Lung resection surgery is a procedure often performed as a treatment for lung cancer.
It is also employed in the treatment of other pathologies,
such as advanced pulmonary emphysema or bronchiectasis with abundant episodes of overinfection (1).
Pulmonary resection techniques include pneumonectomy,
lobectomy,
and partial resection.
The choice of each technique is based on the staging of lung cancer and the patient's respiratory function (2).
After lung resection surgery,
there are a range of changes in the hemithorax undergoing the procedure that are necessary to know for a correct evaluation of these patients.
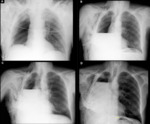
In the case of pneumonectomy,
the control chest radiograph usually shows a gradual opacification of the residual cavity.
First,
in the immediate postoperative period (day 1-2),
the pneumonectomy space contains air and a small amount of fluid and some congestion of the contralateral pulmonary vessels is noted.
During the first week,
an increase in the air-fluid level is observed up to half or two-thirds of the hemithorax.
After the first week,
the fluid continues to increase until the residual cavity is almost completely filled,
although there is often some residual air left in the vertex (Figure 1).
This process may last for weeks to months.
At the earlier stage,
the mediastinum remains centred,
and gradually moves into the residual cavity as fluid in the cavity is reabsorbed and the contralateral lung expands.
In one-third of cases,
the liquid is completely reabsorbed,
leaving a small residual cavity filled with air or occupied by fibrous tissue or mediastinal structures,
whereas in the remaining two-thirds a certain amount of residual fluid persists (1,
4).
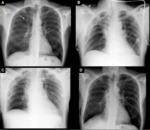
In the case of lobectomies,
apart from the presence of surgical clips,
the main change observed is the progressive loss of volume caused by the elevation of the corresponding hemidiaphragm and the slight displacement of the mediastinum,
followed by a re-expansion of the adjacent lobes.
This distorts the normal anatomy and makes it difficult to assess the resection performed without the complete series of radiographic images (Figure 2) (1).
Imaging techniques play an important role in the diagnosis and follow-up of lung resection surgery.
The radiograph is the technique of choice during the early postoperative period,
and the only one necessary if the postoperative period is normal.
However,
in case of complications,
a more sensitive technique such as contrast enhanced-CT should be performed (2).
COMPLICATIONS:
Early and late postsurgical complications may vary depending on the time that has elapsed after surgery (Table 1) (1,
3,
4).
EARLY COMPLICATIONS:
PULMONARY OEDEMA:
Postoperative pulmonary oedema is a life-threatening complication that develops 2-3 days after lung resection,
usually following pneumonectomy,
lobectomy,
or bilobectomy.
The causal mechanism,
although undetermined,
seems to be related to the increase in hydrostatic pressure and the permeability of the capillary barrier (1).
Predisposing factors include an overload of perioperative fluid,
frozen plasma transfusion,
arrhythmias,
marked postoperative diuresis and low serum osmotic pressure (3,
4).
Clinically,
it manifests itself as a rapid respiratory deterioration,
with dyspnoea and hypoxemia that is accompanied by pulmonary infiltrates,
interstitial oedema and consolidations (3).
Without adequate management,
it can evolve into Acute Respiratory Distress Syndrome (ARDS) (2).
Pulmonary oedema is an exclusion diagnosis and is only valid if there is no clinical or radiological evidence of other acute pathologies (4).
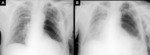
On serial chest radiographs,
findings of mild postpneumonectomy pulmonary oedema resemble those of hydrostatic pulmonary oedema,
with Kerley's lines,
peribronchial cuffing,
ill-defined blood vessels,
and pleural effusion (Figure 3).
In severe forms,
central or widespread pulmonary opacities are observed,
which,
if rapidly established,
can be indistinguishable from ARDS or aspiration pneumonia (1).
In pulmonary oedema,
with correct management,
these images tend to disappear within a few days (3).
PNEUMONIA:
Approximately 30% of pulmonary infiltrates in the intensive care unit are due to respiratory infection.
