Imaging,
particularly CXR,
the fist instance imaging modality in this kind of clinical practice,
is essential during the follow-up of patients undergoing thoracic surgery,
especially in the early phase.
CXR,
performed bedside supine in the very first postoperative days and,
as soon as possible,
upright in the two orthogonal incidences,
allows monitoring of the evolution of physiological findings,
as well as identifying “not expected” signs,
even without clinical suspicion: for this purpose,
the retrospective analysis of serial X-rays as an extreme utility.
However,
CXR has relatively low sensibility and specificity so,
in case of findings inconclusive or discordant with the clinical setting,
chest CT allows an accurate diagnosis in most of the cases.
Particularly multidetector CT (MDCT) scans enable the use of ultrathin section thickness (≤1 mm),
that exalts spatial resolution and permits high-quality multiplanar (MPR) and 3D reconstruction [4].
Therefore it is necessary that Radiologists know how to recognize “expected” and “not expected” findings,
in order to detect even subtle modifications of radiographic picture that may represent a complication and to provide indications to continue the diagnostic iter and the treatment planning properly
 .
.
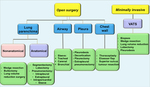
Surgical procedures in thoracic surgery and “expected” postoperative findings
Chest surgical procedures are several and can be divided into two main categories,
which are open surgery,
performed by way of thoracotomy,
and video-assisted thoracic surgery (VATS).
VATS is a minimally invasive surgical technique,
mainly used for biopsies,
for treatment of pneumothorax and recurrent pleural effusion and for removal of peripheral low stage lesions (Table 1).
In this work we are going to describe the major lung resection procedures [5]:
- Pneumonectomy (intrapleural,
extrapleural,
intrapericardial and “sleeve-pneumonectomy”);
- Lobectomy;
- Segmental resections (segmentectomy);
- Nonanatomical resections (wedge resections);
- Lung resections associated with bronchial or bronchovascular anastomosis (“sleeve-lobectomy” e “double sleeve resection”);
- Tracheobronchial resections;
- Bullectomy.
Lung resections themselves can be distinguished in anatomical (with ligation of segmental artery and vein,
closure of segmental bronchus and removal of the lymph nodes) and nonanatomical,
in which big staplers are used to resect “geographical areas” of lung parenchyma.
In the management of lung cancer,
the procedure selection depends mainly on the extent of cancer and on its anatomical relationships with central bronchi and large mediastinal vessels,
but also on patient’s clinical conditions (comorbidities,
cardiac and functional respiratory reserves).
Surgical flaps are used in thoracic surgery to strengthen bronchial anastomosis potentially at risk of leakage,
increasing blood flow and promoting healing; they are commonly used in case of sleeve-resections,
pneumonectomy or after neoadjuvant therapies,
to repair tracheoesophageal,
bronchopleural and esophagopleural fistulas or esophageal rupture,
to strengthen chest wall after extended bone resections and to prevent or contain pleural empyema.
Their knowledge is essential because flaps may simulate a pathological condition in an early postoperative phase or a recurrence of cancer later.
The most common surgical flaps are the pedicle muscle ones,
which appearance and localization primarly depend on the type used; they are the intercostal muscle flap,
the serratus anterior one,
the latissimus dorsi one,
the pectoralis major one and the rhomboid muscle flap; other flaps are pericardial or omental flaps and microsurgical free-flap [6].
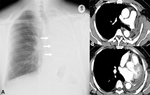
- Intercostal muscle flap: it originates along the posterior chest wall and extends towards the resection site; it appears as a soft-tissue thickening along the spine and mediastinum or directly across the thoracic cavity; it may contain calcifications,
often linear,
oriented along the long axis of the flap,
that can be seen within a few weeks after surgery and can increase over time.
- Serratus anterior muscle flap: it is flat and fan-shaped and it is obtained completely detaching muscle from the ribs anteriorly and the scapula posteriorly; to facilitate the entrance into the thoracic cavity,
a small part of the II or the III rib can be resected.
