery encasement,
with visible tumor lateral to the vessel,
is the most specific sign of cavernous sinus invasion by a macroadenoma.
Other signs of invasion are an expanded sella or destroyed inferior cortex on computed tomography as well as absence of cavernous sinus enhancement con contrast enhanced MRI.
Fat suppressed post contrast T1WI is usefull for assessing marrow and cavernous sinus invasion as well as internal carotid encasement.
By far the most common lesion in the pituitary gland are adenomas,
representing around 15% of intracranial tumors.
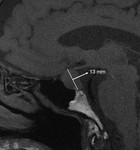
They are divided into microadenomas (below 10 mm) (Fig. 3) and macroadenomas (diameter greater than 10 mm) (Fig. 4).
They are usually detected by imaging when bigger than 4 mm and although microadenomas are usually small and do not create mass effect,
they signal their presence by signs and symptoms of hormonal excess,
nonfunctioning adenomas comprising 25-50% or all surgically resected pituitary tumors.
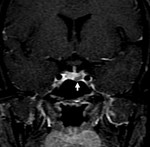
Microadenomas are rounded,
oval,
triangular of flattened hypointense regions in the anterior gland and isointense to the grey matter in the temporal cortex on T1,
but it can also be isointense making dynamic enhancement with contrast medium very useful.
Because they are less vascular than the surrounding tissue,
so almost immediately upon contrast administration,
normal pituitary gland will enhance,
leaving a microadenoma hypointense compared with normal pituitary gland (Fig. 3).
That is why scans are taken at 20-30 second intervals after administration of intravenous injection of gadolinium contrast agent,
and at some point after the administration the adenoma “wash in will equal the pituitary gland “wash out”.
Other indirect signs described in the literature are deviation of the infundibulum,
upward bulging of the sellar content into the opticochiasmatic cistern,
or a localized depression of the sellar floor.
They can have hemorrhagic or cystic changes,
altering the signal intensity.
There is potential for misinterpretation,
mainly due to the small size of the lesions that alter the intensity of the gland in ways that can mimic other lesions (e.g.,
cysts of the pars intermedia or due to artifacts,
such as partial volume effects),
thus such findings deserve special attention.
Pituitary macroadenomas are easier to detect,
since they are bigger,
and are reliably visualized with routine MR imaging.
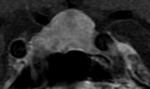
They may be inhomogeneous (Fig. 5) due to regressive changes and because of their size they can displace the chiasm,
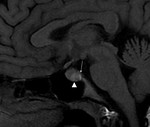
in some cases even the visual pathways may not be detected (Fig. 6).
Craniopharyngiomas
They are congenital,
benign but locally invasive,
often partially cystic,
epithelial neoplasms that arise from squamous epithelial rests of the Rathke pouch.
They account for approximately 3% of all intracranial tumors.
There are 2 types,
the Adamantinomatous (cystic mass),
Papillary (solid mass) and mixed.
Most tumors often have both cystic and solid components.
Craniopharyngiomas exhibit a bimodal age distribution,
with one peak in childhood (5-15 years) and the other in people over 50 years.
Children often present with symptoms of intracranial pressure,
growth disturbance and visual dysfunction,
while adults more often present with symptoms of endocrine dysfunction.
Characteristically,
craniopharingiomas demonstrate well defined lobular lesions in the suprasellar space.
They are the 90% tumors,
roughly 90% are cystic,
90% are calcified and 90% enhance with contrast media.
The papillary type rarely calcifies and is found mainly in adult population.
The variable composition of these tumors is reflected in the heterogeneous and variable internal architeture on CT and MRI.
Solid portions of craniopharyngiomas have variable intensity on T1WI and are generally hyperintense on T2WI.
Cystic components are T2 hyperintense.
There is homogeneous enhancement of the solid portions and rim enhancement of cyst walls with contrast administration (Fig. 7).
When the diagnosis is doubtful,
use of non-enhanced computed tomography is useful to detect calcifications because they can escape MRI detection.
Magnetic resonance angiography is useful for evaluation of vascular displacement and encasement and spectroscopy can show a broad lipid spectrum in the cystic component.
To differentiate with a Rathke’s cleft cyst,
they are entirely cystic lesions,
well defined in the midline and do not have solid or calcified portions.
Also,
craniopharyngiomas are usually bigger (>2 cm in diameter),
tend to be more lobulated and can produce compression of the third ventricle.