The absence of infiltrations in the radiograph does not exclude the possibility of pneumonia since the radiological signs of pneumonia appear after the clinical manifestations (3).
Frequently,
it appears in patients with mechanical ventilation,
or tracheobronchial secretion retention.
Its appearance on chest radiographs is very variable (1),
ranging from bronchopneumonia to lobar consolidation.
CT is especially relevant in patients with aspiration pneumonia due to a BPF since it allows a better assessment of this complication (4).
ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS):
This complication has a low incidence (5%) and a high mortality rate.
Its mechanism is still unknown,
but it is due to a severely damaged alveolus-capillary barrier (1,
4).
Diagnosis is established by the imaging features and the presence of a pulmonary artery wedge pressure less than 18 mmHg,
although this parameter may be mistakenly decreased after pneumonectomy (1).
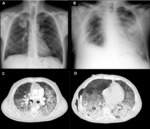
On postoperative radiographs,
a rapid and progressive opacity of the remaining lung may be observed,
along with a clinical deterioration.
CT depicts ground-glass opacities and patched pulmonary consolidations predominantly in declines areas and peripheral regions.
As it evolves,
CT shows an increase of consolidations,
with a gravitational anterior-posterior gradient,
and the appearance of tractional areas and subpleural and intraparenchymal cysts (Figure 4) (4).
ARDS may develop in combination with other postoperative complications such as pneumonia,
BPF or empyema (4).
Radiographic findings of pneumonia,
pulmonary oedema and ARDS may overlap,
and in fact,
the first two may coexist with the latter.
The absence of pleural effusion helps distinguish ARDS from pulmonary oedema (1).
HAEMOTHORAX:
Postoperative bleeding is an uncommon complication (less than 1%) after lung resection surgery (1).
It can occur in the early hours of the immediate postoperative period with significant haematocrit drop,
hypotension and the presence of intracavitary massive pleural effusion with a displacement of the mediastinum.
The appearance of any of these signs requires a quick surgical revision.
In the milder forms,
there is a progressive increase in pleural effusion,
which may go unnoticed as it overlaps with the usual evolutive findings in the normal postoperative period.
On non-contrast CT,
a pleural effusion with high attenuation areas (clots) can be observed,
which can be differentiated from pleural masses by their lack of enhancement in contrast-enhanced CT.
Occasionally,
it is possible to observe a haematocrit effect or the presence of a mass (clot) with a high attenuation filling the residual lobectomy cavity (Figure 5) (1,
3).
PERSISTENT AIR LEAK:
One of the best-known complications of lung resection is a persistent air leak.
It occurs when alveolar space communicated with the post-surgical cavity exists.
It is more frequent when there are incomplete or absent interlobar fissures.
Imaging techniques show a persistent pneumothorax,
pneumomediastinum and subcutaneous emphysema.
Those that originate in the pulmonary periphery,
usually resolve in the first 24-48 hours,
however,
if they do not do so in the first week they are called persistent air leakage (3).
EARLY BRONCHOPLEURAL FÍSTULA (BPF):
It is one of the most important complications of this type of surgery and its mortality varies from 30 to 70% (3).
Fistulas are the result of communication between the bronchial stump and the residual cavity.
It is most common after a right pneumonectomy,
due to increased length of the bronchial stump,
increased dehiscence rate,
and less mediastinal coverage.
BPF is also frequently associated with postoperative mechanical ventilation,
uncontrolled preoperative pleuropulmonary infection,
trauma and preoperative radiotherapy.
BPF is the leading cause of postpneumonectomy empyema.
In the early postoperative period,
BPF is rare and generally occurs in the first postoperative week,
usually due to the lack of adequate bronchial closure.
A late BPF is more common and is usually caused by infections or tumour recurrence on the bronchial stump (1,
3,
4).
The definitive diagnosis is usually made by bronchoscopy (3).