Its use results in some impairment of arm abduction of more than 90° and in ipsilateral scapular winging.
This flap localizes in the apex of the thoracic cavity.
- Latissimus dorsi muscle flap: it is less frequently used,
mainly when obliteration of surgical cavity with vascularized tissue is required,
such as in case of empyema or bronchopleural fistula or when previous flaps have failed; it requires an extensive dissection but because of its large size,
the muscle can be easily transposed to any site within the thoracic cavity.
The flap extends from the lateral chest wall through the thoracotomy incision to the resection site.
It results in mild shoulder impairment.
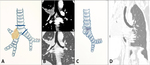
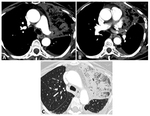
At imaging,
thoracic muscle flaps appear as homogeneous opacity with lengthened morphology at CXR and as structures that show muscular fibres mixed to fat-containing areas in their context at CT; the amount of fat usually increases over time,
maybe because of the atrophy and fatty replacement of the muscle fibres,
particularly in the serratus anterior muscle flap (Fig. 1).
Pneumonectomy consists of surgical excision of an entire lung and its lymph nodes and is indicated in case of lung cancer involving proximal vessels or main bronchi.
The overall incidence of complications is higher (20-60%) than other thoracic surgical procedures,
although mortality (6-8%) has decreased during last decades [7].
Depending on disease extension and type of pathology,
four different types of pneumonectomy can be carried out,
with different possible complications [5]:
- Intrapleural pneumonectomy - the most frequently used – consists of resection of the lung and its visceral pleura; it is performed in case of lung cancer without chest wall or pericardial invasion or in case of late-stage non-neoplastic disease,
such as tuberculosis or bronchiectasis;
- Extrapleural pneumonectomy involves en-bloc resection of the lung,
parietal and mediastinal pleura,
and,
if necessary,
pericardium and ipsilateral hemidiaphragm; it is employed in case of resectable pleural mesothelioma or tubercular empyema;
- Intrapericardial pneumonectomy involves resection of the lung and its visceral pleura with opening of pericardium,
then repaired with a mesh; it is performed in case of pericardial invasion or intrapericardial ligation of pulmonary artery and veins;
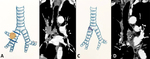
- Sleeve pneumonectomy is rarely performed,
in case of a bronchogenic carcinoma with carinal invasion; it involves resection of the lung and the carina,
with end-to-end anastomosis between the distal portion of trachea and the contralateral main bronchus (Fig. 2).
In case of cancer involving the ipsilateral main bronchus,
the contralateral main bronchus and the distal portion of trachea,
“tracheal sleeve pneumonectomy” can be employed,
but only in case of involvement of no more than 1 cm of the contralateral main bronchus and no more than 2 cm of the trachea.
After pneumonectomy,
the surgical cave,
at first replenished with nothing but air,
is gradually filled with fluid,
coming from passive transudation,
lymphatic vessels or small bleedings; the pleural fluid accumulation rate is extremely variable [8].
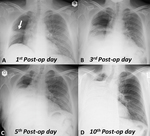
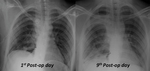
In normal circumstances,
CXR performed on 1st post-operative day should show a postpneumonectomy space filed with air,
a midline position or a slightly contralateral shift of the mediastinum,
normal expansion of the remaining lung.
A rapid mediastinal shift toward remaining lung,
especially in the immediate post-operative period,
is indicative of a complication,
such as atelectasis or abnormal accumulation of air and fluid in the postpneumonectomy space (bronchopleural fistula,
empyema,
hemothorax,
etc.); for this reason precociously recognizing this sign is essential.
The following serial radiographs should show the progressive appearance of pleural effusion in the lower part of the surgical space and,
in the upright CXR,
an air-fluid level gradually rising [9,
10].
