The most common thoracic findings in the ICU patients are pulmonary edema,
barotrauma,
atelectasis,
pneumonia,
aspiration,
pulmonary embolism,
pulmonary hemorrhage,
pleural effusion and pneumothorax.
Radiological findings in an ICU patient with acute lung disease may be nonspecific and it is essential to know the clinical history to establish the appropriate diagnosis.
1) PULMONARY EDEMA
Frequently,
pulmonary edema is classified as cardiogenic (due to cardiac insufficiency) and non-cardiogenic (due to fluid overload,
acute respiratory distress syndrome (ARDS),
toxic inhalation…).
But there is another classification of pulmonary edema that is correlated better with physiology and radiological findings [3]:
1.1.
Hydrostatic edema
1.2.
Permeability edema with diffuse alveolar damage (DAD).
1.3.
Permeability edema without diffuse alveolar damage (DAD).
1.4.
Mixed edema.
1.1.
Hydrostatic edema [3,
4]
-Physiopathology: it is produced by alterations in the normal relationship between intravascular and extravascular hydrostatic and oncotic pressures.
-Etiology: left heart failure,
left atrium obstruction,
pulmonary venous obstruction,
fluid overload in renal insufficiency,
hyperhydration,
hypoalbuminemia...
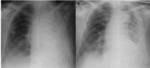
-Radiological findings: signs of interstitial edema,
alveolar edema or both.
1.1.1.
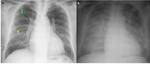
Signs of interstitial edema (Fig.
1a):
*Kerley-B lines: due to thickening of the interlobular septa.
They are seen as lineal horizontal opacities from 1 to 2 cm in length,
reach the pleural surface and are better seen at the costofrenic angles.
*Kerley-A lines: they are less frequently seen,
are oblique lineal opacities,
measure several centimeters in length and are seen in central or peripheral pulmonary areas.
*Subpleural edema: due to thickening of the subpleural interstitium.
It is recognized as a thickening of the fissures.
*Peribronchial cuffing: secondary to the thickening of the peribronchovascular interstitium that gives an appearance of thickening of the bronchial wall.
*Perihilar haze and loss of definition of the hilar vessels.
*Ground-glass opacities: secondary to the increase of intrapulmonary fluid.
1.1.2.
Signs of alveolar edema (due to extension of edema into the alveolar spaces) (Fig.
1b):
*Nodular or acinar areas of increased opacity that coalesce into consolidations which are usually bilateral symmetrical,
and predominantly perihilar.
*Air bronchogram may be present.
*Causes of asymmetric or unilateral alveolar pulmonary edema are: in the patient in decubitus edema accumulates in the most declining area; in the case of mitral insufficiency,
edema is accumulated in the right upper lobe; and if the patient has pulmonary emphysema,
the edema tends to accumulate in the respected areas.
-Evolution: As the liquid is relatively poor in proteins,
the edema presents a rapid onset and resolution.
1.2.
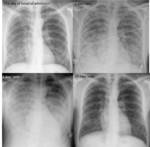
Permeability edema with diffuse alveolar damage (DAD): ARDS [4].
-Physiopathology: there is a lesion in the alveolocapillary membrane that leads to a loss of fluid and proteins in the pulmonary interstitium.
The ARDS represents the most severe form of edema with increased permeability associated with DAD (Fig.
3 and 4).
ARDS
There is a diffuse pulmonary lesion with progressive dyspnea and hypoxemia in a period ranging from hours to days.
The diagnostic criteria are:
1. Existence of a triggering pathology.
2. Hypoxemia despite high concentrations of inspired oxygen.
3. Bilateral alveolar infiltrates.
4. Exclusion cardiogenic pulmonary edema.
ARDS may have a lot of causes: it can be secondary to a pulmonary (pneumonia,
toxic fume inhalation…) or extrapulmonary injury (sepsis,
shock,
trauma…).