Plain radiographs show: a poor evolution of the air-fluid level in the hemithorax operated; a persistent or progressive pneumothorax,
subcutaneous and mediastinal emphysema despite the pleural tube functioning; a decrease in fluid level greater than 2 cm with mediastinal displacement to the contralateral side; a reappearance of a pneumothorax or recurrence of air in a previously opacified hemithorax (Figure 6) (3,
4).
In addition,
CT can show communication from the bronchial stump to the pleural cavity or the presence of small air bubbles around the bronchial stump (1).
EARLY EMPYEMA:
Its incidence is low due to current surgical techniques and available antibiotics.
However,
when it occurs,
it can have deadly consequences (4).
It occurs most often in the following situations: right pneumonectomy,
completion pneumonectomy after previous lobectomies,
preoperative radiotherapy,
prior pleural infection,
postoperative sepsis,
lymphadenectomy,
long bronchial stump,
and mechanical ventilation.
It can occur in both postoperative periods and even months and years after surgery (3).
In the immediate postoperative period,
it is usually related to a residual infection in the pleural cavity or intraoperative contamination,
although it can also occur secondary to a BPF or an OPF.
In the late phase,
it is usually secondary to the haematogenous spread of an infection or associated with late BPF.
In fact,
the risk factors associated with the onset of BPF also increase the risk of empyema.
Patients show signs of sepsis,
fever,
increased acute phase reactants and clinical deterioration (1,
4).
On plain radiographs,
a rapid filling of the residual cavity,
with a mediastinal shift towards the remaining lung,
is observed.
CT shows an irregular pleural thickening with atypically distributed convex pleural collections,
with enhancement after contrast administration (Figure 7) (3,
4).
Due to inflammatory changes in the residual cavity,
empyema may communicate with adjacent structures,
resulting in a BPF or in an empyema necessitatis,
when drainage is to adjacent muscle planes and skin.
In this context,
the appearance of a new air level may indicate the presence of a BPF or gas-forming organism.
BPF,
therefore,
can be both cause and consequence of empyema (4).
CARDIAC HERNIATION:
It is a very rare complication which requires urgent surgery.
In most cases,
it is due to a pericardial defect secondary to an intrapericardial pneumonectomy.
It occurs fundamentally in the immediate postoperative period.
A change of position,
coughing,
extubation,
rapid re-expansion of the remaining lung,
or an intrathoracic drainage tube can trigger this complication (1,
3,
4).
The defect side will determine the clinic: on the right,
there will be signs of obstructed venous return; on the left side,
can lead to arrhythmias,
myocardial ischemia,
and heart attacks (1).
When it occurs toward the left,
the radiograph shows a rounded-shaped left border with a marked cleft between the cardiac border and the large vessels; and a lateral or posterior apex displacement.
When it occurs toward the right,
a cardiac apex displacement to this side is observed.
In both cases,
the pericardial sac can be filled with air,
and there is often an abnormal displacement of the catheters (1,
4).
Possible immediate differential diagnoses include massive intrathoracic bleeding,
residual lung atelectasis,
and acute cardiac tamponade (4).
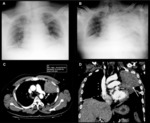
LOBAR TORSION:
It is a rare and serious complication of the immediate postoperative period.
It occurs after a lobectomy,
and most often in the middle lobe.
Clinically,
respiratory deterioration,
and sometimes fever and increased acute phase reactants are observed.
It occurs when a lobe rotates on its pedicle,
causing a torsion of the bronchovascular structures (1,
3).
The findings on plain radiographs are suggestive but not specific and may mimic atelectasis or intrapleural hematoma (1).
The CT with contrast allows a better characterization of this complication,
visualizing an obliteration of the bronchovascular pedicle.
Sometimes a suture malposition can be observed.
The affected lobe is enlarged and shows ground-glass opacities,
consolidations with no enhancement,
inter and intralobular septal thickening,
and a bulging of the fissure (Figure 8).
Recognition of this complication is essential since the absence or delay of surgery increases mortality (1,
3).