Treatment is usually surgical with radiotherapy especially useful for incomplete resection.
Craniopharygiomas frequently recur after treatment and can cause local invasion to adjacent structures.
Special attention should be placed on post treatment imaging in search for hypothalamic injury,
endocrine symptoms,
vasa variorum injury and pseudoaneurysm.
Rathke’s Cleft Cysts
Also known as pars intermedia cysts they are frequent congenital lesions,
found in around 15-22% of autopsies in normal population.
They arise from Rathke’s cleft,
and,
like craniopharyngiomas,
they are a remnant of the Rathke pouch,
an ectodermal extension of the embryonic oral cavity.
The Rathke pouch is a precursor of the anterior lobe,
pars intermedia and pars tuberalis of the hypophysis.
They are usually incidentally identified.
They are small lesions and thus asymptomatic,
rarely they have the size to produce vision disturbances,
headache,
diabetes insipidus or hypopituitarism.
They are well circumscribed,
spherical or ovoid lesions arising from the pars intermedia,
with midline location and they can be purely intrasellar or have suprasellar extension,
the latter slightly more common.
They are often identified between the anterior and posterior pituitary gland in the region of the pars intermedia.
They contain serous or mucoid material,
which determines their radiological appearance.
Thus the CT attenuation is low (serous) with low T1WI intensity (Fig. 20) or intermediate CT attenuation (mucoid) with hyperintense T1WI.
They rarely enhance with contrast administration,
but a thin peripheral rim of enhancement has been attributed to inflammation,
squamous metaplasia of the cyst wall or to a circumferential rim of displaced pituitary gland (Claw sign).
If a nodule is found inside,
it must not enhance to contrast administration.
If the patient is asymptomatic with normal endocrine function they may be safely ignored.
When symptoms are present,
surgical drainage and partial excision is necessary.
They have a low rate of recurrence.
Pituitary apoplexy
Pituitary apoplexy is a clinical diagnosis of an acute syndrome of pituitary hemorrhage or infarction.
It is an emergency with possible repercussion to the patient’s vision and life.
Symptoms include headache (the most common,
up to 95% of patients),
hypopituitarism,
vision loss,
fatigue,
ophtalmoplegia,
altered mental status,
emesis or diabetes insipidus.
This condition is often associated with a preexisting pituitary lesion,
most commonly a macroadenoma (outgrows the arterial supply) and patients are unaware of its existence until the event.
This coupled with the lack of direct arterial blood supply to the anterior pituitary create a vulnerable place for ischemic events and also hemorrhage.
There are other theories,
as the pathophysiology is incompletely understood.
The mean age of onset is 57 years with a male to female ratio of 2:1.
Imaging can show an enlarged pituitary.
Hyperdensity of the pituitary is not specific since other conditions can have the same imaging appearance.
Clinical information is decisive.
The pituitary mass can have a patchy or confluent hyperdensity due to a hemorrhagic component in most of the lesions on non-enhanced CT.
In one study,
identification of a pituitary tumor was possible with CT in 94% of cases,
but features of hemorrhage or infarction were only detected in 46% of cases.
Contrast administration may show a rim of enhancement that can be suggestive (but not diagnostic) of pituitary apoplexy.
CT does not differentiate cystic or degenerative changes from previous hemorrhage,
something that can be done with MRI.
CT may be normal.
MRI better depicts both a pituitary lesion and the hemorrhagic degeneration with a sensitivity of 88-90%.
We have to be aware when imaging in the hyperacute stage,
when we may not have signal alteration on MRI.
In the acute phase the lesion appears isointense on T1 and hypointense on T2.
Subacute phase will show an hyperintense lesion on T1 and on T2 (Fig. 8) (Fig. 9) and in the chronic phase both T1 and T2 will appear hypointense.
As in CT,
gadolinium administration can show slight contrast enhancement (Fig. 10).
T2 weighted gradient-echo MR imaging is sensitive for identifying brain hemorrhage and can show acute and chronic hemorrhages not seen with conventional techniques.
Restricted diffusion may appear in the setting of solid infarcted components and may be an early sign of apoplexy.
A fluid-debris level because of sedimented red blood remnants suggests late subacute hemorrhage with high specificity for pituitary apoplexy.
Treatment can be done with steroids and electrolyte replacement with surgical decompression showing the best results.
Pituitary metastasis
Pituitary metastasis have an incidence ranging from 0.14 to 28% with the highest percentage observed in autopsy series.
