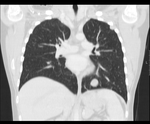
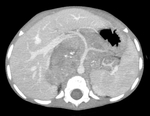
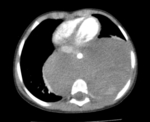
Diagnostic imaging of suspected Wilms tumour
Plain film has minimal contribution,
it may show bowel displacement and typical changes of adjacent ribs caused by compression by a slowly growing tumour.
Intravenous urography was used in the past and it may show distortion of the pyelocalicial system but it is not sufficient for diagnosis and staging.
Ultrasound (US) is a very useful diagnostic tool and imaging generally begins with ultrasound.
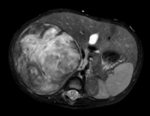
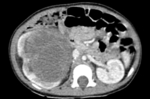
Computed Tomography (CT) and Magnetic Resonance (MR) are the gold-standard imaging modalities for diagnosing Wilms tumour (Fig.
1 and 2).
Wilms tumour imaging findings
Wilms tumour usually presents at ultrasound as an heterogeneous mass with hypoechoic areas due to necrosis and hyperechoic areas due to hemorrhage,
fat and / or calcification.
It is important to localise the mass to the kiney,
which at times can be challenging.
In many cases US,
CT and MR can demonstrate the "claw sign" wich refers to the sharp angles on either side of the mass with the surrounding normal parechyma indicating that the mass has arisen from this same parenchyma and not from an adjacent structure (Fig.
3).
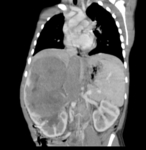
Vascular invasion occurs in approximately 5-10% and an effort should be made to detect potential tumour thrombus on the renal vein and inferior vena cava even on US examination (Fig 4.).
CT also shows an heterogeneous mass with soft-tissue density and hypodense necrotic areas (Fig.
3).
Calcifications may be seen in 10-15% of cases.
Administrataion of intravenous contrast wil show patchy enhancement and allows for better delineation of the relationship between the mass and the kidney.
Intravenous contrast also helps detecting nodal or hepatic metastases,
other foci of Wilms tumour and tumour extension into vessels.
Rarely there can be massive intra-tumoral hemorrhage or even rupture readily seen as spontaneous hyperdense material on unenhanced CT (Fig.
5).
MR usually shows a mass with low signal intensity on T1W and variable but mainly high signal intensity on T2W.
Like US and CT,
the mass is heterogeneous and may show areas of higher signal intensity on T1W due to necrosis and / or hemorrhage and areas of signal void due to calcifications.
Areas of necrosis also show higher signal intensity on T2W and non-enchancement after administration of gadolinium contrast.
The non-cystic components of the mass typically show restricted diffusion on DWI.
While the mass under study is readily seen on both unenhanced CT and MR with T1W and T2W sequences,
small multiple or bilateral tumour foci and liver metastases may be hard to detect and are often best seen after administration of intravenous contrast.
Staging of Wilms Tumour
Staging is the most important factor affecting prognosis and survival of patients.
The staging system currently used has been developed by the National Wilms Tumour Study (NWTS) and it relies heavily on post-operative histopathologic findings (Table 1).
The ability of CT to stage Wilms tumour accurately has been questioned mainly because it can be difficult to distinguish extension through tumour margins (which can be microscopic) and distinguishing reactive from metastatic regional lymph nodes.
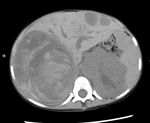
A CT of the chest is also recommended during staging since metastases occur more frequently in the lungs (85%) and 20% of cases have lung metastases at the time of diagnosis (Fig.
6).
Metastases are also most frequent in regional lymph nodes and in the liver (Fig.
5).
Both the North American Children Oncology Group (COG) and the European Societe Internationale D'oncologie Pediatrique (SIOP) are based on NWTS staging altough COG uses an upfront surgery approach and SIOP uses pre-operative chemotherapy on children over 6 month-old to reduce tumour size and the chances of intra-operative tumour spillage.
Both staging systems have proven value with almost equivalent clinical outcomes.
For children with bilateral disease several surgical options exist such as hemi-nephrectomy and wedge-ressection so each patient needs to be individualized to spare any normal renal parenchyma where possible.
When renal salvage procedures are impossible,
and bilateral nephrectomies are necessary,
they should be followed by renal transplant.
Adrenal and retroperitoneal Neuroblastoma imaging findings
Describing the whole clinical features and staging of neuroblastoma is beyond the scope of this exhibit but knowledge of imaging features is essential for distinguishing neuroblastoma from Wilms tumour.
Calcifications are very common on neuroblastoma occuring in 80-90% of cases and can be recognized on US but are better depicted on CT.
Calcifications may increase after pre-operative chemotherapy.
Plain radiograph may depict calcifications in 30% of cases and it is also important when suspected bone metastases wich are usually lytic and ill-defined altough rarely they can be sclerotic.
Either CT or MR may be used for Neuroblastoma diagnosis and staging depending on local availabilty and radiologist preference.
Currently there is no consensus about the optimal imaging modality although some consider MR to be preferable for determination of the organ of origin and assessment of regional invasion.
CT shows an heterogeneous mass frequently with calcification and areas of low atenuation due to necrosis and / or hemorrhage.
On MR the mass will show heterogeneous low signal intensity on T1W and heterogeneous hyperintense signal on T2W with necrotic areas more hyperintense on T2W.
Signal voids may be evident due to calcifications.
Intravenous contrast administration will depict heterogeneous variable enhancement on both CT and MR.
Neuroblastoma tends to encase vessels and insinuate itself beneath the aorta increasing the distance between the aorta and the vertebral column.
Adjacent organs are displaced altough in more agressive tumours there may be direct invasion of adjacent organs.
Adenopathies are more frequent in the renal hillum,
porta hepatis and retroperitoneum.
Imagiologic distinguihing features between Wilms tumour and adrenal or retroperitoneal Neuroblastoma
Even though both these tumours present as abdominal masses,
closely related to the kidneys,
in early childhood,
there are some clinical and imagiologic features that can help radiologists distinguish them.
Refer to Figures 7 and 8 for neuroblastoma typical features:
- Although both tumours occur in early childhood,
Wilms tumour has a peak incidence at a slightly older age (3-4 years old) than neuroblastoma (2 years old);
- Wilms tumour,
more often than not,
is a well circunscribed mass while neuroblastoma usually is poorly marginated and more frequently shows intrathoracic extension;
- Like previously mentioned,
the "claw sign" is a usefull sign that helps determine the origin of a mass and is commonly present in Wilms tumour;
- Wilms tumour displaces structures and vessels without insinuating between them or crossing the midline.
Neuroblastoma however,
is known for displacing and encasing structures and crosses the midline characteristically by insinuating behind the aorta;
- While Wilms tumour can invade and extend through the renal vein and inferior vena cava,
neuroblastoma usually encases and compresses vascular structures without invading them;
- Calcifications are less frequent in Wilms tumour (occurring in approximately 15% of cases) than in neuroblastoma (ocrurring in 80-90% of cases).

![Table 1: Wilms tumour NWTS staging system. References: Erginel B. Wilms Tumor and Its Management in a Surgical Aspect. In: van den Heuvel-Eibrink MM, editor. Wilms Tumor [Internet]. Brisbane (AU): Codon Publications; 2016 Mar. Table 1. [The NWTS staging system*]](https://epos.myesr.org/posterimage/esr/ecr2018/142652/media/737014?maxheight=150&maxwidth=150)