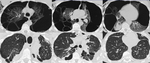
In most cases,
within the first 4 or 5 postoperative days,
approximately half of the postpneumonectomy space is filled with fluid,
with an air-fluid level expected in the mid-thoracic portion; fluids progressively rises and within the 10th-14th postoperative day most of the gas is reabsorbed with an almost complete opacification of the hemithorax; mediastinum is moderately attracted toward postpneumonectomy space,
with normal radiolucency of the remaining lung (Fig. 3).
Beyond the 14th postoperative day,
a progressive reabsorption of air and fluid in the space is observed; it happens during weeks,
up to 6 months,
in which the hemithorax is completely opacified but mediastinum is progressively shifting toward the postpneumonectomy space,
with a posterior rotation of the heart,
retraction of chest wall and elevation of the ipsilateral hemidiaphragm; the remaining lung is going to hyperdisted,
herniating across the midline to a position anterior to the heart and mediastinal big vessels [11,
12].
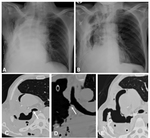
After months,
in 2/3 of cases CT scans show a fluid-filled space with a smooth fibrous rind,
while in the remaining 1/3 of cases the space is completely obliterated by fibrotic tissue and mediastinal structures (Fig. 4) [8]; a periosteal reaction of the ribs is also possible.
The extent of mediastinal shift is not constant,
depending on compliance and hyperexpansion of the contralateral lung,
which is larger in young people and women,
and on the rate of fluid reabsorption.
Late shift of mediastinal structures is also generally larger after right pneumonectomy,
due to the greater dimension of the postpneumonectomy space [9].
Lobectomy is the surgical resection of a single lung lobe with its regional lymph nodes,
with preservation of fissures: it is considered the optimal procedure for the treatment of surgically resectable lung cancer,
with a mortality rate of 2-4% [5].
After lobectomy,
the remaining lobes tends to rapidly hyperdistend.
“Sleeve lobectomy” is a particular type of lobectomy with different variants according to the localization (“right upper sleeve lobectomy”,
“left upper sleeve lobectomy” e “medium-inferior sleeve lobectomy”).
In this procedure the resection of a lung lobe is associated to resection and anastomosis of the airways; it represents an alternative to pneumonectomy in case of a central cancer involving not only the lobar bronchus but also the main bronchus [13,
14]; this procedure consists of resection of the main bronchus upstream of the lesions with an end-to-end anastomosis between the main bronchus itself and the part of the lobar bronchus not involved.
It carries a postoperative morbidity and mortality ranging from 2% to 11%.
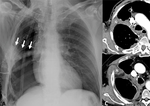
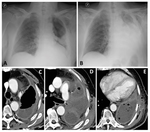
The right upper “sleeve lobectomy” is the most frequently performed,
representing 75% of cases (Fig. 5) [5].
“Double sleeve resection” or “bronchovascular sleeve resection” is performed to avoid pneumonectomy in case of cancer involving not only the common airway but also the main pulmonary artery; it implies both bronchial and vascular anastomosis [4].
After lobectomy,
CXR performed during the 1st post-operative day should show a mild volume reduction of the operated lung,
with ipsilateral pleural effusion and pneumothorax; mediastinum is in midline position or only slightly shifted toward the operated lung.
Remaining lung parenchyma shows a more or less conspicuous reduction of the normal radiolucency,
due to hypoventilation and parenchymal contusions after surgery.
During the 1st postoperative week,
pneumothorax,
pneumomediastinum and subcutaneous emphysema of the chest wall or even of the supraclavicular space may be seen.
During the following postoperative days,
approximately until the 14th day,
a progressive accumulation of fluid in the surgical space is seen,
with sometimes an air-fluid level.
Beyond the 2nd postoperative week,
a gradual reabsorption of gas and fluid is observed,
with obliteration of the surgical space; in this phase mediastinum is slightly shifted toward the operated side; this volume reduction of the surgical space has the same radiographic appearance of a lobar atelectasis.
This picture is associated with a mild reduction of the operated hemithorax,
which varies depending on the resected lobe,
intercostal space narrowing and hyperexpansion of the contralateral lung.