Breast metastasis are the most common,
identified in 6-29% of breast cancer patients.
Other reported cancers that provide metastases to the pituitary are lung (Fig. 11),
prostate,
melanoma,
pharynx,
pancreas,
thyroid,
larynx,
renal,
liver and ovarian.
7 cases of hepatocellular carcinoma have been reported.
They happen mostly in patients that have bone metastases or systemic dissemination.
Symptoms are rare.
Metastases are more prevalent in the posterior pituitary because of direct systemic blood supply leading that can lead to diabetes insipidus.
The patient’s history helps to differentiate metastases to lesions with similar characteristics,
usually macroadenomas,
but they can also be the initial presentation in patients with unknown primary tumors.
On MRI they can frequently look like pituitary macroadenomas,
but metastases grow faster.
Characteristically they are isointense on T1 and T2 weighted imaging (Fig. 11) or they can be hypointense on T1 with variable intensity on T2.
They enhance with contrast (Fig. 13).
They can cause thickening of the infundibulum and destruction of the sella turcica instead of enlargement or remodeling,
suggesting more than an adenoma (Fig. 13).
A dumbbell shaped appearance has been described due to indentation by the diaphragm sella.
Evaluation of with MR spectroscopy,
especially in lesions with a diameter greater than 20 mm can be useful.
Micro and macroadenomas can manifest as hypermetabolic foci on FDG-PET so they must be considered in the differential when evaluating for brain metastases.
Management includes surgery,
radiosurgery,
radiation,
chemotherapy,
medical therapy and endocrinological replacement and the prognosis is related to the histological subtype and stage of the primary malignancy.
Pituitary abscess
Pituitary abscess is rare but potentially life threatening,
representing less than 0.6% of patients undergoing surgical treatment,
associated with sepsis,
sinusitis,
osteomyelitis of the sphenoid bone,
cavernous sinus thrombophlebitis,
meningitis or surgical interventions.
They can form in a pre-existing lesion like an adenoma,
craniopharyngioma or a Rathke cleft cyst.
Other risk factors are an underlying immunocompromised condition and irradiation of the pituitary gland.
The most common symptom is headache followed by visual disturbances.
Some patients experienced meningism.
Patients may exhibit signs of infection with fever and leukocytosis,
but some series estimate only 24-33% of patients will have clear signs of infection,
so even in their absence,
they must be considered in the differential.
Anterior hormone deficiencies or diabetes insipidus can also be present.
Computed tomography shows a homogeneous,
low density mass with rim enhancement at contrast administration.
MRI also shows a sellar mass with central hypodensity or isodensity,
heterogeneous center and also a rim enhancing periphery after gadolinium administration (Fig. 15).
The lesion is hypo or isointense on T1 and exhibit high signal intensity in T2.
As expected,
there is restricted diffusion,
but the closeness of the adjacent bone limits its usefulness (Fig. 16) (Fig. 17).
The differential diagnosis must include adenoma,
carcinoma,
arachnoid cyst,
colloid cyst,
Rathke cleft cyst,
craniopharyngioma and metastasis.
Trans-sphenoidal decompression provides the most effective treatment,
improvement in visual fields may be seen in 80% of patients.
Management with craniotomy shows less favorable results.
Hypophysitis
Hypophysitis comprises the spectrum of inflammatory diseases of the pituitary gland and can be primary and secondary.
The former,
also known as lymphocytic hypophysitis is a rare autoimmune disorder most often seen in women in the peri or postpartum period.
Secondary hypophysitis refers to inflammation secondary to systemic inflammatory processes (infectious agents,
systemic disease).
Primary hypophysitis can be classified in three main histological categories: lymphocytic,
granulomatous and xanthomatous,
with lymphocytic being more than twice more common than granulomatous and xanthomatous.
Clinical symptoms usually include diabetes insipidus,
amenorrhea,
hypopituitarism,
headache,
mass effect and visual impairment.
It can be misdiagnosed as adenoma but the development of symptoms is faster in hypophysitis.
Lumbar puncture can reveal monolymphocitic cells in the cerebrospinal fluid.
MRI findings include the appearance of a sellar mass with an abnormally thickened infundibular stalk.
Both avidly enhance with contrast media.
The enhancement is homogeneous in appearance (unlike adenomas) both on pre and post gadolinium images.
Frequently there is loss of the brightness of the neurohypophysis (Fig. 18).