ANASTOMOTIC DEHISCENCE:
The performance of a lymphadenectomy in lung cancer surgery is a major cause of anastomotic dehiscence,
as it increases the risk of bronchial wall ischemia (3).
It is usually an early complication,
although it can also be late (1),
especially after a sleeve lobectomy,
and can lead to BPF or bronchovascular fistula.
The most sensitive and specific finding in CT is the presence of a defect in the bronchial wall (Figure 9) (3).
LATE COMPLICATIONS
POSTPNEUMONECTOMY SYNDROME:
It is a rare syndrome associated with pneumonectomies,
which involves the appearance of respiratory symptoms secondary to intrathoracic airway compression,
due to an excessive mediastinal displacement (3).
It usually occurs in the late postoperative period (around the first year) and manifests as breathlessness,
inspiratory stridor,
and recurrent lung infections (4).
It is more common in young people or women due to the greater flexibility of mediastinal structures (1,
3,
4).
Most occur after right pneumonectomy,
due to the increased volume of this hemithorax.
The left lung and mediastinum move to the right side and the trachea and left bronchus are compressed by the aortic arch and the left pulmonary artery anteriorly and by the vertebral column and the descending aorta posteriorly (1,
4).
On chest radiographs,
a pronounced displacement of the mediastinum in the pneumonectomy space will be seen,
with the cardiac apex in a posterolateral situation (1,
4).
CT is the technique of choice for obtaining information on the position of mediastinal structures and to determine the narrowing of the distal trachea and left main bronchus (3).
OESOFHAGOPLEURAL FISTULA:
It is a rare and serious complication following pneumonectomy,
that usually originates in the first two years after treatment (4).
It is mainly due to three mechanisms: surgical damage,
tumour recurrence or chronic infection (3,
4).
It must be confirmed by several tests such as chest radiographs,
oesophagoscopy,
oesophagography and bronchoscopy.
Radiographic findings are similar to those of BPF.
CT scans can rule out fistula and other abnormalities such as tumour recurrence,
empyema,
or aspiration pneumonia.
These patients should interrupt oral feeding and undergo reparative surgery to close the fistula and manage complications from it (3,
4).
LATE BRONCHOPLEURAL FISTULA:
Nearly half of all BPF occur early in the first few weeks after surgery.
In the late period,
it is usually due to tumour recurrence or infection.
Radiological findings are similar to those of early postoperative fistula (Figure 10) (1,
4).
LATE EMPYEMA:
This complication can occur from the first postoperative day for weeks or months after surgery (3).
As in the case of early empyema,
it is closely related to BPF.
In fact,
in this period the empyema is usually due to the existence of BPF or haematogenous spread of infection (1,
4).
It should be noted that in up to two-thirds of patients,
there is pleural thickening and a certain amount of residual fluid months after surgery,
without an infectious process.
For this reason,
in certain cases,
the diagnosis of empyema could be difficult (4).
CT is the imaging technique of choice,
as it will help identify empyema,
and assess the presence of BPF or EPF,
as well as other thoracic alterations (Figures 10,11) (1,
4).
In the absence of typical images,
diagnostic thoracentesis will be necessary (1).
PULMONARY ARTERY STUMP THROMBOSIS:
In patients suffering from lung resection,
thrombosis of the bronchial stump occurs with some frequency and may be identified incidentally in late postoperative.
Among the factors that influence its appearance,
the length of the arterial stump stands out (Figure 12) (1).
Normally these thrombi do not evolve,
although there is controversy as to whether or not they should be treated,
given the potential risk of pulmonary thromboembolism in pneumonectomized patients (1,
5,
6).
LOCAL TUMOUR RECURRENCE:
Tumour recurrence after curative surgery usually occurs within 2 years after surgery.
Local recurrence includes ipsilateral metastatic lymphadenopathies,
recurrence in the chest wall or parietal pleura,
as well as in the surgical margin (Figure 13).
CT is the technique of choice for assessing this complication (3).