Right-side lobectomy yields a greater reduction in hemithorax size and a more pronounced mediastinal shift compared to left-sided lobectomies,
with greater mediastinal shift after right lower lobectomy if compared to right upper lobectomy [15].
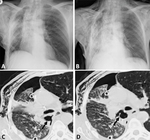
In case of sleeve lobectomy,
radiographic appearance is the same of a regular lobectomy,
while CT scans,
particularly MPR,
show an abrupt reduction in calibre,
up to stenosis,
of airway and,
in case of double sleeve lobectomy,
of airway and vessels,
at the level of the anastomosis (Fig. 6).
Segmentectomy envolves resection of a pulmonary segment or every anatomical resection less than an entire lobe,
with its regional lymph nodes.
Segmentectomies also include the basal segmentectomy (resection of all basilar segments),
lingulectomy and upper division left lobectomy,
sparing the lingula.
Segmentectomy can be performed via VATS,
with associated lower morbidity and shorter hospital stays.
Wedge resection is performed via VATS or,
more recently,
via robot-assisted thoracoscopic surgery and consist of the resection of a nonanatomical wedge of lung parenchyma containing tumour with a margin of adjacent healthy parenchyma; it is used for resection of small tumours and metastatic lesions; it is preferred to lobectomy in patients with low pulmonary reserve who are not eligible for lobectomy [4].
Radiographic appearance in patients undergoing wedge resection is similar to lobectomy.
Both CXR and chest CT scan may show the surgical suture line,
with or without associated consolidation of adjacent lung parenchyma; CT scan shows the resection at the level of a segmental bronchus,
sparing lobar bronchus.
As after lobectomy,
CXR frequently shows areas of reduced normal radiolucency,
due to hypoventilation and/or contusion of the parenchyma adjacent to the resection.
Nonanatomical wedge resection are performed mostly using surgical staplers,
with a suture line inside a part of parenchyma not surrounded by visceral pleura; for this reason nearly all patients experience some degree of air-leakage in the immediate postoperative period [8].
Due to the limited parechymal resection,
the tracheal shift from the midline position toward the operated lung is a sign of atelectasis [10].
Tracheobronchial resection consists of a limited resection of a bronchus or trachea,
with reconstruction of carena or a portion of trachea; this procedure is used in case of squamous cell carcinoma,
cystic adenoid carcinoma or iatrogenic tracheal stenosis.
Following this type of resection,
trachea appears shorter than normal,
with an indentation at the level of the anastomosis,
carena rises and bronchi may appear elongated.
Finally,
bullectomy consists of surgical removal of one or more subpleural bullae (measuring more than 1 cm),
generally followed by pleurodesis; the most common indication is the presence of subpleural emphysema with bullae associated with passive atelectasis of the adjacent lung parenchyma.
After bullectomy,
CT scan shows the surgical clips used for the suture and re-expansion of surrounding lung (Fig. 7).
Imaging of postsurgical complications
Postsurgical complications can be distinguished early complications (within the first 30 days from the surgery) and late ones (after 30 days) [3]; they more often occur in the early phase and their incidence depends on the surgical procedure and the clinical condition of the patient. Table 2 summarises major early and late complications.
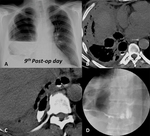
Persistent air leak is characterised by the persistence of pneumothorax,
pneumomediastinum and/or subcutaneous emphysema beyond the 7th postoperative day (Fig. 8).
The air leak is common after lobectomy and after segmentectomy/wedge resection; it occurs more frequently in case of patients with emphysema or with incomplete interlobar fissures.
Usually it is a transient complication,
but when associated with prolonged pneumothorax and/or pneumomediastinum (which require prolonged drainage) it is associated with an increase morbidity [5].
Anastomotic dehiscence occurs more often after sleeve lobectomy (in more than 6% of cases) [16].
An extended lymph node dissection increases the risk of bronchial wall ischemia and therefore anastomotic dehiscence; also an extended resection can result in excessive tension on the bronchial suture line.