Cystic components and extension of the lesion to the base of the hypothalamus have been described.
Differentiating adenoma from hypophysitis is one of our main goals since treatment is different.
A radiologic score evaluating the posterior pituitary bright spot,
pituitary mass volume and symmetry,
stalk size and mucosal swelling within the sphenoid sinus,
allows correct classification between the two in more than 95% of cases.
There is a small association of aseptic meningitis and internal carotid artery occlusion.
Treatment is high dose corticosteroids and hormone replacement therapy.
For refractory patients,
the procedure of choice is transsphenoidal surgery for biopsy,
decompression and partial resection of the mass.
Germinomas
They are the most common intracranial germ cell tumor.
Together with Langerhans cell histiocytosis,
they are the most common causes of central diabetes insipidus in the pediatric age group.
Germ cell tumors in general comprise just 0.15% of lesions in major transsphenoidal series.
They also present with hypopituitarism and visual symptoms.
They can also produce idiopathic diabetes insipidus with no evidence of a focal lesion on MRI or just infundibular thickening.
Serum CSF β-human chorionic gonadotropin and α-fetoprotein aid in diagnosis and sometimes obviate biopsiy.
They are well demarcated,
homogenous lesions,
that because of their high cellularity,
they are hyperdense in CT and can be hypointense in T2 relative to brain parenchima,
although both intrinsic T1 and T2 signals are variable with isointensity being more comon.
Tumor cysts and hemorrhage are rare.
They avidly enhance with contrast material (Fig. 19).
Multimodal intervention is frequently required and is based on the histological tumor components with endoscopic endonasal transsphenoidal approach used for biopsy or resection.
Pituitary lymphoma
Primary central nervous system lymphoma represents 1% of primary brain tumors in immunocompetent patients and 3% in patients with acquired immunodeficiency syndromes.
The incidence in immunocompetent patients has been increasing for unknown reasons.
Most of them are non-Hodgkin B-cell type.
Sellar lymphoma is exceedingly rare and is more frequently found as part of more widespread central nervous system disease.
Primary intrasellar lymphoma can present with fever,
endocrine dysfunction,
cranial nerve paresis and diabetes insipidus.
It can simulate pituitary apoplexy.
MRI is the modality of choice.
Iso to hypointense T2 signal and reduced diffusion are typical features of lymphoma,
result of hypercellularity and high nucleus to cytoplasm ratio.
They tend to enhance homogeneously in the brain,
but in the sellar region they have been described as enhancing more heterogeneously (Fig. 14).
They are often erosive,
showing infrasellar,
suprasellar and cavernous sinus invation.
FDG PET/CT can be useful in diagnosis.
Surgery is done for biopsy purposes and tumor debulking,
especially for patients with visual loss.
Radiation and chemotherapy are the main treatment.
If the patient has primary intrasellar lymphoma,
transsphenoidal resection is possible.
Langerhans Cell Histiocytosis
Rare disorder of unknown etiology that has an initial presentation of central diabetes insipidus followed by a deficit in at least one anterior pituitary hormone.
MRI findings include abnormal posterior pituitary bright spot,
diffuse or focal thickening of the infundibulum or more rarely,
thinning.
Hypointensity in T1 and hyperintensity in T2 is seen on MRI with homogeneous contrast enhancement.
This findings are not specific.
Pituitary hyperplasia
Pituitary hyperplasia is a nonneoplastic enlargement of one or more type of adenohypophyseal cells that results of enlargement of the gland.
The cells don’t have a major qualitative change in their biology.
It is reversible if the cause is corrected.
It can cause mass effect and clinical changes related to the enlarged cells.
It can also be physiologically normal,
as during pregnancy or lactation.
Imaging characteristics can simulate a pituitary adenoma,
but hyperplasia enhances homogeneously and the enlargement is symmetric.
Medical treatment of the cause is often enough,
if the mass effect is severe,
surgery for biopsy and decompression are performed.
Pituitary carcinoma
With an incidence of 0.2% of symptomatic pituitary tumors,
they arise from the anterior pituitary and can be hormonally active.
They are indistinguishable from adenomas and diagnosis is difficult,
often following prolonged treatment.
To establish the diagnosis,
metastases inside and outside of the central nervous system must be found.
Clinical presentation is usually recurrence after initial pituitary surgery,
rapid local growth and tumor extension.
Imaging presents as invasive macroadenomas with no distinguishing features.