Acute interstitial pneumonia is a fulminating disease of unknown cause,
which usually occurs in previously healthy people.
It is associated with the histological pattern of DAD.
In ARDS,
there are several phases,
and they have a close radiological and pathological correlation [4] (Fig.
2).
-Evolution: slow resolution because it is an edema rich in proteins.
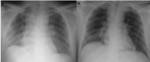
1.3.
Permeability edema without diffuse alveolar damage (DAD) [3,
4].
-Physiopathology: it occurs due to an increase in permeability without epithelial damage.
-Etiology: adverse reactions to drugs,
interleukin-2 treatments (Fig.5a-b),
transfusion reactions or pulmonary syndrome due to hantavirus,
toxic shock...
-Radiological findings: it usually resembles a hydrostatic pulmonary edema.
The thickening of the interlobular septa is the predominant sign in the majority of cases.
-Evolution: the edema resolves quickly due to the absence of epithelial lesion.
1.4 Mixed edema [3,
4].
-Physiopathology: it is an edema that occurs secondary to increased permeability and hydrostatic,
as a consequence of an increase in intravascular pressure and a vascular endothelial lesion.
-Etiology: neurogenic pulmonary edema,
edema due to high altitudes,
edema associated with tocolytic treatment,
post-transplant edema...
-Radiological findings: the findings are a mixture of the different types of edema and they are very variable.
-Differential diagnosis of the different types of pulmonary edema [4].
Although it is often impossible to distinguish between the different types of pulmonary edema,
the search of certain signs allows to help to the differentiation between hydrostatic edema and ARDS (Fig.
6).
2) MECHANICAL VENTILATION AND PULMONARY BAROTRAUMA.
Mechanical ventilation is frequently used in the ICU as a treatment of respiratory failure.
Pulmonary barotrauma is produced by the use of mechanical ventilation with high pulmonary pressures,
which can cause alveolar rupture and the subsequent development of pneumomediastinum,
subcutaneous emphysema,
pneumothorax,
air in the retroperitoneum or free air in the peritoneal cavity (Fig 7).
3) OTHER COMMON THORACIC PATHOLOGICAL FINDINGS IN THE ICU.
3.1.
Atelectasis.
Atelectasis are very frequent in the ICU due to neurological depression,
pain,
orotracheal intubation,
mechanical ventilation and supine position.
In X-rays,
they may be seen as an indistinguishable consolidation of pneumonia or aspiration,
unless there are signs of volume loss.
They can change very rapidly,
unlike pneumonias (Fig.
8).
Segmental or subsegmental atelectasis are very frequent.
They are seen as narrow and well-defined opaque bands of horizontal disposition,
They are usually in the lower lobes,
lingula or middle lobe,
usually due to hypoventilation or mucous impacts.
3.2.
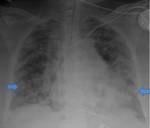
Pneumonia.
They occur in 10-20% of patients admitted to the ICU [4].
In X-rays,
they are seen as localized or diffuse zones of alveolar consolidation that appear and progress in days (FIg.
9).
On rare occasions,
they may show a rapid worsening in hours,
which is more typical of atelectasis,
pulmonary edema,
aspiration and hemorrhage.
3.3.
Aspiration of gastric content.
It is seen as patchy or homogenous consolidations of rapid appearance that become progressively confluent and predominate in the declining areas of the lung (posterior segments of the upper lobes and upper segments of the lower lobes in bedridden patients) [4].
3.4.
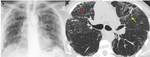
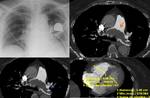
Pulmonary embolism.
It is included in the differential diagnosis of ARDS even though the diffuse consolidation characteristic of ARDS is rarely observed in the embolism.
In cases of doubt,
a CT angiography of pulmonary arteries have to be performed to distinguish among them (Fig.
10) [4].
3.5.
Pulmonary hemorrhage.