The anastomotic dehiscence my lead to bronchopleural or bronchovascular fistula.
On CXR it is suspected in case of persistent air leak with increasing pneumothorax or increasing air-fluid levels in the surgical space [2].
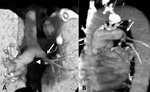
The most sensitive and specific CT finding for anastomotic dehiscence is detecting a defect in the bronchial wall; extraluminal air surrounding the anastomosis in equally sensitive but less specific for this complication [17] (Fig. 9).
An anastomotic dehiscence often requires convertion to pneumonectomy,
with an high rate of mortality.
Tracheo- or bronchopleural fistula (BPF) is a severe complication,
with a prevalence rate of 2-13% ad a mortality that ranges from 30% to 70%,
mostly due to aspiration pneumonia and subsequent ARDS [18].
BPF is more common after right pneumonectomy,
probably because of the anatomic features of the right main bronchus with a shorter and wider bronchial stump,
decreased degree of mediastinal coverage,
and greater propensity to ischemia due to its single bronchial artery.
In the early period it usually arises from of a faulty closure of the bronchial stump,
so it is preceded by an anastomotic dehiscence; it occurs more frequently between the 8th and the 12th postoperative day and the most significant risk factor is postoperative mechanical ventilation.
Delayed BPF is usually due to neoadjuvant radiation therapy,
infections or tumour recurrence.
On CXR,
BPF can be suspected in case of failure of the surgical space to fill with fluid,
in case of persistent and progressive air leak,
a drop in the air-fluid level exceeding 1,5 cm or the reappearance of an air-fluid level after a complete opacification of the hemithorax.
CT can identify fluid collections in the pleural space with air-fluid levels and demonstrate direct communication from an airway to the pleural space ( Fig. 10 ).
Pleural empyema occurs in 2-16% of cases of pulmonary resections,
with most works reporting an incidence rate less than 5% [5].
Mortality rates are quite high,
ranging from 16% to 70%,
especially if associated to a BPF [9].
BPF is the main cause of postpneumonectomy empyema,
identified in over 80% of patients.
In the early phase,
empyema can be attributed to intraoperative pleural contamination,
if lack of fistula is observed.
In delayed phase,
possible causes are BPF,
esophagopleural fistula and hematogenous spread; persistence of fluid in the surgical space increases risk of late-onset pleural empyema.
Most common radiographic findings suggesting for empyema are rapid surgical space opacification or hyperdistension of a complete opacified space,
with contralateral mediastinal shift; in case of late-onset empyema without BPF,
CXR findings can be only subtle or completely absent.
CT findings are convexity of the normally concave mediastinal border of the surgical space; irregular thickening with contrast enhancement of the visceral and parietal pleura,
separated by an empyema or exudative effusion (“split pleura” sign); air-fluid levels with a multiloculated aspect of the space (Fig. 11).
Air-fluid level may be also find inside chest wall due to the dissection of the empyema through the soft tissues of the chest wall (empyema necessitatis).
Hemothorax is characterised by very low mortality rates,
lower than 0.1% [5],
usually results from inadequate hemostasis of a bronchial artery or systemic vessels of the chest wall,
less frequently from ligature slippage from a major pulmonary vessel or unrecognized intrasurgical injury to a systemic vein [5].
On CXR,
it manifests as rapid increase of the pleural effusion till hemithorax opacification.
CT can identify a hemorrhagic effusion,
distinguishing it from a simple fluid one by its inhomogeneity,
with hyperattenuating areas and fluid-fluid levels.
Mediastinal hemorrhage may be due to the manipulation of the pulmonary artery and vein or injury to the intercostal,
internal thoracic or the azygos vein system vessels.
It represents a surgical emergency that has to be suspected in case of mediastinal widening,
confirmed on CT with the evidence of high-attenuation infiltration of the mediastinal fat.
Chylothorax usually occurs with a delay of up to 10 days after surgery [9],
due to injury to the thoracic duct or a main lymphatic vessel.