The focus is to detect metastasis and disease progression.
Depending on the subtype of the tumor,
nuclear medicine studies are sometimes useful.
It is important to exclude the presence of metastatic tumor elsewhere before making the diagnosis of pituitary carcinoma.
Management is surgery,
external beam radiotheraphy and adjuvant medical treatment.
It is mainly palliative.
Pituicytoma
Previously known as infundibuloma or posterior pituitary astrocytoma,
it is a new pathological entity defined by the WHO in 2007 classified as low grade (WHO Grade 1) and originates in the neurohypophysis.
Symptoms include visual disturbanes,
headache,
decreased libido,
fatigue,
amenorrhea and seizures.
CT shows a heterogeneous mass in the sellar and suprasellar regions with contrast enhancement.
MRI shows a well circumscribed,
solid,
posteriorly placed intrasellar mass with suprasellar extension.
They are typically isointense to gray matter on T1and iso to hyperintense on T2.
They can appear separate from the anterior pituitary gland and show fairly homogeneous enhancement on post contrast T1.
Pituicytomas are amenable to surgical resection.
Aneurysm
It is important to exclude a vascular lesion prior to interventions and because of its high morbidity.
They can mimic a cystic pituitary mass.
Aneurysms of the sellar region usually originate from the cavernous or supraclinoid portions of the internal carotid artery and account for approximately 2-10% of all cerebral aneurysms.
Diagnosis can be made with CT,
MR or conventional angiography.
Usually they can be adequately visualized with non-invasive imaging.
MR is usually diagnostic because of its ability to detect flowing blood,
it may show a “flow void” signal.
Partialy thrombosed aneurysm may cause confusion,
because of lack of perfusion in the thrombus with a heterogeneous signal intensity on T1 and T2 weighted images.
There can be calcification of the wall of the aneurysm and even remodeling of the sella turcica.
Management depends on location,
morphology and rupture status,
and treatment options include endovascular coiling procedures,
flow diversion,
surgical bypass and trapping and direct surgical clipping.
Meningiomas
Meningiomas are slow growing extra axial tumors originating from the arachnoid cap cells of the dura.
They are the most common nonglial primary brain tumor,
representing 15% of all intracranial neoplasms.
Aproximately 5-10% are in the sellar region.
They usually generate mass effect (changes in vision or signs of elevated intracranial pressure with pituitary disfunction).
It has been reported that they can mimic a pituitary apoplexy.
They are typically benign.
Being a non pituitary sellar lesion,
they can arise from the diaphragma sella,
tuberulum sellae,
planum sphenoidale,
lesser wing of the sphenoid,
anterior clinoid,
clivus,
cavernous sinus or optic nerve sheath.
Intrasellar meningiomas are rare.
Their size is variable.
Meningiomas are dural based lesions,
the dural attachment can be seen as the “dural tail” sign on MRI.
On non enhanced CT they tend to be hyperdense compared to brain parenchyma with some of them showing intramural calcifications and with intravenous contrast,
like in MRI,
shows strong,
uniform enhancement.
In MRI they vary from isointense (more common) to hypointense on both T1 and T2 weighted MRI although the T2 signal can be variable depending on the protein and water content of the tumor.
They show intense homogeneous enhancement.
This can help in the differentiation with a pituitary macroadenoma,
because as we mentioned earlier the enhancement tends to be inhomogeneous in a macroadenoma,
but beware,
up to 10% of meningiomas can also be inhomogeneous due to necrosis,
scarring,
cystic degeneration or calcifications.
Meningiomas tend to constrict the carotid lumen,
something adenomas rarely do.
Also looking for the dural tail sign and a suprasellar rather than an intrasellar tumor epicenter is important.
Dynamic contrast enhanced MRI can help to distinguish from neurinomas; meningiomas show early enhancement while neurinomas present gradual enhancement.
Angiography shows a tumoral blush and is used for preoperative embolization to reduce intraoperative blood loss.
Surgical approach is dictated by the relationship to the neurovascular structures.
There is a possibility of recurrence more than a decade after surgery; radiological surveillance is recommended.
Empty sella syndrome
Incidental finding of no clinical significance caused by an incomplete or absent diaphragm sellae which permits arachnoidal herniation inferiorly into the sella turcica.
MRI shows a flattening of the pituitary with occasional optic chiasm prolapse into the sella.
Etiologies include elevated intracranial pressure,
and pituitary atrophy.