It may be occur in anticoagulated patients,
patients with hemorrhagic diathesis,
pulmonary vasculitis or Goodpasture syndrome.
Radiologically,
it manifests as rapid onset of diffuse bilateral consolidations and associate fall of the hematocrit.
The resolution can be slow but not as slow as ARDS [4].
3.6.
Pleural effusion.
It can be extremely difficult to distinguish from basal consolidations,
because in X-ray in decubitus the clamping of the costophrenic angles is usually absent,
and the only visible sign is often a poor definition of the ipsilateral hemidiaphragm or a homogeneous opacity of the lower hemithorax (which does not erase the parenchyma vessels) [4].
3.7.
Pneumothorax.
In the chest X-ray in decubitus,
the air is usually located in the anterior pleural space and is difficult to assess.
A subpulmonary pneumothorax with a visible visceral pleural line at the base of the lung may be seen.
Another signs of pneumothorax in decubitus chest x-ray are (Fig.11) [5]:
-Deep sulcus sign: the costophrenic angle may appear abnormally deep and radiolucent.
-Double diaphragm sign: visualization of the anterior costophrenic angle separated from the diaphragm but parallel to it.
-Better delimitation of the hemidiaphragm.
-Air in the minor fissure.
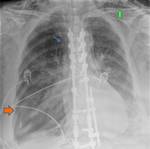
4) TUBES,
CATHETERS AND THEIR COMPLICATIONS.
4.1.
Orotracheal tubes.
Mechanical ventilation makes it necessary to use endotracheal tubes.
The distal end of the tube should be several centimeters above the carina (at least 2 centimeters).
Complications [4,
6]:
-A tube that is too low can enter the main bronchus,
causing hyperinflation of the lung,
alveolar rupture or pneumothorax,
and collapse of the contralateral lung due to the absence of ventilation.
Intubation of the left main bronchus is less frequent (Fig.
12).
-A tube that is too high may be located in the hypopharynx or the larynx,
causing poor ventilation or gastric distension.
-Hyperinflation of the endotracheal tube balloon may cause ulceration of the tracheal wall with perforation or late complications (tracheomalacia or tracheal stenosis).
-Tracheal perforation rarely occurs during intubation,
but when this happens the endotracheal tube leads to a pneumomediastinum,
pneumothorax or subcutaneous emphysema (Fig.
13).
4.2.
Tracheostomy tubes.
They are placed when the patient needs chronic mechanical ventilation or in case of upper airway obstruction.
The location of the tip of the tube is not important and depends on the placement of the tracheostomy stoma (it is recommended that the tip of the tube is placed half or two thirds of the distance between the stoma and the carina) [4,
6].
A small subcutaneous emphysema or pneumomediastinum after an uncomplicated placement of a tracheostomy tube may be normal [6].
4.3.
Pleural drainage tubes.
They are used to drain both pneumothorax and pleural effusions.
In the case of pneumothorax,
the tube should be in the least declining position of the pleural space,
and in the case of effusion the tube should be in the most declining position of the pleural space [4].
Complications [6]:
-If the tube does not work properly,
it may be because it is occluded,
bent,
lodged in the chest wall,
with its lateral hole outside the thoracic cavity,
or located in the greater fissure.
-Sometimes,
the lung can be punctured during the introduction of the tube (Fig.
14).
4.4.
Nasogastric tubes.
They are used to suction the gastric contents,
feed and administrate of medication.
The ideal position of these tubes is inside the stomach surpassing the cardia [6].
Complications [4,
6]:
-They can lodge in the bronchial tree (Fig.
15),
which can cause a perforation of the visceral pleura and a pneumothorax,
a bronchial obstruction and atelectasis or severe pneumonia due to the administration of nutrition to the lung.
-Bending of the nasogastric tube inside the hypopharynx can cause aspiration.
-Less frequent are pharyngeal,
esophageal or gastric perforations.
4.5.