Chylothorax is diagnosed based on chylous leakage from the drainage tube,
with a triglyceride concentration greater than 110 mg/dl [19].
CXR can not distinguish chylothorax from fluid pleural effusion,
while the CT attenuation varies depending on its fat content,
that increases after a fatty meal,
reducing the effusion attenuation.
Atelectasis is commonly observed after lobectomy and other pulmonary resection; it results from retained secretions or lack of re-expansion of the remaining lung [2].
It occurs in 5%–10% of sleeve resections,
due to local edema at the anastomosis,
interruption of the ciliary epithelium and lymphatic vessels and partial denervation of the reimplanted lobe [20].
A prominent mediastinal shift toward the operated lung (or the contralateral one,
in case of pneumonectomy) is highly suggestive for atelectasis,
as well as the presence of an homogeneous opacity with a lobar,
segmental or subsegmental disposition.
Reporting the presence of collapsed areas is important because of the possibility of superimposed infection; atelectasis usually resolves within 2 or 3 days after bronchoaspiration.
Pneumonia of the remaining lung parenchyma after resection or of the contralateral lung after pneumonectomy has a reported incidence of 2-22%.
After pneumonectomy it is associated with a 25% mortality rate and is the leading cause of postoperative mortality [21].
The most common causes of postoperative pneumonia are aspiration of gastric secretions,
superimposed infection of atelectatic areas and BPF.
CXR shows patchy bronchopneumonic areas with reduced radiolucency,
with segmental or lobar distribution,
or less often homogenous opacities.
Prolonged infection course or large aspirations may result in severe necrotizing pneumonia or abscess formation.
Pulmonary edema is a severe complication,
that usually occurs in the 2nd-3rd postoperative day (2.5-5% incidence rate after pneumonectomy; <1% incidence rate after lobectomy or other limited resections); after pneumonectomy,
it has a mortality rate of 80-100% [22].
It is probably caused by fluid overload and hyperperfusion of the remaining lung,
as well as altered permeability of capillaries.
Predisposing factors include an excessive perioperative fluid load,
transfusion of fresh frozen plasma,
arrhythmia and low serum colloidal osmotic pressure [7].
Postpneumonectomy pulmonary edema more commonly occurs after right pneumonectomy,
probably due to an increased pulmonary blood flow through the left lung,
which normally receives only about 45% of the total pulmonary blood flow and contains about 45% of the total lymphatic capacity of the lungs and therefore it is not able to support the increased blood flow [22,
23].
In severe cases,
postsurgical pulmonary edema radiographically resembles adult respiratory distress syndrome,
with confluent alveolar opacities with air bronchogram,
while in less severe cases,
the CXR findings are those typical of the hydrostatic pulmonary edema,
such as ill-defined vessels,
peribronchial cuffing and Kerley B lines.
CT demonstrates interlobular septal thickening,
peribronchial cuffing,
and ground-glass opacities; in case of survival,
this findings disappear within a few days.
ARDS (adult respiratory distress syndrome) is a complication with a 5% incidence rate,
with mortality rate of 80% or more,
even higher during the postoperative period than overall mortality rate for all patients with this disease (65%) [24].
It is an increased permeability edema,
with irreversible damage of the alveolar-capillary barrier due to sepsis or high oxygen concentration during assisted ventilation.
The diagnosis of ARDS is based on severe refractory hypoxemia,
low pulmonary artery wedge pressure (<18 mmHg) and confluent alveolar opacities on CXR.
After lobectomy,
an asymmetric form of ARDS is possible,
with relative sparing of the postlobectomy lung [25].
Lung torsion presents a reported incidence of less than 0.4% and occurs when a lobe or the entire lung rotates 180° around hilar structures (although instances of 90° and 360° torsion have been described) [2].
The torsion compromises the airways,
the arterial blood supply and causes lymphatic and venous stasis,
resulting in interstitial pulmonary edema,
followed by alveolar effusion and hemorrhagic infarction in the affected parenchyma.
The lobar torsion more frequently involves the middle lobe after right upper lobectomy or the lingula after left upper lobectomy with sparing of the lingula itself.
On CXR lobar torsion should be suspected in case of a rapid lobar opacification,
with an increase in volume of the involved lobe,
sometimes with change in the vessel configuration or position of the opacified lobe on serial X-rays.
On CT scan,
the affected lobe shows an increase in volume,
with bulging of the neofissure,
ground-glass areas,
poorly enhancing consolidations,
interlobular and intralobular septal thickening; narrowing and occlusion of the lobar artery and vein with tapering or occlusion of the lobar bronchus (Fig. 12).
Intraperative pneumopexy can prevent this complication.
Cardiac herniation is a very rare,
usually early,
complication of the intrapericardial pneumonectomy with defective closure of the pericardium,
or after exposition of the intrapericardial portion of pulmonary vessels,
with a 40-50% overall mortality rate [7].
It typically manifests suddenly with shock,
often after a change in the patient’s position,
coughing,
extubation,
or connection of a drainage chest tube to a negative pressure source.
When the heart herniates to the right,
the heart rotates in a counterclockwise manner around the axis of the vena cava,
causing a superior vena cava syndrome; when the heart herniates to the left,
part of the heart protrudes through the pericardial defect,
producing a bulge in the cardiac contour with sharp demarcation by the pericardial edge [9].
Another sign on CXR is the presence of pneumopericardium inside of an empty pericardial sac.
Lung herniation is characterised by the protrusion of lung parenchyma through a chest wall defect; it is a rare but possible early complication – usually due to the thoracotomic or VATS suture line dehiscence – or more often late complication,
depending on chest wall muscle weakness joined to an increased intrathoracic pressure.
It can be lateral or posterior and can evolve to strangulation and infarction.
Esophagopleural fistula (EPF) is a very severe complications,
which occurs in 0.2%-1% of patients after pneumonectomy [7].
It is rarely an early complications,
usually due to an underestimated iatrogenic intrasurgical esophageal injury or parietal ischemia; it occurs more commonly as a delayed complication,
resulting from cancer recurrence with mediastinal invasion or chronic infection.
CXR findings are similar to those of a BPF with empyema,
with the reappearance of an air-fluid level in a previously opacified hemithorax; esophagography can show direct passage of the oral contrast medium from the esophagus to the pleural space,
with the leak generally located at the middle portion of the esophagus.
CT is the examination of choice,
because can directly show the fistula and its associated alterations,
such as empyema and aspiration pneumonia (Fig. 13).
Anastomotic stenosis is the most frequent complication after sleeve and double sleeve lobectomy,
with an 18-20% prevalence [14].
It may occur in the early phase due to secretion accumulation and perianastomotic edema or,
in the late phase,
due to exuberant granulation tissue,
partial anastomotic dehiscence with secondary healing and stricture formation or tumour recurrence.
MDCT scan,
with MPR,
MIP (“maximum intensity projection”),
MinIP (“minimum intensity projection”) e 3D reconstructions,
allows appropriate evaluation of location and features of both bronchial and vascular stenosis (Fig. 6).
Postpneumonectomy syndrome is always a delayed complication of pneumonectomy; it usually occurs in children,
young adults and women (probably because of the increased elasticity of the remaining lung in these patients),
generally within a year after surgery,
with an incidence rate of almost 1:640 pneumonectomies; this syndrome presents with dyspnea,
inspiratory stridor and recurrent pulmonary infections.
Most of the cases occurs after right pneumonectomy,
characterised by a displacement to the right and posteriorly of mediastinal structures,
with an anterior overexpansion of the left lung and a counterclockwise rotation of the heart along its major axis.
This displacement causes stretching and compression of the distal portion of the trachea and the left main bronchus,
that leads to stenosis and tracheobronchomalacia [26].
The treatment consists in the placement of silicone breast implants in the postpneumonectomy space.