Central venous catheters.
They are used to measure the central venous pressure or for the infusion of liquids.
They are placed by percutaneous puncture of the internal jugular or subclavian veins.
A catheter will have an appropriate location when it is in the superior vena cava.
It is usually not recommended that the catheter reaches the right atrium,
as it may cause arrhythmias or the injection of undiluted toxins into the heart [4].
Complications [7]:
-Pneumothorax is the most frequent complication because the pulmonary vertex is located about 5mm from the subclavian vein.
-Abnormal localization of the catheter in the azygos vein,
intercostal or internal mammary veins.
-Laceration of the subclavian or carotid artery.
-Mediastinal hemorrhage.
-Pneumomediastinum.
-Layering or loop forming.
-Fragmentation of the catheter with migration of the fragment causing arrhythmia,
vascular injury or pulmonary embolism.
-Venous thrombosis.
The formation of thrombi at the ends of intravenous catheters is frequent and does not involve any risk.
However,
the appearance of venous thrombosis or pulmonary thromboembolism is uncommon and requires removal of the catheter and anticoagulant therapy.
4.6.
Transvenous pacemarkers.
They may be temporary or permanent and are used to treat anomalies in cardiac conduction.
Transvenous placement is the choice for temporary pacemakers in the ICU,
and they are usually placed through the internal jugular or subclavian veins.
The electrodes of pacemakers are usually placed with the tip at the apex of the right ventricle pointing to the left,
anteriorly and inferiorly.
Bipolar pacemakers have a second electrode,
which is normally positioned in the right atrium [4,
7].
Complications:
-Pneumothorax and vascular injury may occur during transvenous insertion of the electrodes.
-Rarely,
pacemaker electrodes perforate the coronary sinus or myocardium,
causing pericardial tamponade or diaphragmatic stimulation (Fig.
16).
-The incidence of cable breakage has decreased nowadays.
4.7.
Pulmonary artery catheters (Swan-Ganz catheters).
They allow the measurement of the pressures of the pulmonary artery.
The catheter is inserted through the internal jugular or subclavian vein.
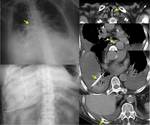
The ideal position of the catheter tip is in the right or left pulmonary artery and the tip of the catheter should not extend beyond the proximal interlobar pulmonary artery (2 centimeters of the hilum) (Fig.17) [4,
6].
Complications [7]:
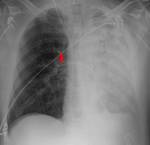
-If the catheter is placed very distally,
occlusion of a branch of the pulmonary artery may occur,
and if the inflated balloon persists or a thrombus forms around the distal tip of the catheter,
a pulmonary infarction may occur.
If this happens,
it will be seen as a consolidation near the end of the catheter without another cause that justifies it.
It will slowly decrease in size for several days,
leaving a residual scar.
-Other complications are: kinking,
loops,
pneumothorax or vascular injury.
4.8.
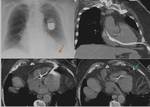
Intraaortic counterpulsation balloon pump.
Intraaortic counterpulsation balloon pump is an intra-aortic catheter with an inflated balloon (about 25cm long) that can be used to improve peripheral blood flow in patients with left heart failure or situations of low cardiac output.
It is introduced through the femoral artery until its tip is in the descending thoracic aorta 2cm distal to the origin of the left subclavian artery.
The balloon with sausage profile may be visible when inflated as a radiolucency within the descending aorta.
The catheter is difficult to see [4,
7].
Complications [7]:
-If the catheter does not progress enough,
it may cause occlusion of the left common carotid or left vertebral arteries resulting in cerebral ischemia.
It can also cause obstruction of the left subclavian artery (ischemia of the left arm).
-If the catheter is located too distally,
it may occlude the renal or mesenteric arteries.
-Aortic dissection may also occur.
-The rupture of the balloon with gas embolization is extremely rare.